Crystalline PV/RR
Access crystallization and formulation information at mL scale with the new Crystalline PV/RR

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Access crystallization and formulation information at mL scale with the new version of Crystalline PV/RR, launched in November 2022.
Designed by scientists, for scientists, the new Crystalline PV/RR features:
- AI-based image analysis
- Improved optics with resolution up to 0.63 microns per pixel
- Advanced analytical features
- Ready for robotic integration
Applications:
- Crystallization process optimization
- Form & habit control
- Particle size determination
- Formulation process optimization
- Stability of liquid formulations
- Solubility determination
- Polymorph, salts, cocrystals
With the new Crystalline PV/RR, every picture tells your story.
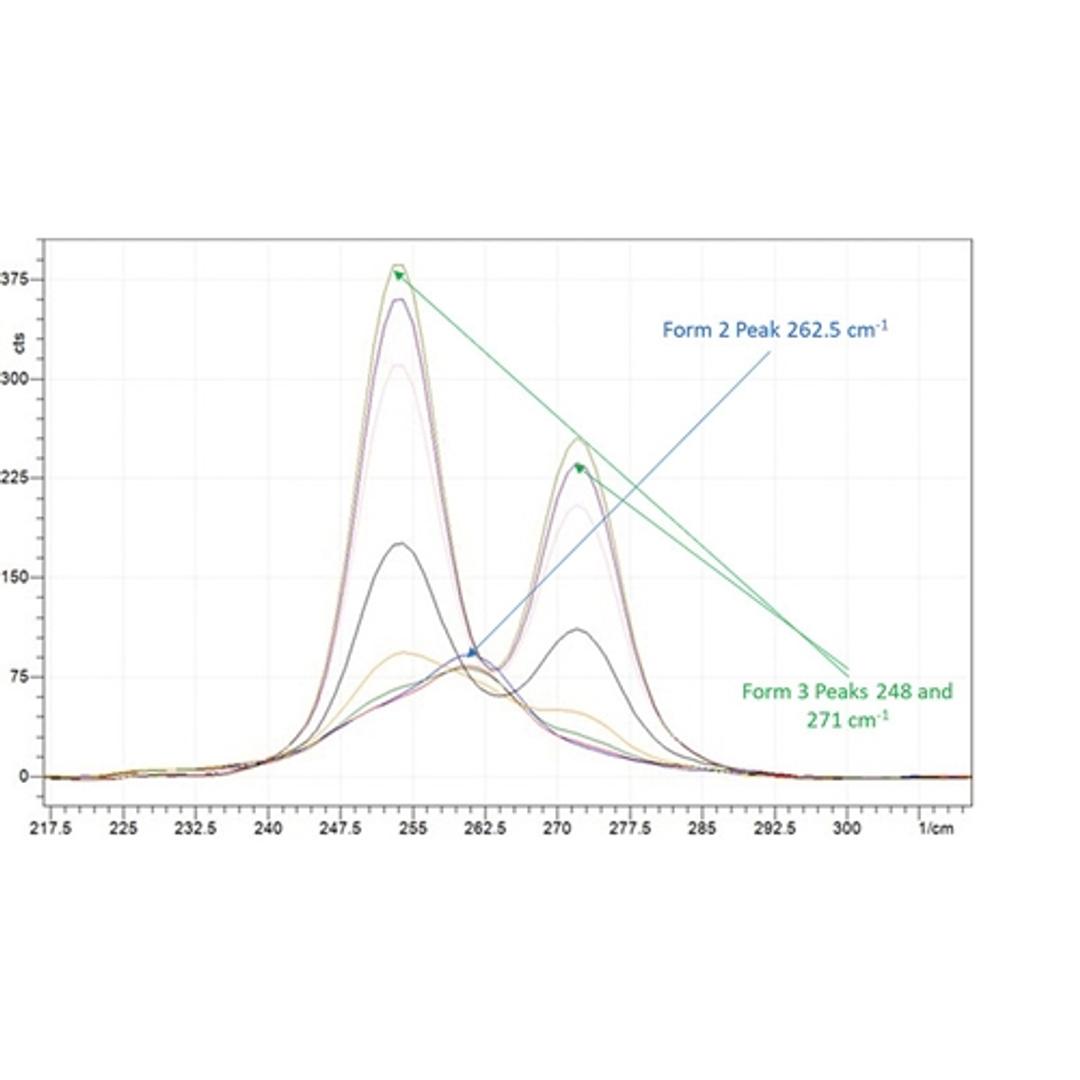
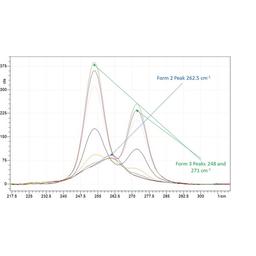
Tracking polymorph and crystal morphology using non-invasive in situ analysis at small scale
In this application note, Technobis Crystallization Systems demonstrate that by utilizing the Crystalline PV/RR device coupled with a Tornado Raman spectrometer, it is possible to monitor and track polymorphism and morphology at low working volumes.
Measuring secondary nucleation through a single crystal seeding approach
Crystallization is a common process in the pharma industry in the manufacturing of solid form doses. In this application note, Technobis Crystallization Systems introduces its novel, single crystal seeding procedure for measuring secondary nucleation. This promises to allow the rapid development of industrial crystallization procedures and facilitate a deeper understanding of the crystallization process.
In-situ crystallization monitoring with CrystallineRR and Raman spectroscopy
In this application note, Dr. Manuel Henry, from Boehringer Ingelheim laboratories explains how Raman spectroscopy and the CrystallineRR instrument can be used to identify polymorphic transformation.
Determining stability of protein formulations using the Crystalline series
This application note provides information on how the Crystalline series of instruments can obtain data on the stability of protein formulations by making use of its real-time digital camera.
Successful parental formulations by preventing crystallization
This application note provides a case study on how the Crystal16 and the Crystalline instrument can be used to produce successful parental formulations by preventing crystallization.
Physical stability of agrochemical formulations and observation of eutectics
Physical stability of formulations is an important parameter during formulation development, especially when testing multiple phase systems (suspensions or emulsions). In this application note, Dr. Martin Viertelhaus from BASF SE, explains the essentials of physical stability of formulations, covering topics such as stability testing, observation of eutectics and how the Crystalline instrument can be used to obtain this information.
Solubility: Importance, measurements and applications
This application note covers two dynamic methods for effective and reproducible solubility data generation: the temperature variation and solvent addition methods. These methods can be easily applied by making use of the turbidity probes integrated in the Crystal16 and particle viewer cameras of the Crystalline instruments.
Crystalline V2
In this video, Technobis Crystallization Systems presents the Crystalline V2.
Enhancing crystallization and formulation processes with new Crystalline PV/RR
Improved optics and state-of-the art software based on AI analysis for monitoring crystallization processes
Conduct more crystallization studies with less research material
Technobis Crystallization Systems launches new modules for benchtop crystallization platform to boost its experimental capabilities
Tiny crystals, big data
New modules for Technobis’s crystallization platform enable scientists to perform vapor diffusion and evaporation crystallization screening