Resources
25
Product Brochures
PAL RSI and PAL RTC - Sample prep and injection
Product Brochures
PAL3 Autosampler: Accessories & smart consumables
Application Note
Immunofluorescence resource guide
Product Brochures
Advancing innovation for smarter manufacturing
Application Note
IHC multiplexing resource guide
White Papers
VectaPlex™ Antibody Removal Kit (VRK-1000)
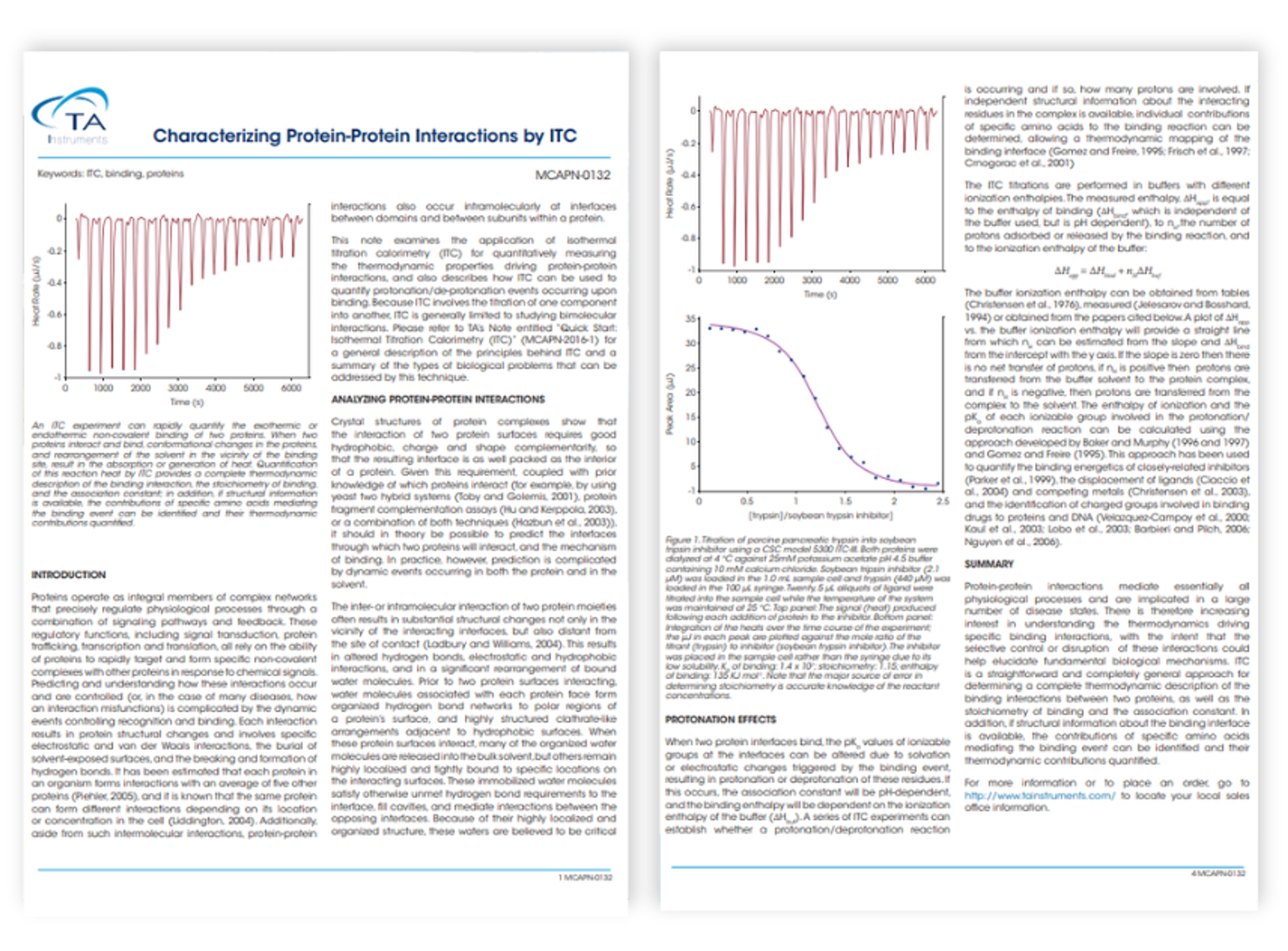
Application Note
Characterizing protein-protein interactions by ITC
White Papers
Agricultural products and crops testing services
White Papers
How to biotinylate with reproducible results
Application Note
Methods for 16S rRNA sequencing
Product Brochures
Galileo Series 2 Microtomes
White Papers
Food testing services
Application Note
Power up your assay development: DOE made easy on icon96™
Application Note