VENTANA MMR RxDx Panel
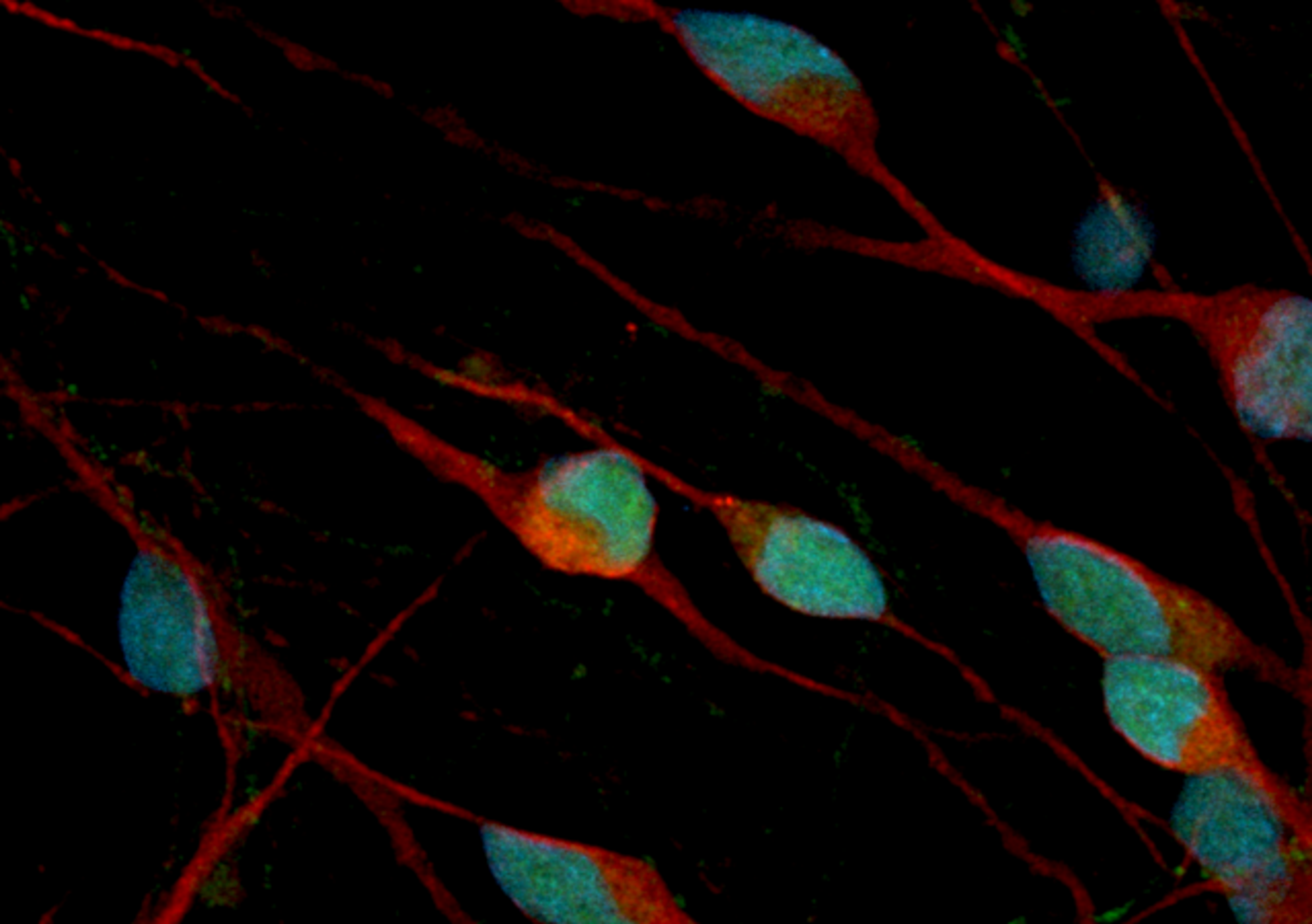
The VENTANA MMR RxDx Panel is the first and only immunohistochemistry-based assay approved to identify endometrial carcinoma patients eligible for treatment with JEMPERLI (dostarlimab).

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Intended use
VENTANA MMR RxDx Panel is a qualitative immunohistochemistry test intended for use in the assessment of mismatch repair (MMR) proteins (MLH1, PMS2, MSH2 and MSH6) in formalin-fixed, paraffin-embedded (FFPE) endometrial carcinoma tissue by light microscopy. The OptiView DAB IHC Detection Kit is used for MSH6, MSH2 and MLH1, and the OptiView DAB IHC Detection Kit with the OptiView Amplification Kit is used for PMS2 on a VENTANA BenchMark ULTRA instrument.
Novel diagnostic assay for gastric and gastroesophageal junction cancer receives CE Mark approval
Roche obtains CE certification for VENTANA CLDN18 (43-14A) RxDx Assay to identify patients with gastric and gastroesophageal junction cancer eligible for targeted treatment with VYLOY