GLP systems Track
AUTOMATION REINVENTED, FLEXIBILITY REDEFINED. In the United States, the GLP systems Track is FDA cleared and approved. Orderability is pending for certain modules and features.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
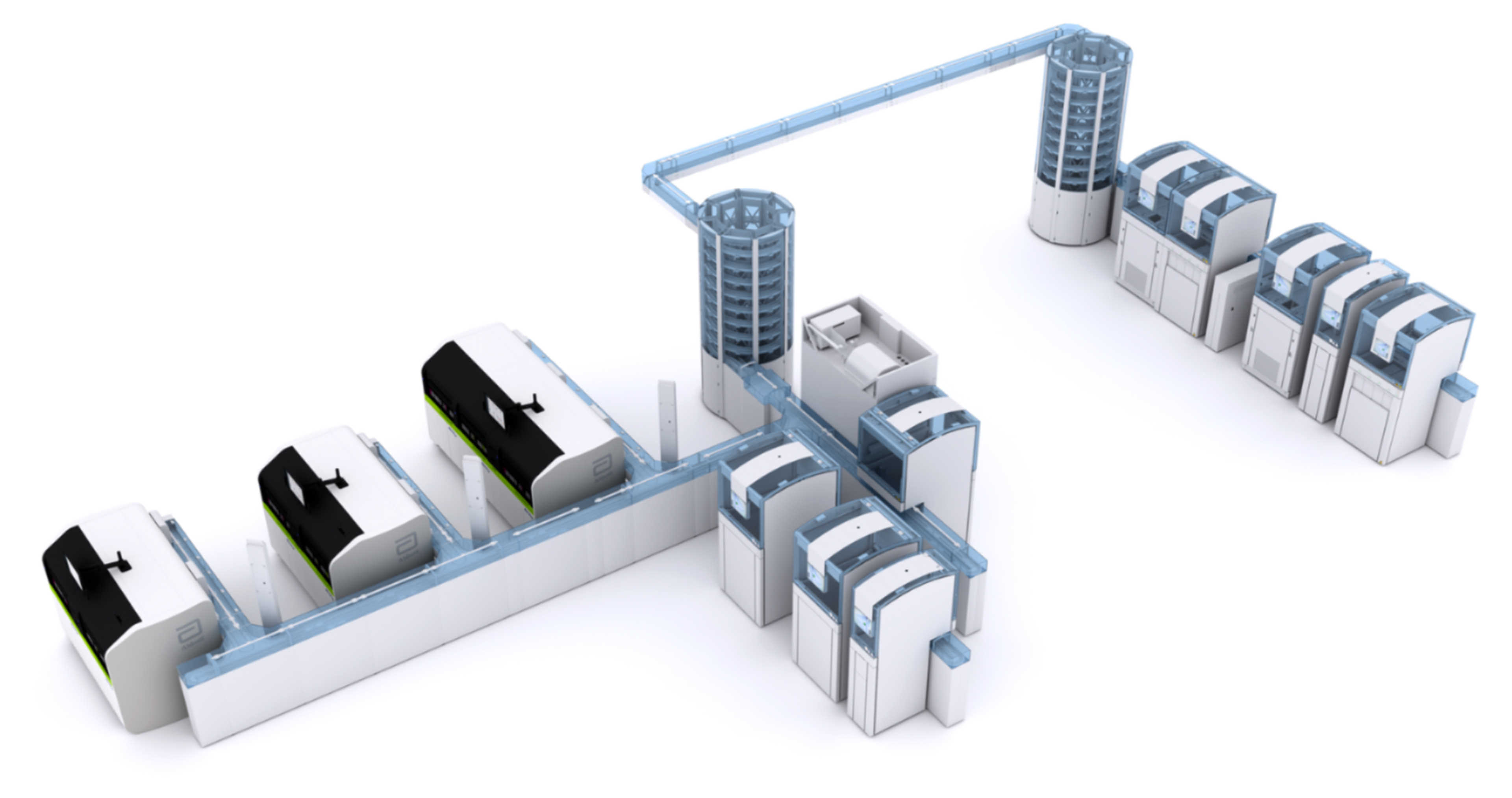
To help your laboratory thrive today and, in the future, Abbott is pleased to introduce GLP systems Track—an innovative total laboratory automation solution, offering you proven technology with greater flexibility and options to meet your high-volume needs.
**In the United States, the GLP systems Track is FDA cleared and approved. Orderability is pending for certain modules and features.
A case study on future proofing the lab with next-generation automation
Lauren Thomas, Executive of Laboratory and Outreach Lab Service, Mercy Southeast describes the installation of the very first Abbott GLP systems Track in the US. The lab’s rapid growth, driven by its outreach program, required technology that could evolve alongside it. Abbott’s next-generation automation systems, particularly the flexible GLP systems Track, provided the adaptability needed to meet changing test and volume demands over time, making them an ideal partner for long-term success.
This video is part of the global #CLINICAL24 2024 conversation.
Clinical Diagnostics Scientists’ Choice Awards winners announced at ADLM 2024
Thermo Fisher Scientific, Beckman Coulter Clinical Diagnostics, and LGC Clinical Diagnostics among those recognized