BioPhase 8800 system
Purpose-built to target quality and efficiency, the BioPhase 8800 system enables parallel sample throughput and analysis for capillary electrophoresis characterization.
Superb product. Allows for faster processing of samples.
Run multiple samples
The SCIEX BioPhase 8800 system offers a solution to the limited throughput that current single capillary CIEF systems provide. The new multi-capillary system enables parallel isoelectric focusing analysis for up to 8 samples at a time, allowing for rapid charge heterogeneity analysis of protein therapeutics.
Review Date: 28 Sept 2021 | SCIEX
The BioPhase 8800 system from SCIEX helps researchers take charge of obtaining relevant information on their molecules with increased separation throughput. From analytical development to quality control (QC), the BioPhase 8800 system allows for the separation of 8 samples in parallel. Achieve high-quality characterization of molecules like RNA-based drugs, next-generation therapeutics and monoclonal antibodies (mAbs) with this solution.
With 8 pre-built capillaries, temperature-controlled cartridges and compatibility with automation systems for 96 well plates, the BioPhase 8800 system enables a smooth set-up and experiment execution with maximum data reproducibility. The ability to easily and quickly switch between UV and laser-induced fluorescence (LIF) detection allows for additional flexibility for the molecules of today and future.
A series of validated kits and application workflows sets new frontiers for simplified usage and minimizing errors in integral product analysis including mRNA purity and integrity, protein size and purity quantitation, protein charge variant analysis and glycan identification.
Key features:
- Robustness: Ensure repeatability and reliability with the latest capillary electrophoresis hardware and software advances.
- Software: Simplify the path from samples to results with an intuitive touchscreen-based software.
- Flexibility: Experience seamless switching between UV and LIF detection for ultimate detector flexibility.
- Compatibility: Take control of compliance needs with the BioPhase 8800 driver for Empower software and the Empower Chromatography Data System (CDS) to control instruments and analyze data.
Applications:
- mRNA and genome integrity and purity analysis with the RNA 9000 Purity & Integrity kit
- Protein size and purity analysis with the BioPhase CE-SDS Protein Analysis kit
- Charge variant assessment with the BioPhase Capillary Isoelectric Focusing (cIEF) kit
- Glycan identification using the BioPhase Fast Glycan Labeling and Analysis kit
Brochures
BioPhase 8800 system 21 CFR Part 11 compliance features
In this product brochure, the data collection system, software, and controls that should be considered to achieve compliance with 21 CFR Part 11 are outlined. The product brochure highlights how the design and features of the BioPhase 8800 system facilitate compliance with 21 CFR Part 11.
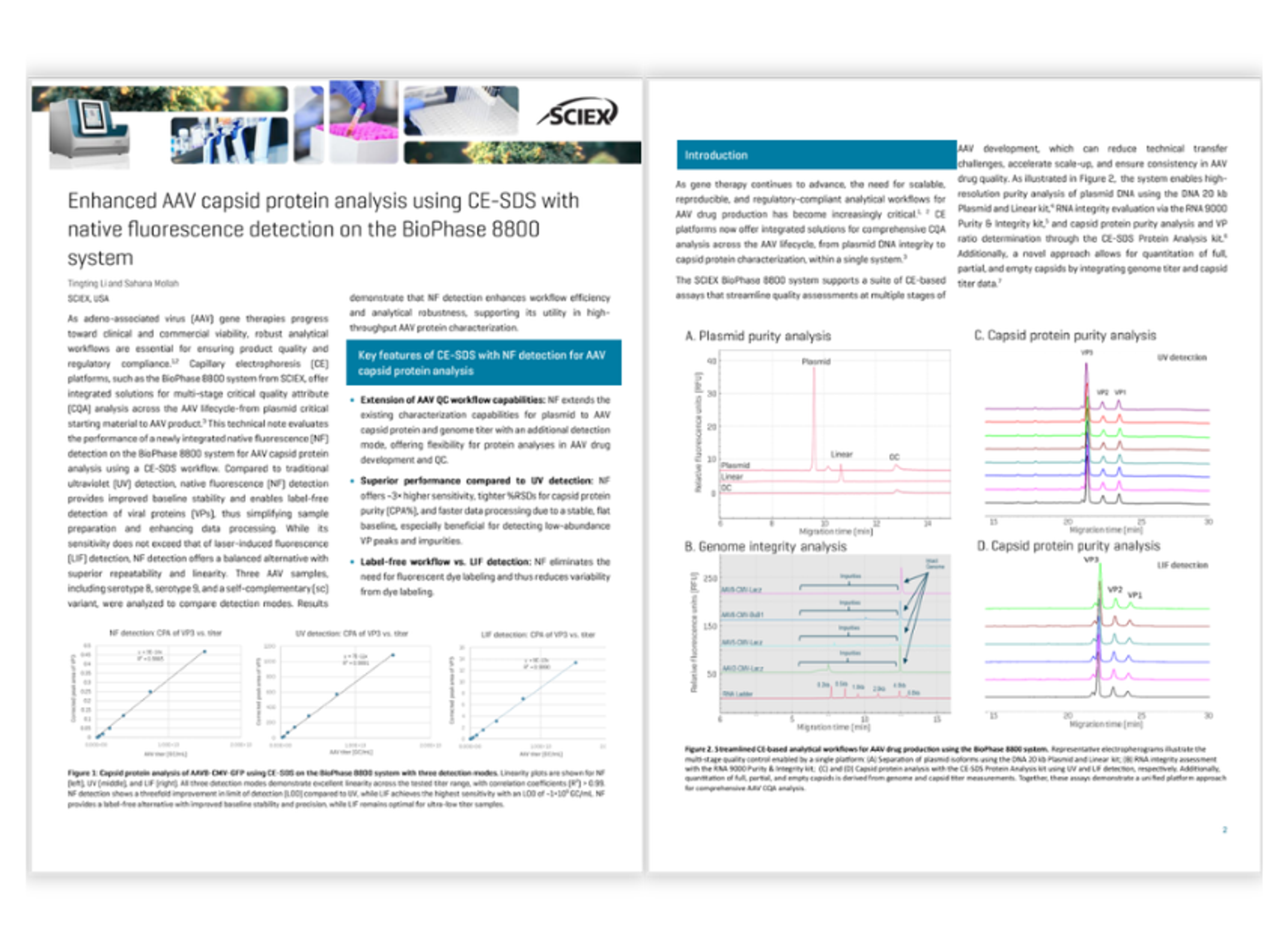
Enhanced AAV capsid protein analysis using CE-SDS with native fluorescence detection on the BioPhase 8800 system
This resource highlights AAV capsid protein analysis using CE‑SDS on the BioPhase 8800 system, enabling high‑resolution separation and quantitation of VP1, VP2, VP3, and VP3 capsid proteins. Comparative results using UV, laser‑induced fluorescence (LIF), and native fluorescence (NF) detection show that NF and LIF provide improved sensitivity, flatter baselines, and more reliable peak integration than UV detection.
The workflow also demonstrates how CE‑based assays for plasmid, RNA, and capsid analysis can be combined on a single platform to support comprehensive AAV critical quality attribute (CQA) assessment throughout gene therapy development and production.
BioPhase 8800 system with native fluorescence – Same foundation, more muscle
Capillary electrophoresis (CE) has long supported biopharma innovation, but evolving therapeutic demands require greater sensitivity and speed. Discover how native fluorescence and streamlined CE workflows enable faster, more confident impurity analysis. These approaches help to reduce noise, simplify preparation, and accelerate charge heterogeneity profiling for advanced biologics, mRNA drugs, and protein therapeutics.
Gaining high throughput without compromising data quality
Teva Pharmaceuticals’ Analytical Development lab needed a higher-throughput capillary electrophoresis (CE) solution to support stability testing, process development and formulation for quality control (QC).
This case study discusses the considerations to make when moving from the SCIEX PA 800 Plus system to the SCIEX BioPhase 8800 system in development labs while ensuring method transfer to QC labs. Plus, learn of Teva's plans to implement the BioPhase 8800 system for its clinical phase programs.
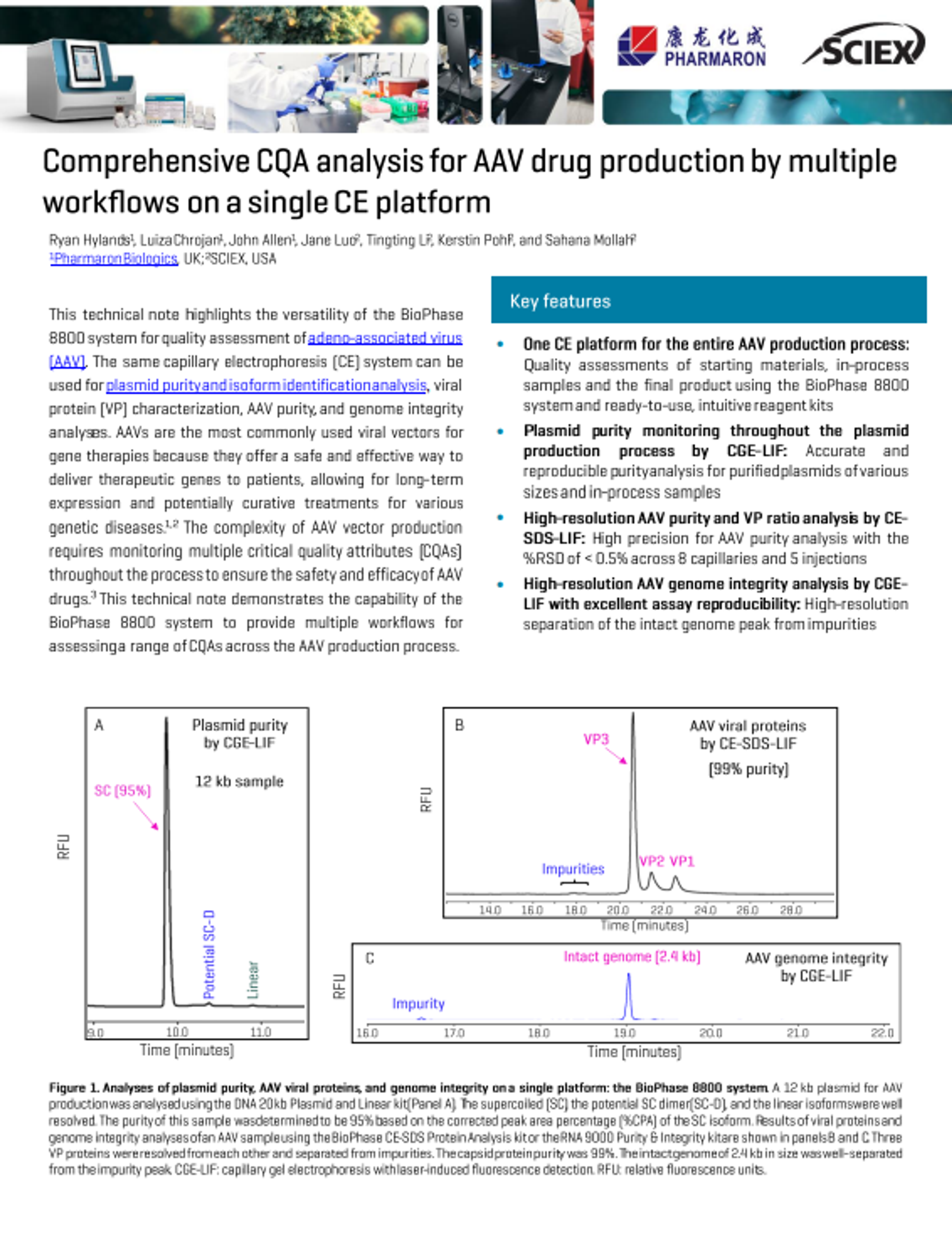
CQA analysis for AAV drug production by multiple workflows on a single CE platform
Adeno associated viruses (AAVs) are used widely in the development of gene therapies for viral vectors due to their safety and effective in delivery of therapeutic genes to patients. Monitoring the critical quality attributes (CQAs) of your AAV therapeutic is therefore critical to meet safety and efficacy and can be performed using capillary electrophoresis.
Discover the BioPhase 8800 system from SCIEX that offers multiple workflows for assessing different CQAs across the AAV production process. Explore its key features and how it can provide reproducible and high-precision results.
Assessment of integrity and purity of mRNAs from lipid nanoparticles
To ensure the quality of LNP-based drugs, comprehensive characterization of these sophisticated molecules is paramount. Explore how to achieve high-quality, robust assessment of the integrity and size of mRNA molecules encapsulated in LNPs.
High-throughput multi-capillary SDS gel electrophoresis of proteins
In this application note, the BioPhase 8800 system is used in the analysis of multiple samples simultaneously by sodium dodecyl sulfate gel electrophoresis (SDS-CGE). The application note also explores how the BioPhase 8800 system allows for analysis using UV and fluorescent detection in the same sequence.
Acceleration of method optimization for AAV capsid purity analysis using a multi-capillary electrophoresis platform
This application note highlights the use of the BioPhase 8800 system for accelerating method development for adeno-associated viruses (AAV). The application note demonstrates the system’s ability to perform 8 separations in parallel allowing for faster processing of samples and overall increased throughput.
Genome integrity analysis of adeno-associated viruses (AAV) using multi-capillary gel electrophoresis
In this application note, it is demonstrated that multiple AAV samples with different serotypes or different genome sizes can be run simultaneously on the multi-capillary BioPhase 8800 system to accelerate the execution of AAV genome integrity analysis.
Rapid and reproducible parallel processing of charge heterogeneity analysis of protein therapeutics by multi-capillary isoelectric focusing
In this application note, a parallel capillary isoelectric focusing analysis using the BioPhase 8800 system and a multi-capillary separation approach is described. Protein therapeutic charge variant assessment is essential at different manufacturing stages as they are subjected to instability, causing alterations in their primary amino acid sequence.
Addressing cell line development barriers with advanced analytics
In this SelectScience interview, explore key challenges, analytical solutions and future directions in cell line development
Optimize monoclonal antibody development with icIEF-UV/MS
Scientists at Johnson & Johnson Innovative Medicine are using the Intabio ZT system for the confident characterization of monoclonal antibodies