BioPharma Solution
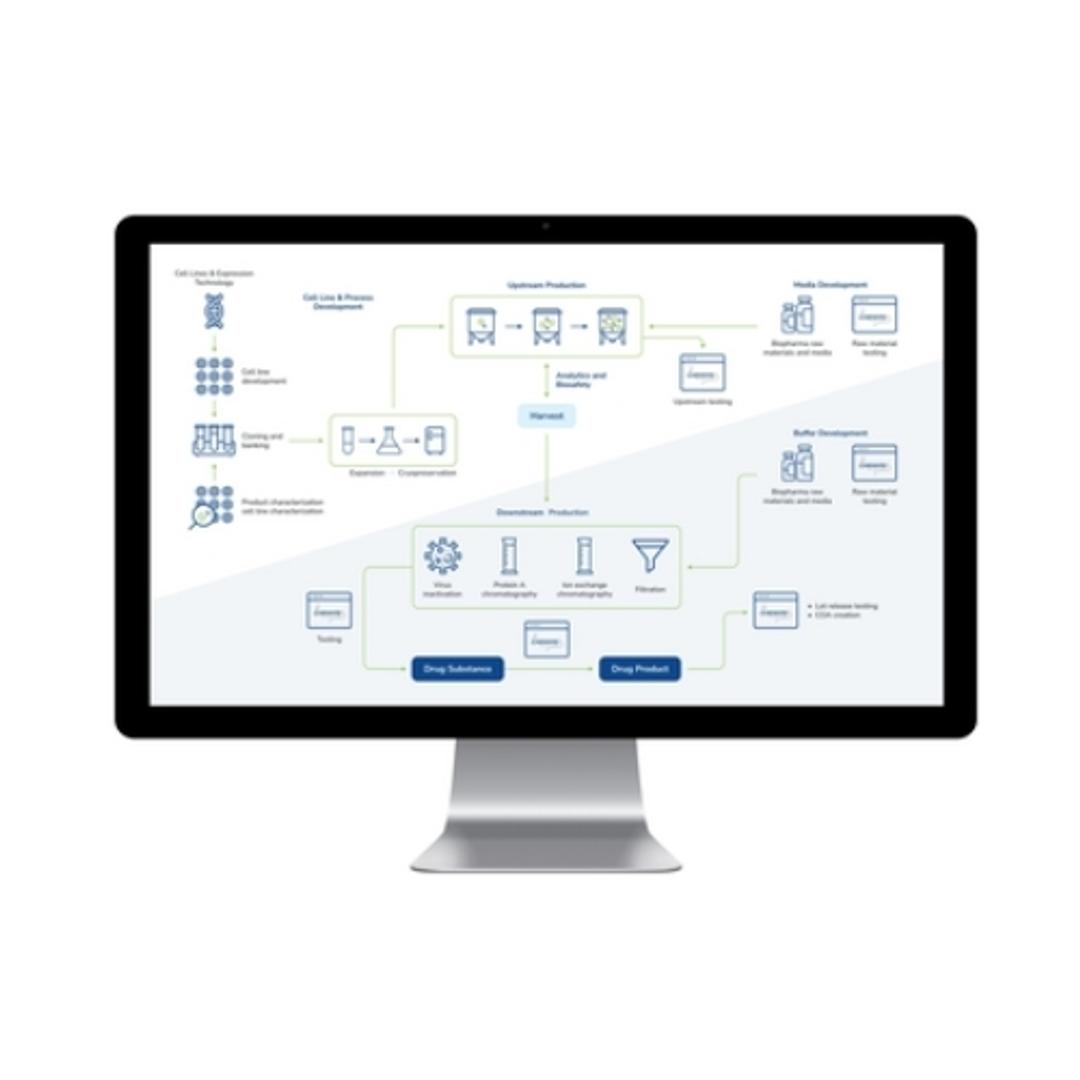
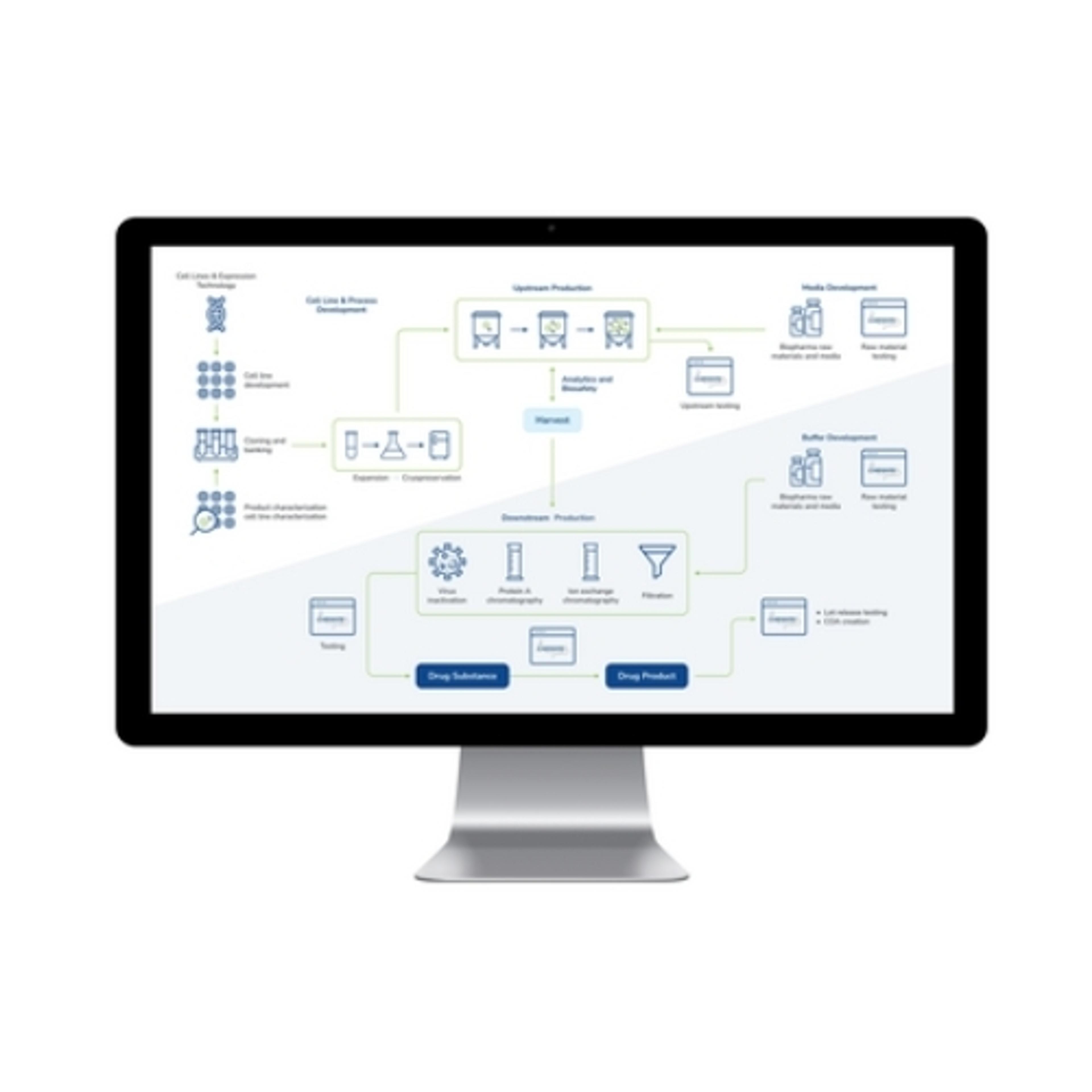
LabWare’s BioPharma Solution is a leading laboratory software platform designed for biopharmaceutical and biotechnology industries, promoting quality throughout the development and production lifecycle. The LabWare Laboratory Information Management System (LIMS) platform streamlines complex processes, from cell line development and banking to the intricate steps of biopharmaceutical manufacturing, including upstream and downst…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
LabWare’s BioPharma Solution is a leading laboratory software platform designed for biopharmaceutical and biotechnology industries, promoting quality throughout the development and production lifecycle. The LabWare Laboratory Information Management System (LIMS) platform streamlines complex processes, from cell line development and banking to the intricate steps of biopharmaceutical manufacturing, including upstream and downstream processing. With its emphasis on automation, real-time data tracking, and stringent compliance enforcement, the LabWare BioPharma Solution is a good choice for laboratories seeking efficiency, accuracy, and regulatory adherence in their operations.
LabWare BioPharma Solution key features include:
- Streamlined Biopharmaceutical Processes: Optimizes cell line development, upstream and downstream processing, and media & buffer formulation, promoting adherence to GMP for high-quality products.
- Advanced Instrumentation Support: Facilitates automation and paperless operations through seamless interfacing with laboratory analyzers and robotics, enhanced by a configurable Rules Engine for increased efficiency.
- Regulatory Compliance and Data Integrity: Through robust data management and audit trails, LabWare promotes comprehensive documentation, traceability, and compliance with critical regulations and industry standards, including 21 CFR Part 11 and ISO 17025.
- Operational Efficiency and Insight: Offers real-time tracking, built-in laboratory KPIs, and configurable dashboards for data review and process optimization, alongside comprehensive tools for stability study management and inventory control.
Data Agility in Biopharma - Strategies for Success
In the rapidly evolving landscape of biopharmaceutical product development, effective data strategies are essential for driving innovation, ensuring regulatory compliance, facilitating efficient decision-making, improving research outcomes, and accelerating development timelines.
In this webinar, Tom Brohan, BioPharma Leader at LabWare, will detail the common and distinctive challenges encountered in biopharmaceutical laboratories and will provide practical strategies for overcoming them. Topics he will cover include data integration, volume, and complexity; data quality and standardization; data security and compliance; and access and collaboration challenges. Furthermore, the webinar will highlight the specific data management challenges faced by R&D, bioanalysis, and QA/QC bioprocessing laboratories, offering solutions to address these intricacies.
Key learning objectives
- Explore the common data challenges scientists face in biopharmaceutical research and development
- Discover practical strategies on how to overcome specific data challenges
- Learn new ways to unlock and interpret information from different data sources
Who should attend?
- Quality managers in biopharma operations
- R&D scientists in biopharma
- Lab managers
- Bioprocess engineers
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.