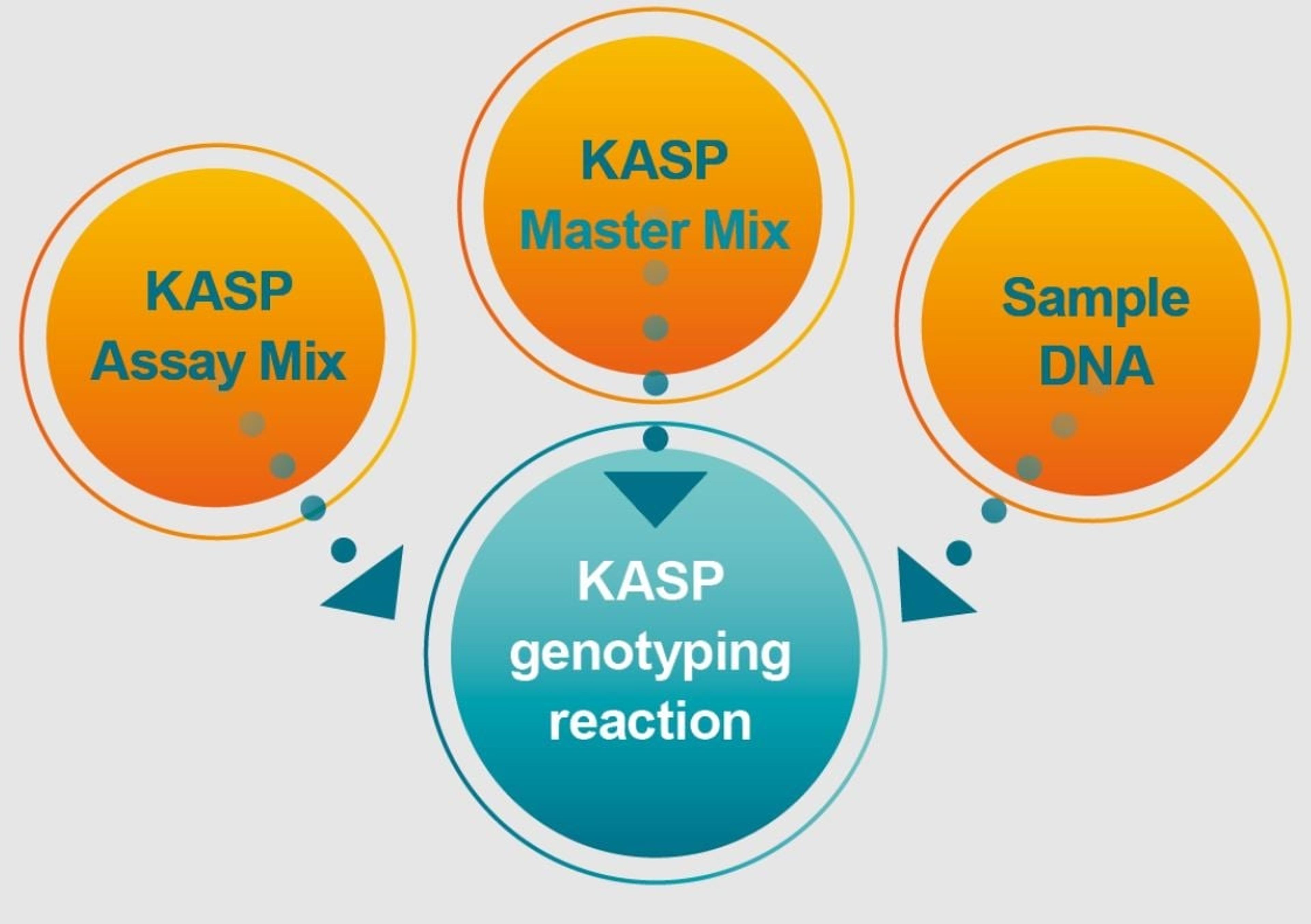
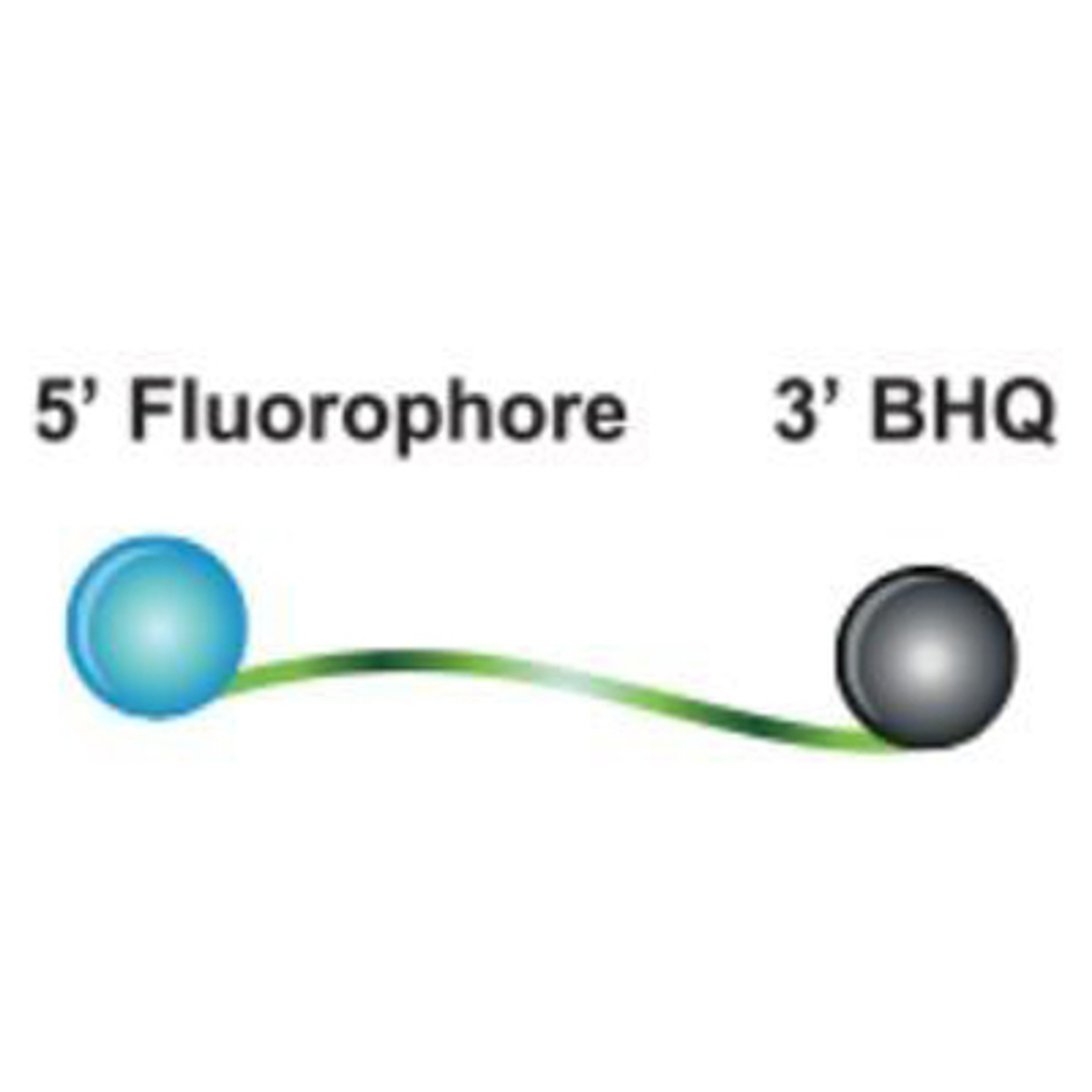
sbeadex™ Pathogen Nucleic Acid Purification Kit
High-throughput, high-quality DNA and RNA purification for detection of multiple pathogens.
Pathogen testing protocols (e.g. for COVID-19, influenza or easy-to-lyse bacterial pathogens) require fast, reliable, high-yield, high-quality and easily automatable sample preparation methods.
Our sbeadex™ Pathogen Nucleic Acid Purification Kits with their optimised chemistry meet these requirements and deliver excellent yields of highly purified nucleic acids from various sample material that can be used in many downstream applications such as PCR/qPCR, RT-PCR/qRT-PCR, and NGS. The purification procedure can be fully automated and is compatible with many robotic platforms.
The sbeadex Pathogen Nucleic Acid Purification Kit is a simple renaming of our proven sbeadex Viral RNA Purification Kit to reflect its continuously expanding usability to purify good yields of high-quality DNA and RNA from a variety of viral and bacterial pathogens.
Key benefits
- Qualified protocol for viral and bacterial nucleic acid purification
- Compatible with most popular robotic platforms, including the Kingfisher™ and our high-throughput oKtopure™ instrument
- Fast – both short (25 min) and standard (45 min) protocols available
- High-throughput batch sizes (up to 10,000 purifications)
- No organic extraction, alcohol precipitation or centrifugation steps
- Water-based wash buffers reduce solvent carryover, resulting in absence of PCR inhibitors in final eluate
- High nucleic acid quality for all downstream applications including PCR/qPCR and NGS
- Ready-to-use kits with high stability (customised and bulk size kits are also available)
Nucleic acid extraction and purification for molecular diagnostics
Molecular testing protocols require reliable and high-quality isolation of genetic material from diverse sample types. In this infographic, LGC Biosearch Technologies explores how its portfolio of products and services can support your molecular testing workflows and shares how to select the best kits for your sample type and downstream requirements.
Automate RNA purification for high-throughput processing of 2019-nCoV samples
This application note discusses how sbeadex™ Pathogen Nucleic Acid Purification kit is used to purify RNA from 2019-nCoV buccal swab and sputum samples for downstream RT-qPCR applications.
Reliably purify nucleic acids from respiratory pathogens across a broad concentration range
This application note discusses how sbeadex™ Pathogen Nucleic Acid Purification Kit is used to purify DNA and RNA from a variety of respiratory disease samples for downstream qPCR, RT-qPCR, and next generation sequencing (NGS) applications.
Lessons on sample accessioning and processing at clinical labs during a pandemic
In this eBook, LGC Biosearch Technologies details the major challenges labs have faced during the COVID-19 pandemic and how they have adapted to handle the sustained demand for large-scale sample accessioning and testing. Nischay Mishra, PhD, an Assistant Professor of Epidemiology and infectious disease diagnostic developer at the Center for Infection and Immunity at the Columbia University Medical Center, provides insight and key takeaways on the next steps to improve testing workflows.
The future of diagnostics: How innovators in molecular techniques are shaping healthcare
From enzymatics to informatics, Hillary Sullivan, Vice President of R&D at LGC Biosearch Technologies, shares her thoughts on some of the latest advancements in molecular diagnostics. With insights from successful partnerships in healthcare, Sullivan provides her perspective on what the most promising areas of scientific advancements might hold for this rapidly developing field in the next 25 years.
Tackling HPV detection and the global cervical cancer prevalence problem
Explore the concerted effort healthcare professionals, pharma, and biotech are making to combat cervical cancer through the development and implementation of new HPV assays
LAMP-based diagnostics: Rapid and truly portable molecular testing at the point-of-need
Discover how a multi-disciplinary team located at Imperial College London created a novel portable diagnostic platform and delivered a rapid and accurate 5-in-1 respiratory pathogen detection test panel