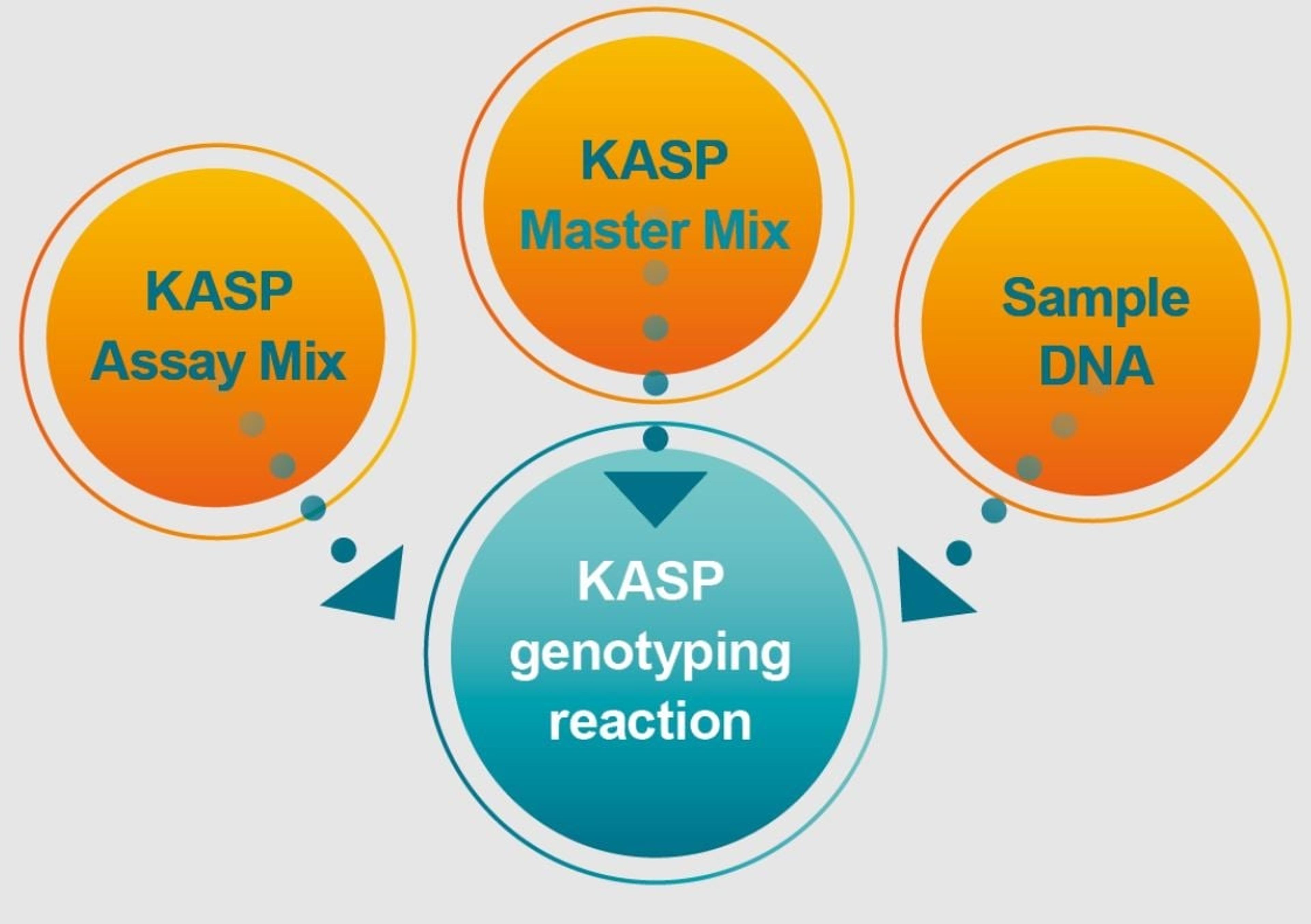
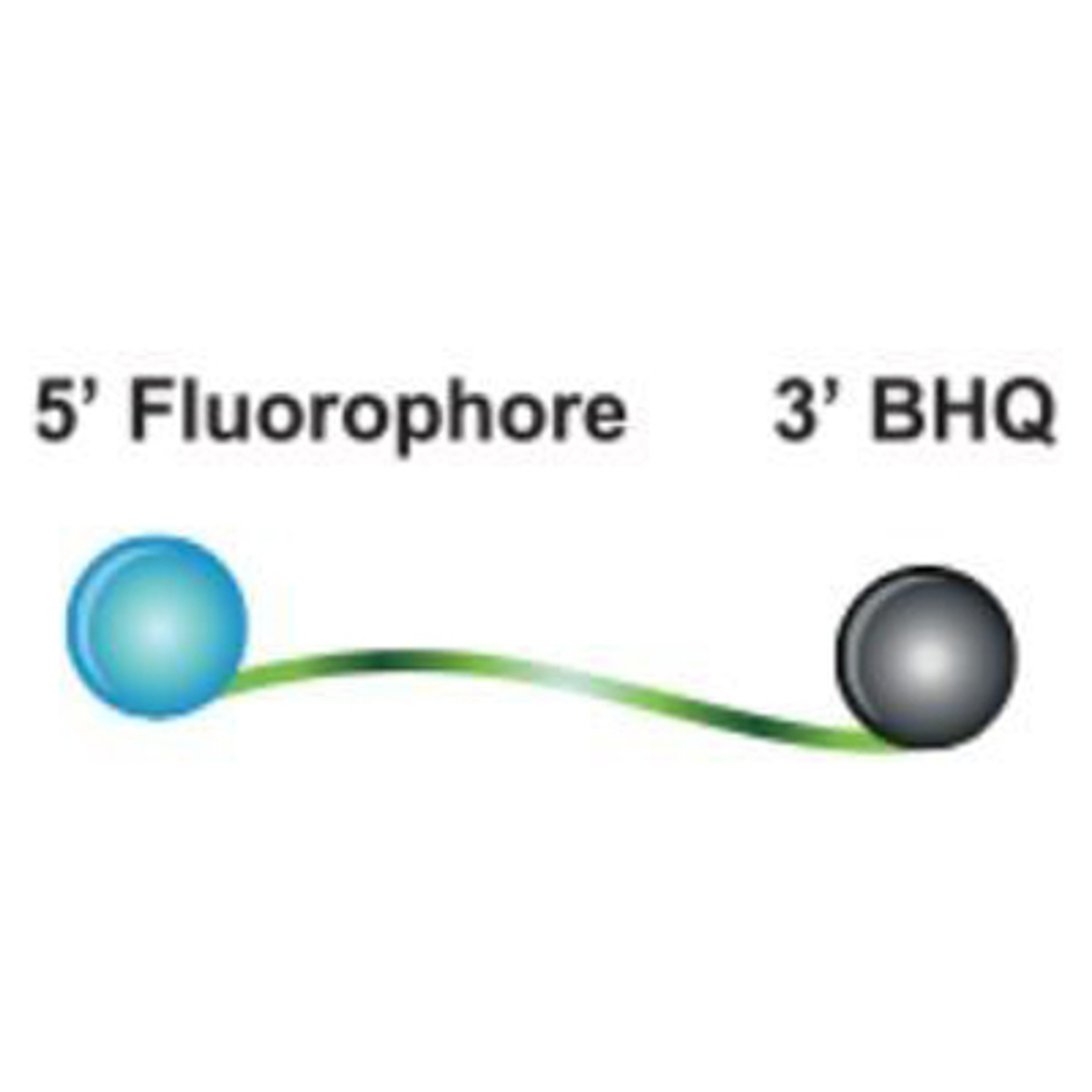
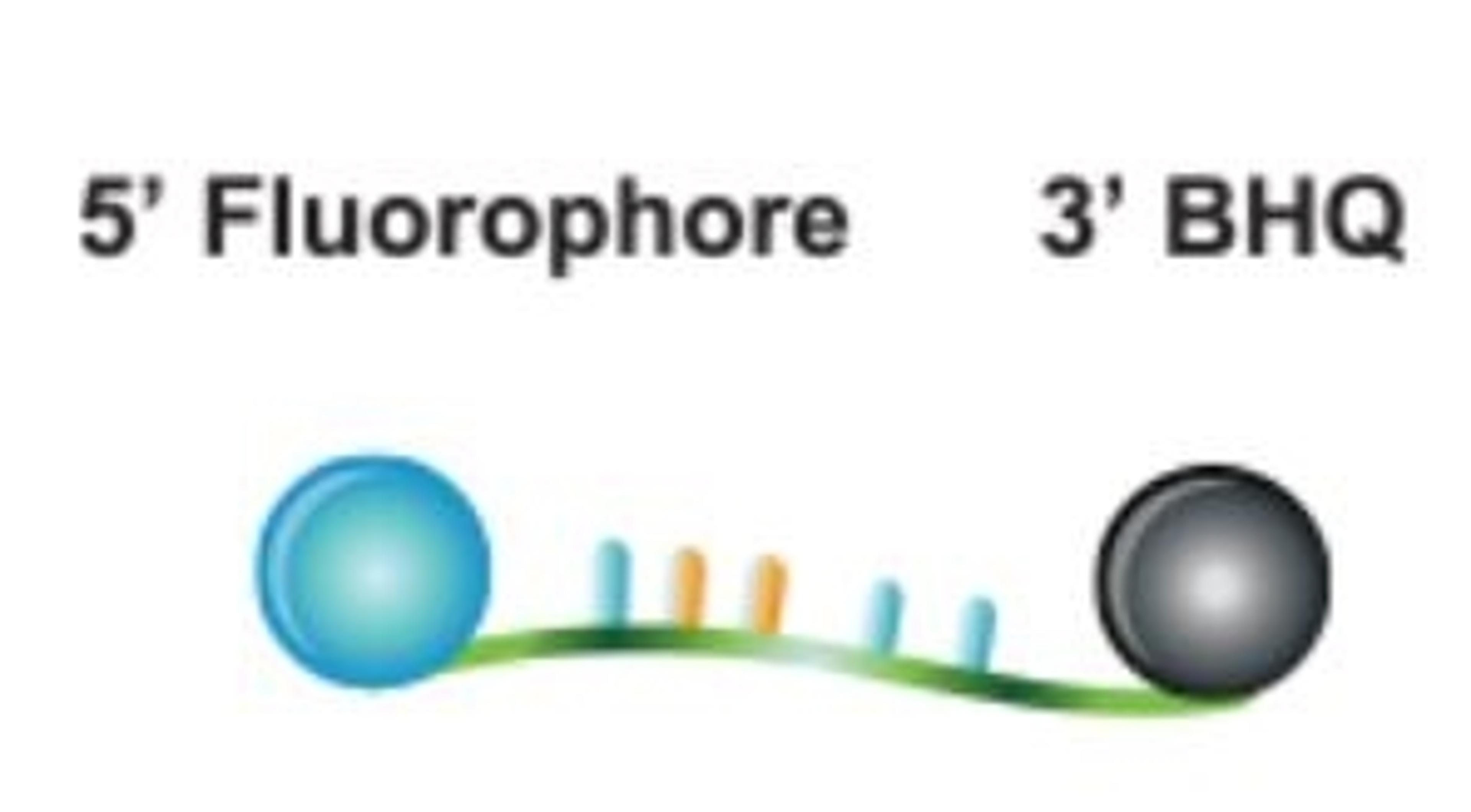
Custom oligos
Custom design your oligonucleotide according to your specifications. Our probes and primers are manufactured at unmatched quality under ISO 9001 and 13485 compliance. Whether you need RUO or GMP grade oligos, choose from a vast selection of modifications to customize your oligos for your application.
Great product
Analysis for DNA
Good product
Review Date: 13 Jan 2023 | LGC Biosearch Technologies
Overall, I am very satisfied with the product and excellent customer service!
Amplification of tissue culture DNA
Very comfortable to use and service and price is great!
Review Date: 1 Nov 2017 | LGC Biosearch Technologies
High quality - happy customer!
RT-PCR testing of GMOs in grain
High quality custom oligos. Easy, accurate & efficient to order with friendly customer service.
Review Date: 4 Aug 2017 | LGC Biosearch Technologies
Great products, but could use more versatility in concentration/volume.
Real-Time PCR
If a company is potentially ordering larger batches of custom oligos, it may be useful to offer the option of either creating more highly concentrated vials, or vials with larger volumes. At times, we create LARGE batches of in-house master mix, and it is valuable to us to not have to open numerous small vials of custom oligos.
Review Date: 1 Aug 2017 | LGC Biosearch Technologies
Oligos are easy to order and a high quality product.
PCR Testing
These products are easy to use and we've seen good results.
Review Date: 1 Aug 2017 | LGC Biosearch Technologies
great results
Polymerase Chain Reaction
The oligonucleotides come lyophilized with a certificate of analysis provided. This makes it much easier to verify the quality of the product and the ability to resuspend the oligonucleotides. The oligos are of high quality and have always given consistent results in PCR.
Review Date: 1 Aug 2017 | LGC Biosearch Technologies
Great results and good price!
smFISH probes
I have used biosearchtech for oligo plates multiple times. I like the product, and customer service.
Review Date: 1 Aug 2017 | LGC Biosearch Technologies
Satisfactory
biosensor for biomarker detection
The order was reviewed critically and delivered within my expected time. With the products, I can produce consistent results. The customer service is responsive and helpful as well.
Review Date: 1 Aug 2017 | LGC Biosearch Technologies
Great results
Biosensing
High quality oligos with different modifications. The costumer service is fantastic
Review Date: 1 Aug 2017 | LGC Biosearch Technologies
Custom oligos from this company are good for imaging
RNA FISH to Study RNA level and localisation
I received my custom oligos tagged with flourochrome in time. They provided detailed information about the product and how to suspend oligos. My RNA FISH images with custom oligos were quite good and reproducible.
Review Date: 31 Jul 2017 | LGC Biosearch Technologies
Custom design your oligonucleotide according to your specifications. Our probes and primers are manufactured at unmatched quality under ISO 9001 and 13485 compliance. Whether you need RUO or GMP grade oligos, choose from a vast selection of modifications to customize your oligos for your application.
Simplifying the journey from molecular diagnostic assay development to commercialization
Molecular diagnostics (MDx) is rapidly advancing, with a growing array of DNA- and RNA-based tests now available to physicians, pharmacists, geneticists, research scientists, and other healthcare professionals.
As labs work with human and microbial genes, test developers must comply with clinical guidelines and regulations. Both established companies and start-ups face challenges, including meeting regulatory standards, ensuring quality, and managing supply chain issues. Early planning and robust partnerships are key to overcoming these obstacles.
This essential guide explores these challenges through detailed case studies. It covers everything from early development and assay formulation, to supply chain security, offering expert insights on successfully bringing PCR, next-generation sequencing (NGS), and other nucleic acid-based diagnostic tests to market.
Developing the 5-minute point-of-care PCR test: A technology revolutionising molecular diagnostics
With the challenges faced at the peak of the COVID-19 pandemic still very fresh in the memory, delivering point-of-care testing with speed, convenience, and reliability remains a pressing need in primary care. A resurgence in other respiratory pathogens, including influenza A and B, has left no respite for urgent care centers, general practice, and pharmacies as their testing services remain under pressure. The development and implementation of a PCR platform that could produce results in just minutes rather than hours would therefore be transformative.
In this SelectScience® webinar, Andrew Anyakwo, Head of Reagent Development at LEX Diagnostics, will give a unique insight into how they have been able to achieve just that - developing a low-cost, point-of-care PCR test that aims to diagnose SARS-CoV-2 and Flu A/B in just 5 minutes with high sensitivity and specificity.
Key learning objectives
- Understand the challenges the rapid development of assays/devices presents, how they can be overcome, and what can be learned in the process
- Gain insight into how to deal with inhibitors
- Explore the benefits of partnering with a manufacturer to develop a solution with GMP at scale
- Discover how PCR can be improved upon to revolutionise point-of-care molecular diagnostics
Who should attend?
Assay developers and scientists working within the molecular diagnostics field.
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
The future of diagnostics: How innovators in molecular techniques are shaping healthcare
From enzymatics to informatics, Hillary Sullivan, Vice President of R&D at LGC Biosearch Technologies, shares her thoughts on some of the latest advancements in molecular diagnostics. With insights from successful partnerships in healthcare, Sullivan provides her perspective on what the most promising areas of scientific advancements might hold for this rapidly developing field in the next 25 years.
Decentralizing healthcare through point-of-care diagnostics
Portable testing platforms are set to drive precision medicine and community health into 2024, so when will they see a more widespread adoption amongst healthcare providers and individuals alike?
Globalization presents challenges and opportunities in infectious disease outbreak preparedness
How is globalization impacting the transmission of infectious diseases and what response is required from global health networks for greater security?
Transforming molecular diagnostics: Unique primer technology dramatically increases the accuracy and specificity of PCR tests
More affordable diagnostic tests for multiple diseases could soon be available thanks to innovative Co-Primers technology
LAMP-based diagnostics: Rapid and truly portable molecular testing at the point-of-need
Discover how a multi-disciplinary team located at Imperial College London created a novel portable diagnostic platform and delivered a rapid and accurate 5-in-1 respiratory pathogen detection test panel
A revolution in diagnostics – The race for five-minute point-of-care PCR testing
Learn how UK-based diagnostics company, LEX Diagnostics, is working with LGC Biosearch Technologies to precipitate a step-change in PCR usability and speed
Saliva-based open-source tests for COVID-19: How saliva sampling improves compliance, saves costs, and relieves the strained supply chain
A microbiologist at Yale University decided to test saliva samples for COVID-19 detection, and it resulted in a widely used, cost-effective, and accessible test for the public
Combating antibiotic-resistant sexually transmitted infections with molecular diagnostics
Explore how a partnership between a lead researcher in sexual health and a molecular diagnostics innovator helped to overcome the therapeutic hurdles that come with antibiotic resistance