SARS-CoV-2 Variant ValuPanel™ assays
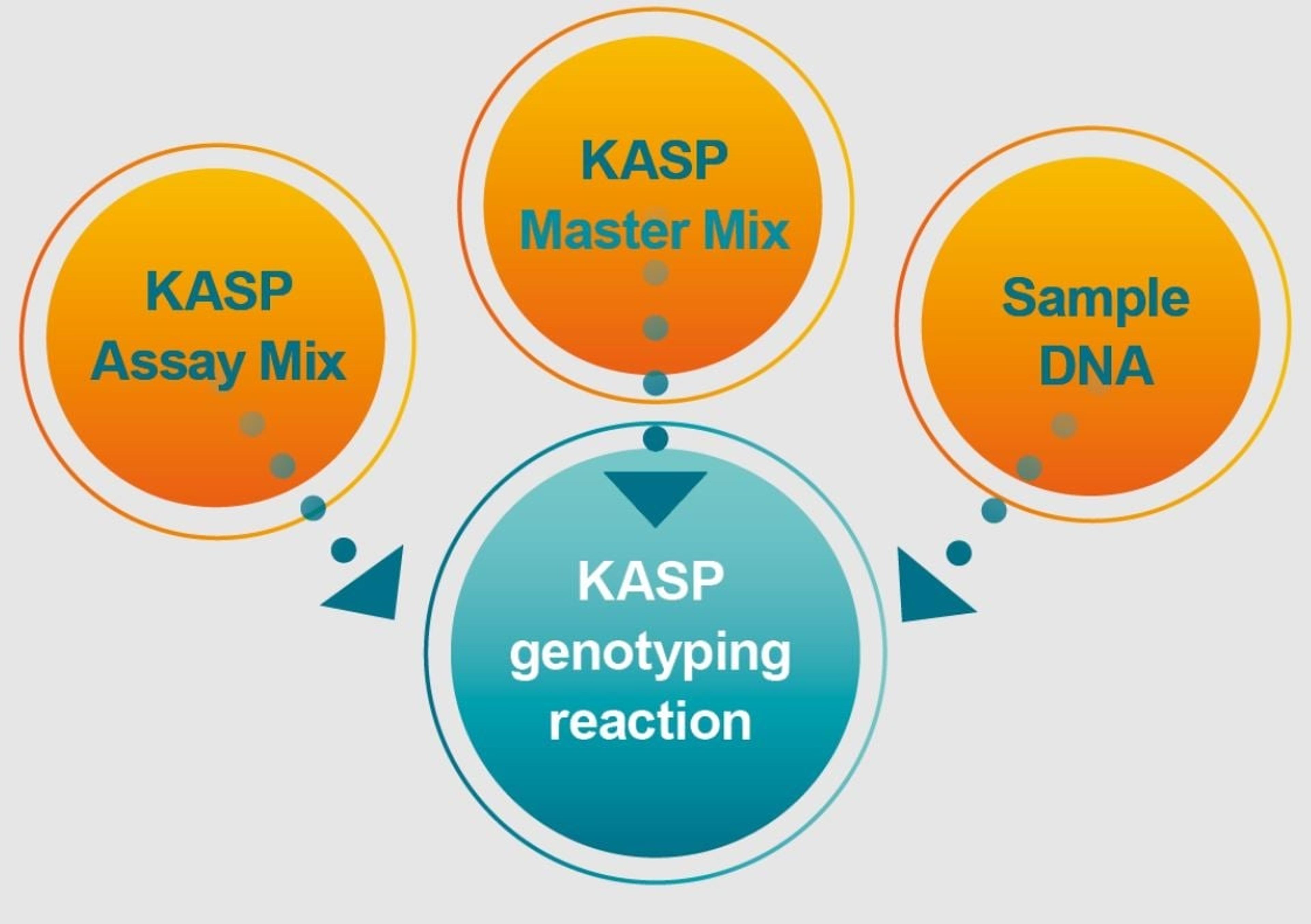
SARS-CoV-2 Variant ValuPanel™ assays consist of separately delivered probes and primers that are designed for qualitative detection of specific SARS-CoV-2 mutations by genotyping using reverse transcription-polymerase chain reaction (RT-PCR).
Great product with high quality results
Used for qualitative detection of specific SARS-CoV-2 mutations
Easy to understand the instructions. SARS-CoV-2 Variant ValuPanel™ assays gives highly specific and quality results. Highly recommended
Review Date: 11 Sept 2021 | LGC Biosearch Technologies
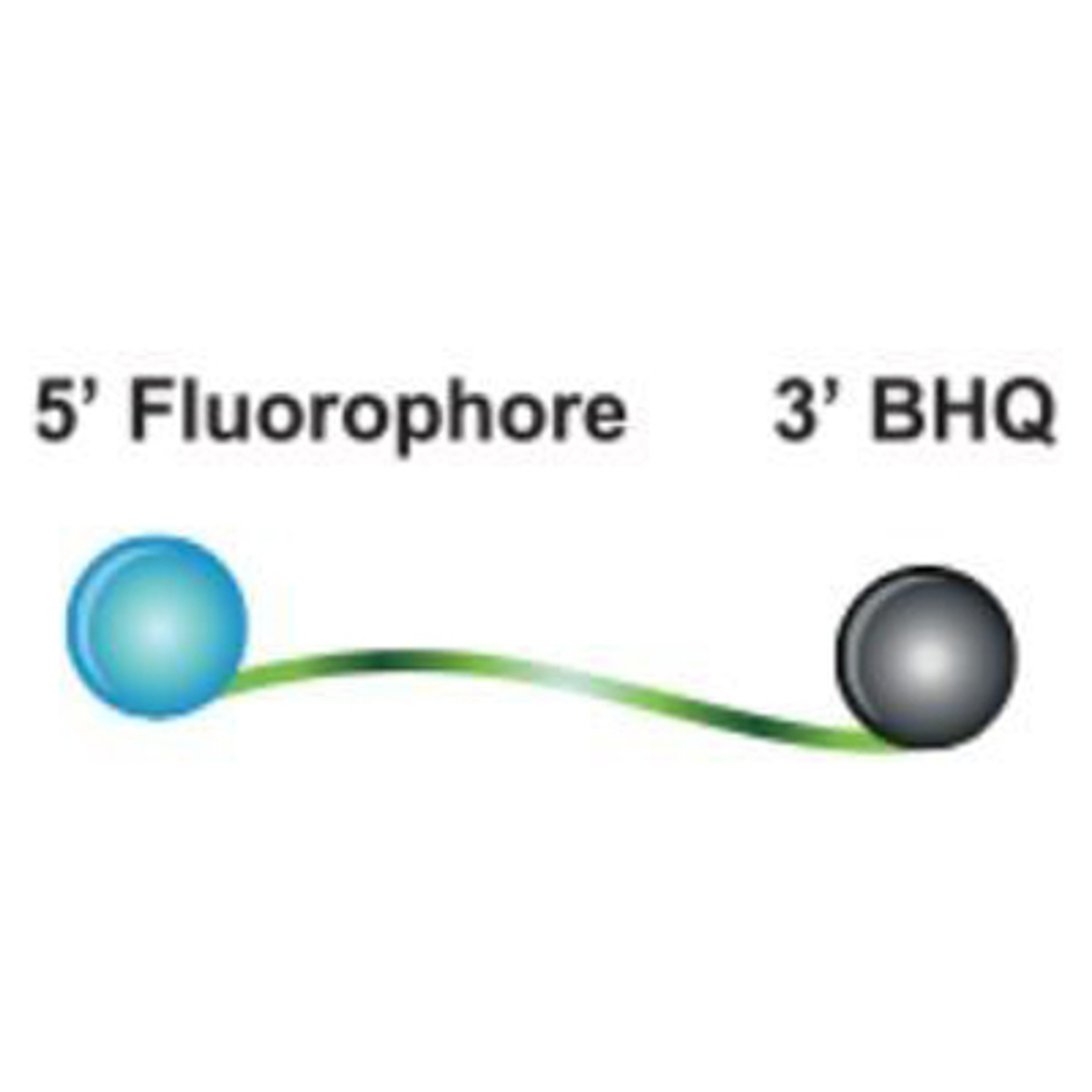
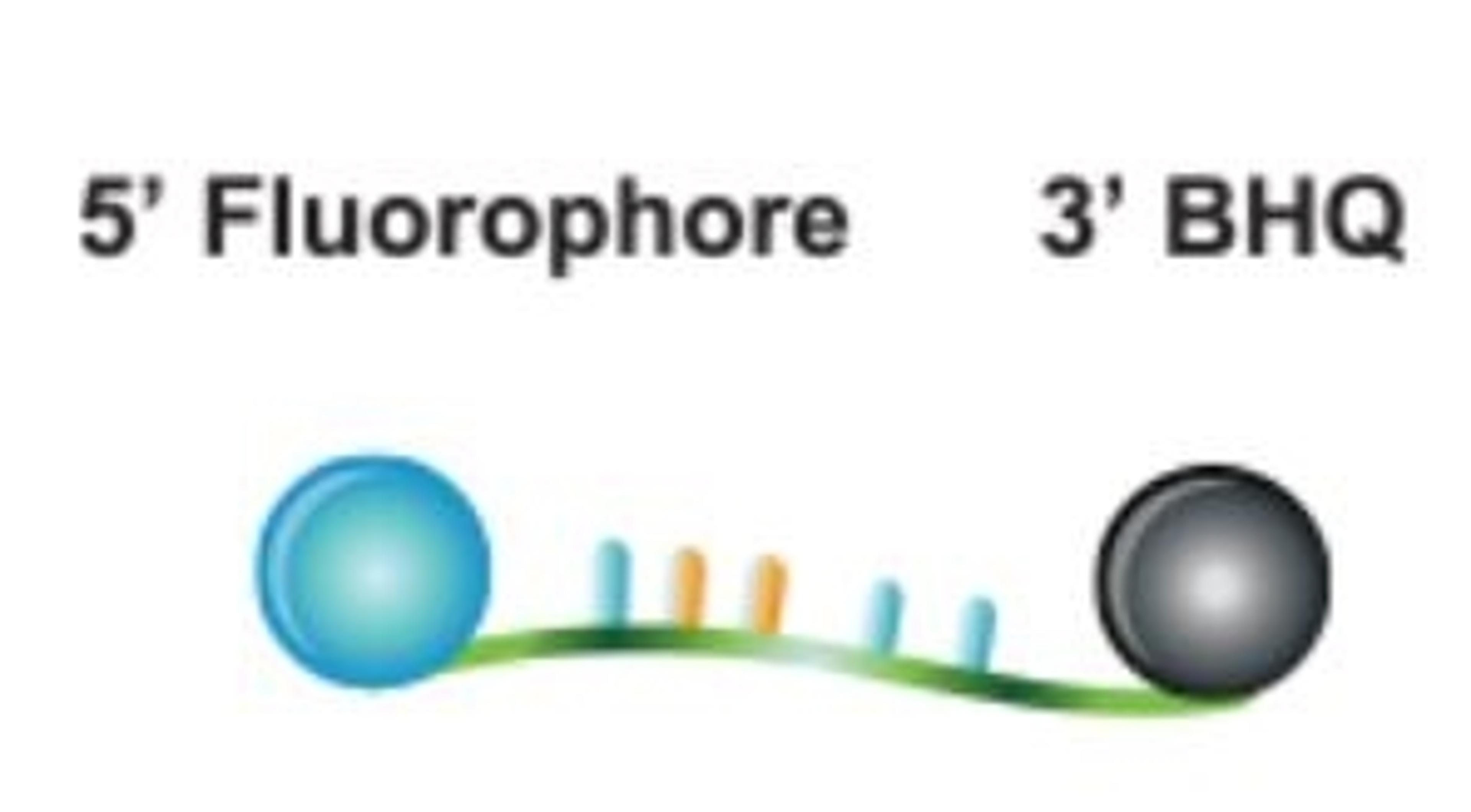
SARS-CoV-2 Variant ValuPanel™ assays consist of separately delivered probes and primers that are designed for qualitative detection of specific SARS-CoV-2 mutations by genotyping using reverse transcription-polymerase chain reaction (RT-PCR). Each SARS-CoV-2 Variant ValuPanel consists of 2 BHQplus™ Probes and 2 primers for amplification and simultaneous discrimination between a specific mutation and the wild-type SARS-CoV-2 sequence.
For Research Use Only. Not for use in diagnostic procedures.
Several variants of SARS-CoV-2 have emerged bringing challenges to diagnostic tests and eradication efforts. Of particular significance are variants B.1.1.7 (first detected in the United Kingdom), B.1.351 (first detected in South Africa), and P.1 or B.1.1.28 (first identified in travelers from Brazil who arrived in Japan). The ability to quickly and reliably identify SARS-CoV-2 mutations will enhance the ability to gather crucial public health information regarding transmissibility kinetics of new variants, and the efficacy of vaccines and therapeutics.
SARS-CoV-2 Variant ValuPanel assays are for research use only, they are not intended for SARS-CoV-2 diagnosis. The SARS-CoV-2 Variant ValuPanel assays are intended for reflex tests only, designed for variant screening in respiratory tract samples that have previously tested positive for SARS-CoV-2 by diagnostic RT-PCR.
Key benefits
- Reliable results: From the trusted inventor of Black Hole Quencher™ (BHQ™) technology referenced in 7 of 8 WHO COVID-19 protocols
- High specificity: Enhanced affinity of the BHQplus Probe technology delivers high-fidelity genotyping results
- Flexible assay design: Increased assay design options with separately delivered probes and primers
- Consistent quality: All probes and primers are HPLC purified and are manufactured and shipped from a facility entirely separate from positive control production
Keeping up with COVID-19: Designing novel monitoring assays to tackle the rise of variants
Dr. Tania Nolan outlines how her team is designing assays to identify new variants and why these are critical to managing the global response