Reconstructed Bone (r-Bone™) 3D Cell Culture Service, Mulitiple Myeloma
Human, bone marrow 3D culture model enabling accurate testing of therapeutic agents against multiple myeloma.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Reconstructed Bone (r-Bone) platform offers 1-to-1 reconstruction of cellular and extracellular compartments of the human bone marrow microenvironment. r-Bone preserves the architecture of human bone marrow and enables accurate evaluation of response to treatment.
Unique Advantages:
- Clinically relevant system demonstrating high correlation with clinical outcomes (in a clinical study, 19 out 21 cases of response vs. resistance to treatment were predicted correctly).
- Supports evaluation of efficacy and toxicity in a single assay.
- Suitable for testing of immuno-oncology agents.
- Comprehensive system to ascertain mechanisms of drug resistance.
Features:
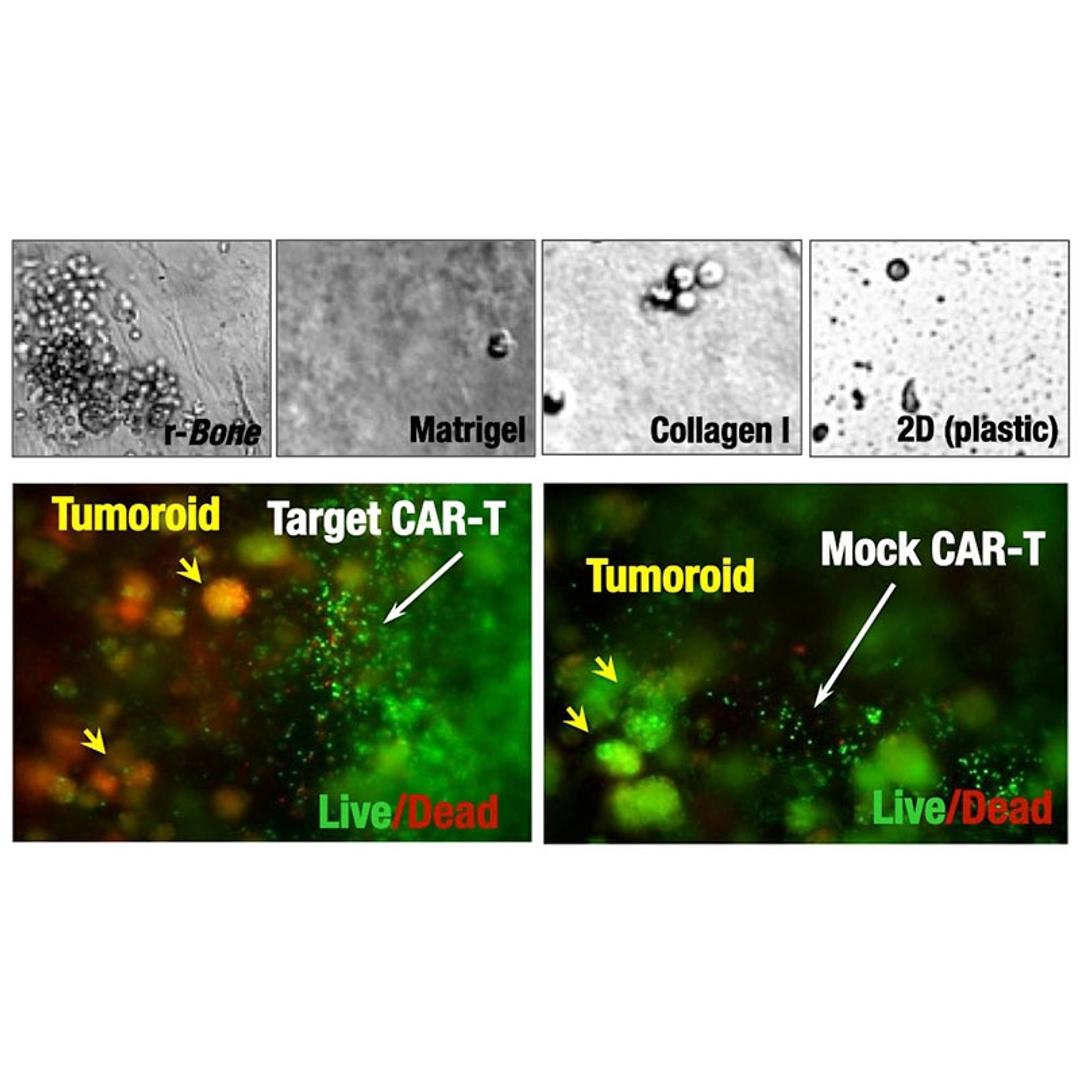
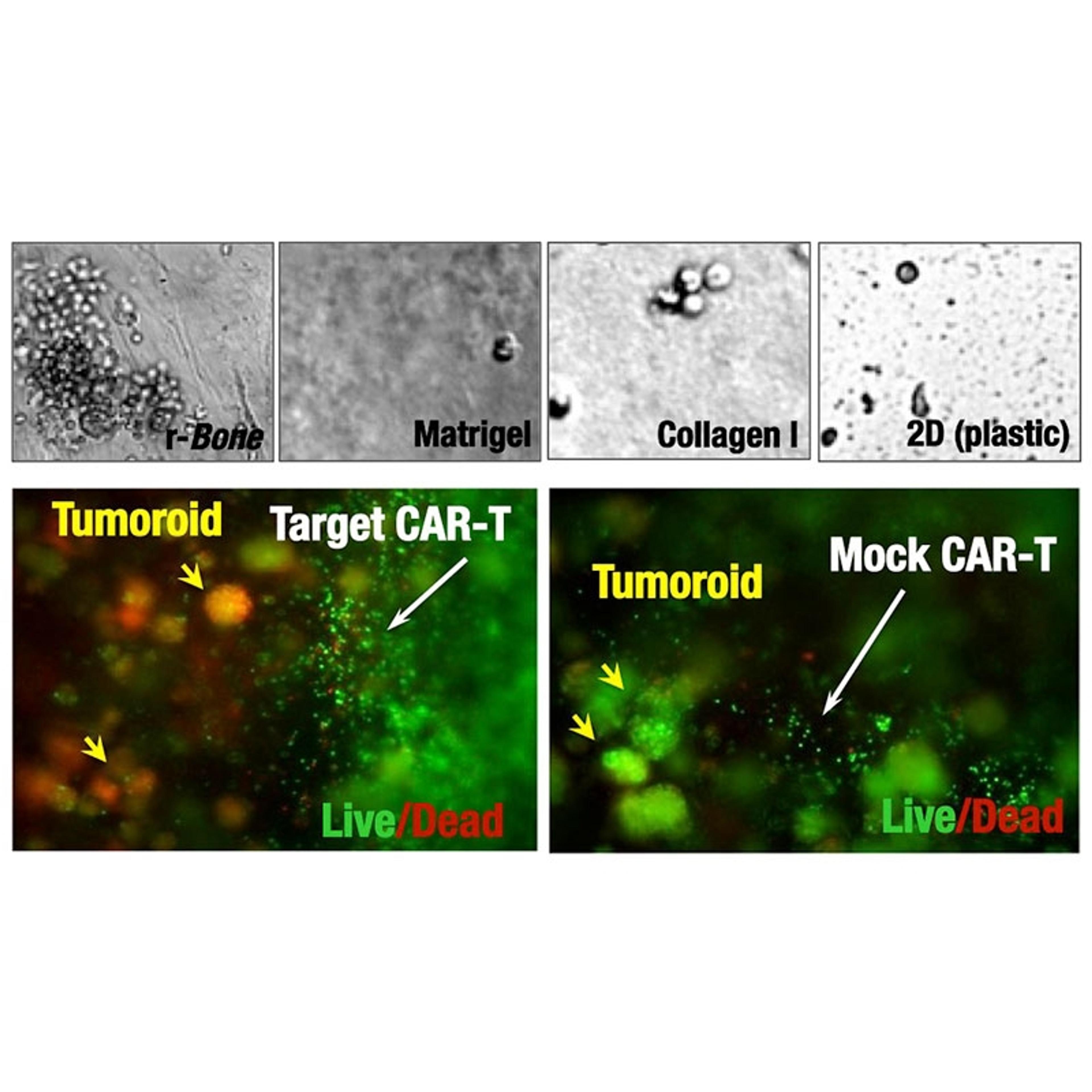
- Bone marrow-specific extracellular matrix (ECM) drives native organization of primary multiple myeloma cells ex vivo.
- Multiple myeloma medium supplement enables reconstruction of a disease-specific mileu.
- Long-term survival of primary patient cells (viability is maintained for >30 days as compared to 1-4 days in standard 2D culture or Matrigel).
- Maintains cellular heterogeneity of the primary human bone marrow.
- Suitable for evaluation of single agents and combination treatments.
- Compatible with multiple therapeutic modalities, e.g. small molecules, antibody-drug conjugates (ADC), immuno-oncology agents, including CAR-T cells.
- Compatible with standard readouts, such as FACs, imaging, bioassays, etc.
Next generation 3D patient-derived cell culture services
In this overview, zPREDICTA demonstrates their 3D culture serivces based on their unique proprietary technology, where organ-specific extracellular matrix and disease-specific medium supplements provide a physiologically relevant microenvironment for accurate testing of anticancer compounds.
Testing immuno-oncology agents in organ-specific 3D cell culture models
In this application note, zPREDICTA demonstrates their 3D culture services based on their tumorspecific technology, where tissue-specific extracellular matrix (ECM) and tumor-specific medium supplements provide a physiologically relevant tumor microenvironment for accurate testing of anticancer compounds.
r-Bone: 3D culture platform for testing agents against AML and multiple myeloma
In this application note, zPREDICTA demonstrates their reconstructed bone marrow (r -Bone) platform, which holds the ability to sustain long-term viability and preserve proliferative properties of primary bone marrow. r-Bone replicates the comprehensive 3D microenvironment of the human bone marrow from patients with multiple myeloma and AML, allowing for more in-depth research to be performed.
Reconstructed Bone (r-Bone™) technology
In this application note, zPREDICTA describes the Reconstructed Bone (r-Bone™) technology, a patient-derived 3D tissue culture model where the human bone marrow microenvironment is reconstructed in vitro. This technology is capable of supporting long-term survival and expansion of primary multiple myeloma cells and cells from other hematological malignancies, meaning the complex interactions between cells and their microenvironment may be studied.