RapiDxFire qPCR 5X Master Mix GF
Formulated with diagnostic kit developers in mind, RapiDxFire™ qPCR 5X Master Mix glycerol free (GF) can be combined with gene-specific primers and hydrolysis probes for immediate use in high-throughput laboratory developed test (LDT) workflows or further processed for lyophilisation for use in point of care (POC) devices. Provided at a 5X concentration without passive reference dye, this flexible master mix allows more room…
Formulated with diagnostic kit developers in mind, RapiDxFire™ qPCR 5X Master Mix glycerol free (GF) can be combined with gene-specific primers and hydrolysis probes for immediate use in high-throughput laboratory developed test (LDT) workflows or further processed for lyophilisation for use in point of care (POC) devices. Provided at a 5X concentration without passive reference dye, this flexible master mix allows more room for sample and up to 5 target detection at one time. The RapiDxFire qPCR 5X Master Mix, manufactured in an ISO 13485-certified facility, is available in small development sizes to large scale production batches to assist you throughput the diagnostic test development process.
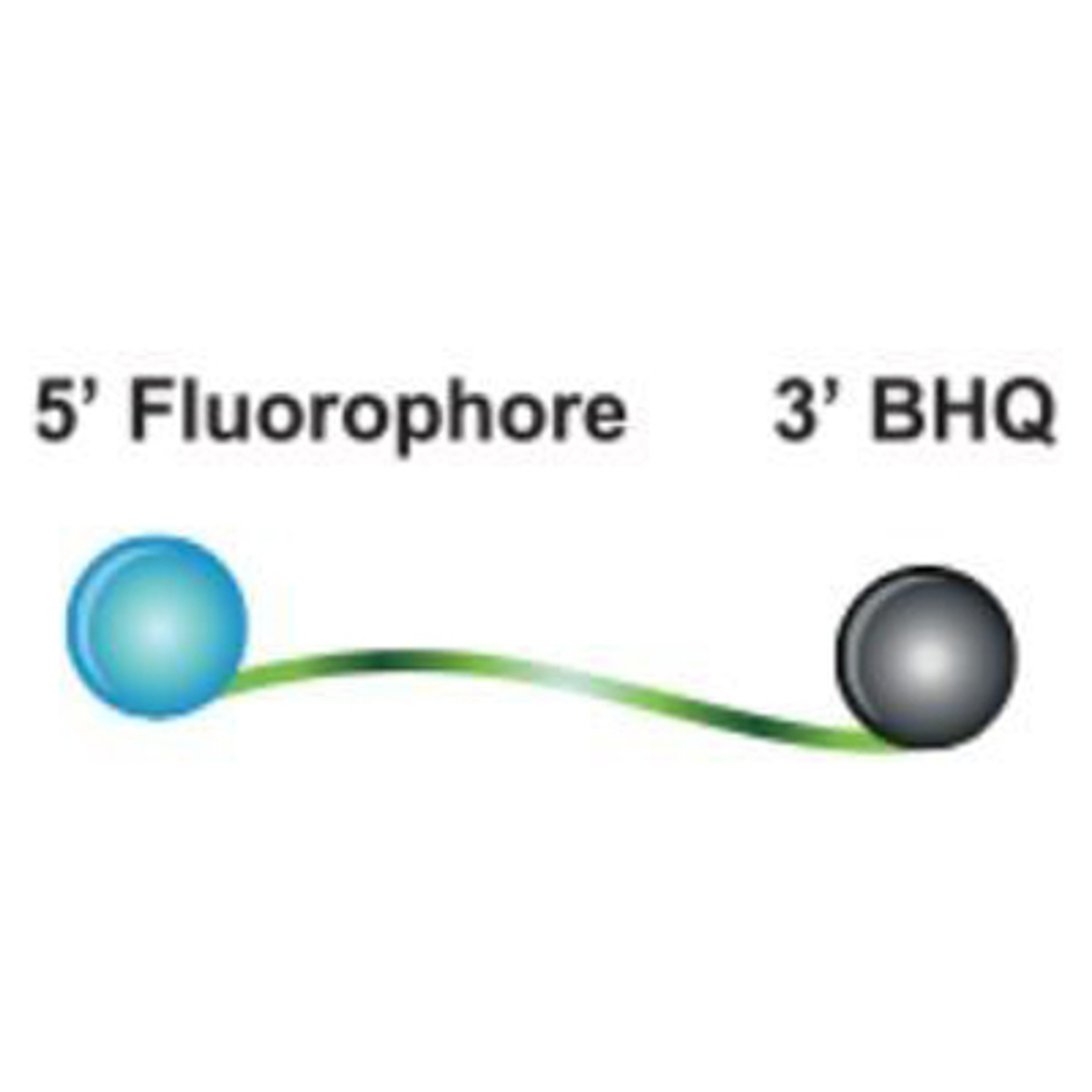
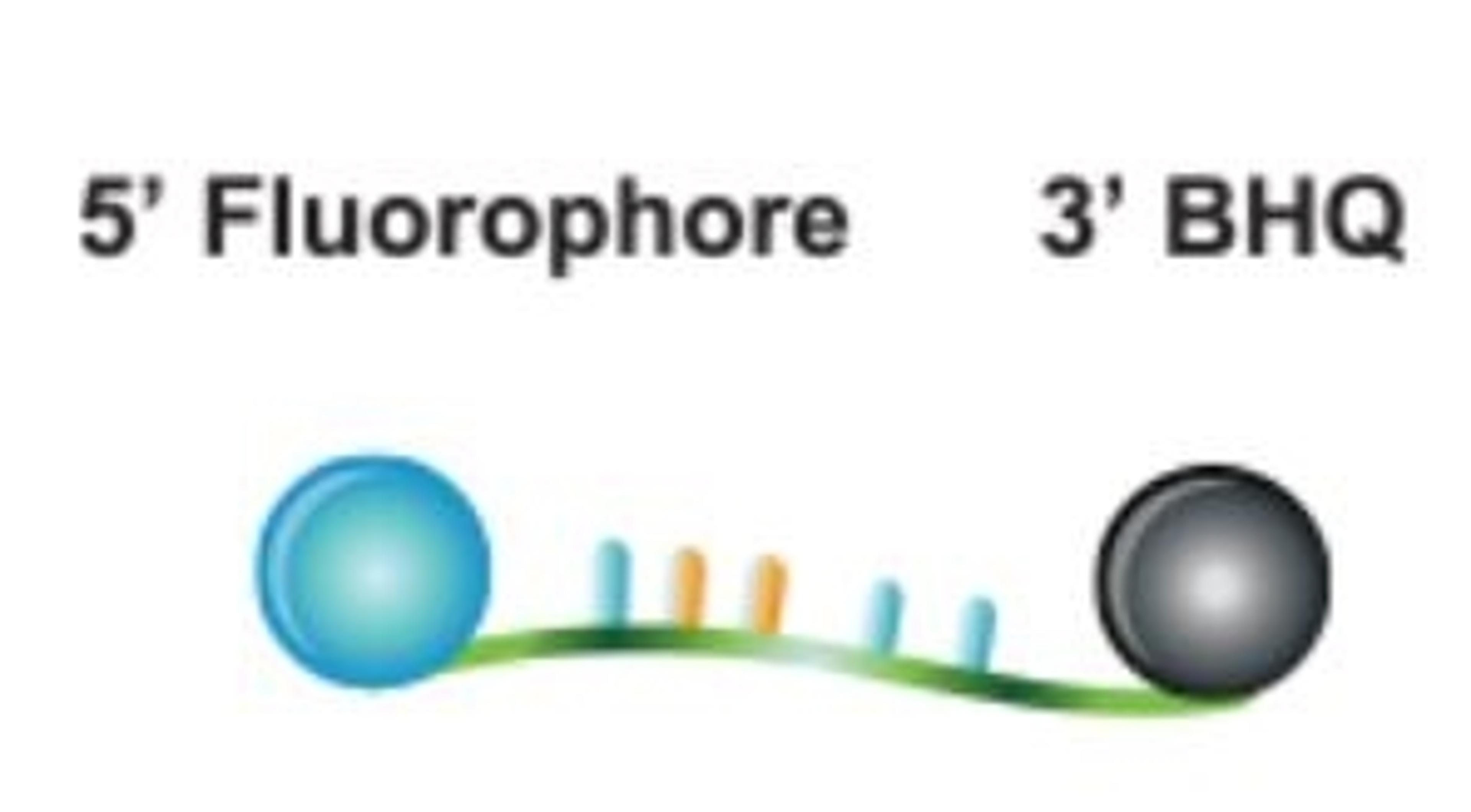
RapiDxFire qPCR 5X Master Mix GF has been optimised for use with all Biosearch Technologies’ probes (Dual-Labelled BHQ, BHQplus, BHQnova and BHQplex CoPrimers), which all contain Biosearch Technologies’ proprietary BHQ quenchers. BHQ dyes have been proven to dramatically reduce low-level background fluorescence due to highly efficient static quenching between reporter and quencher. BHQ dyes are compatible with reporter fluorescent dyes that span the visible spectrum, allowing for broad flexibility in fluorophore selection. RapiDxFire qPCR 5X Master Mix GF can also be used with any probe-based assay. In addition, the Biosearch Technologies RealTimeDesign software is available online to facilitate the design of qPCR assays. Research use only not for use in diagnostic procedures.
High-performing, lyo-compatible master mix for sensitive pathogen detection.
- 5X dye-free formulation offering flexible reaction setups and protocols
- Sensitive detection down to ~10 genomic DNA copies
- Wide dynamic range for multiplexing
- 48-hour reaction benchtop stability ideal for automated workflows
- Glycerol-free, Triton-free, high concentration, and bulk formulations for adaptable test development and lyophilisation options.
- Manufactured in an ISO 13485-certified facility demonstrating batch to batch reproducibility
- Part of the RapiDxFire 1-step RT-qPCR system for effective detection of viral RNA pathogens
Enabling room temperature shipping and storage through compatibility of RapiDxFire qPCR 5X Master Mix GF with lyophilisation
Lyophilization, or freeze drying, is a low-temperature dehydration that involves freezing a reagent at a low temperature, lowering pressure, and then removing any ice by sublimation. The resultant product is either a bead or a cake-like substance and, after rehydration, can be used the same way as before lyophilization. In this application note, LGC Bioresearch Technologies illustrates the suitability of the RapiDxFire™ qPCR 5X Master Mix for lyophilization, and the downstream applicability of this mix to a broader scope of molecular diagnostics workflows.
Developing the 5-minute point-of-care PCR test: A technology revolutionising molecular diagnostics
With the challenges faced at the peak of the COVID-19 pandemic still very fresh in the memory, delivering point-of-care testing with speed, convenience, and reliability remains a pressing need in primary care. A resurgence in other respiratory pathogens, including influenza A and B, has left no respite for urgent care centers, general practice, and pharmacies as their testing services remain under pressure. The development and implementation of a PCR platform that could produce results in just minutes rather than hours would therefore be transformative.
In this SelectScience® webinar, Andrew Anyakwo, Head of Reagent Development at LEX Diagnostics, will give a unique insight into how they have been able to achieve just that - developing a low-cost, point-of-care PCR test that aims to diagnose SARS-CoV-2 and Flu A/B in just 5 minutes with high sensitivity and specificity.
Key learning objectives
- Understand the challenges the rapid development of assays/devices presents, how they can be overcome, and what can be learned in the process
- Gain insight into how to deal with inhibitors
- Explore the benefits of partnering with a manufacturer to develop a solution with GMP at scale
- Discover how PCR can be improved upon to revolutionise point-of-care molecular diagnostics
Who should attend?
Assay developers and scientists working within the molecular diagnostics field.
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
Tackling HPV detection and the global cervical cancer prevalence problem
Explore the concerted effort healthcare professionals, pharma, and biotech are making to combat cervical cancer through the development and implementation of new HPV assays