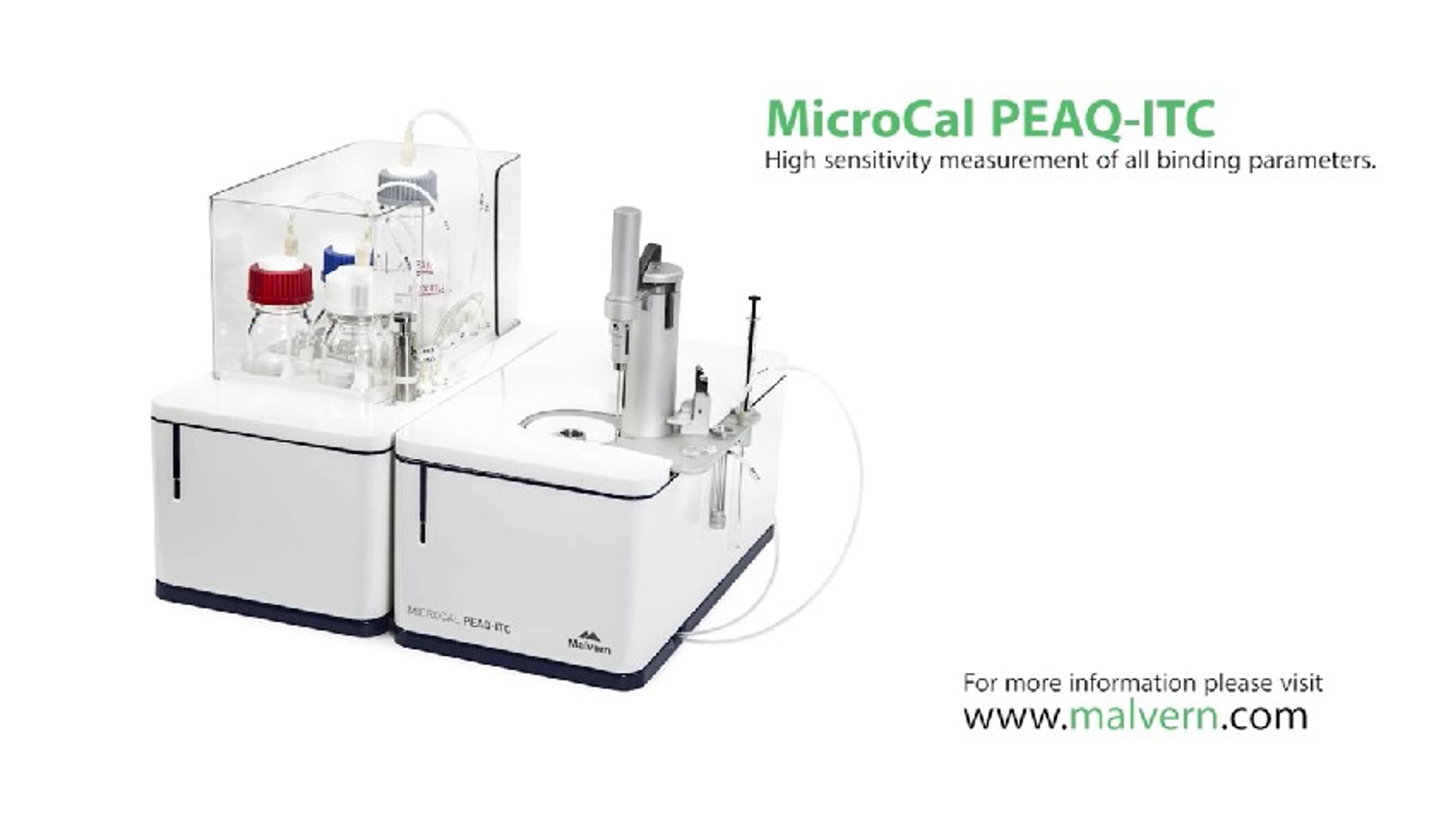
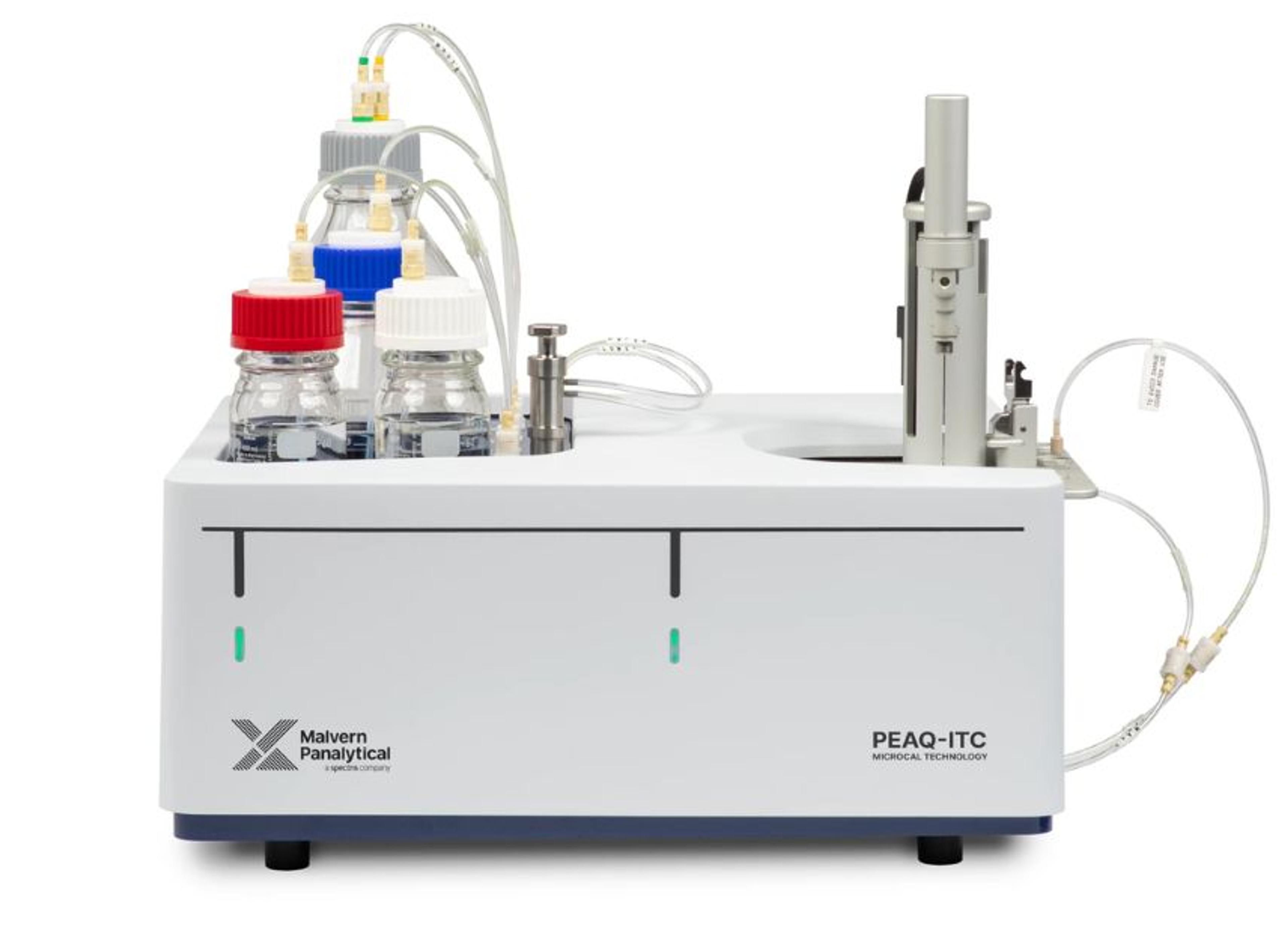
MicroCal PEAQ-ITC Automated
High productivity measurement of multiple binding parameters.
Great outcomes It is at the core of my study.
Analyze Binding interactions of lectin-glycopeptides
I am obtaining the thermodynamic parameters for my research utilizing Malvern's very sensitive ITC equipment. I was able to produce findings of a very high caliber, and Malvern's PEAQ ITC program is fantastic; I adore it. Although it is difficult to analyze the samples because many precautions must be taken, it is acceptable because we are only interested in binding affinities that are micromolar or lower because everything counts at that point. Additionally, customer support is excellent. Whenever I write about a problem, I always receive a response within 24 hours.
Review Date: 19 May 2023 | Malvern Panalytical
From less amount of sample can determine the interaction studies
Binding interaction studies
Malvern Microcal PEAQ-ITC Automated system is very easy to use. I love this instrument very much. Software for analysis is also very easy to determine the binding interaction studies.
Review Date: 10 Feb 2023 | Malvern Panalytical
Manufacturer's Response
Thank you so much for taking the time to send in the review. It was very kind of you. The development team put a lot of work into making the user experience and enjoyable one and will be so pleased to hear that you 'love it'.
We got excellent results and we were able to get some good publications.
Protein drug binding
When we started the protein-drug binding study, we were had the impression 'How do we handle and interpret the data?', but as we went along it was very convenient to perform the experiment and do data analysis. I have been using this machine since my Ph.D. days. It has undergone huge modifications and advances. Excellent instrument !
Review Date: 8 Dec 2020 | Malvern Panalytical
Manufacturer's Response
Thank you for your kind words. The instrument was designed with protein-drug interactions in mind and so it was hugely gratifying to hear that it supported your research in that important field. Thanks again.
Very good instrument, fast and easy to use.
Binding experiments with macromolecules (proteins, peptides)
The instrument is very easy to use, there are videos demonstrating everything you need to use it. Loading is a bit difficult, but because it uses very little material it is not a huge deal to miss a few injections until you get used to loading efficiently. In one day it is possible to get 5 or 6 experiments done, if not more - so very fast compared to other ITC instruments. I find it very reproducible as well. I am overall very happy with the instrument, since I was able to obtain data in a really short time, including binding and kinetic experiments. The only issue I have with it is that the service contract is on the expensive side. It is possible to do routine maintenance without service (there are also videos for that), and Malvern has good phone technical service so overall it is not a major set back since most service contracts are expensive.
Review Date: 31 Jan 2017 | Malvern Panalytical
Manufacturer's Response
Thank you very much for your feedback
The MicroCal PEAQ-ITC Automated is a configurable, low volume, highly sensitive isothermal titration calorimeter which offers the convenience of unattended operation. It delivers direct, label-free in solution measurement of all binding parameters in a single experiment. Applications include characterizing molecular interactions of small molecules, proteins, antibodies, nucleic acids, lipids and other biomolecules. It can also be used to measure enzyme kinetics.
The system directly measures heat released or absorbed during biochemical binding events, from which it calculates binding affinity (KD), stoichiometry (n), enthalpy (ΔH), and entropy (ΔS). Other than preparing samples and reagents to appropriate concentrations, there is minimal assay development and time to first result is fast. Easy-to use software includes optimized automation scripts for robust operation with a variety of sample types and conditions.
Features:
- User-friendly design tools assist in selecting experimental run parameters.
- All binding parameters (affinity, stoichiometry, enthalpy and entropy) in a single experiment.
- Quick to first result with minimal assay development, no labelling, no immobilization and no molecular weight limitations.
- High signal to noise gives more confidence in accessing data quality and relevance of generated affinity and thermodynamic parameters.
- Outstanding data quality for sub-millimolar to picomolar disassociation constants (10-2 to 10-12 M).
- Coin shaped cell optimizes sample mixing.
- Nonreactive Hastelloy for chemical resistance and compatibility with biological samples.
- Sensitivity to investigate any biomolecular interaction using as little as 10µg of protein.
- Samples presented in temperature controlled 96-well plate.
- Fully automated system with walk-away operation for enhanced productivity.
Brochures
MicroCal ITC Range brochure
This product brochure introduces the MicroCal ITC range by Malvern Panalytical.
Enzyme kinetics assays with MicroCal ITC systems
In this application note, discover how to perform enzyme kinetics experiments with a MicroCal isothermal titration calorimetry (ITC) system.
MicroCal ITC Systems: Understanding Biomolecular Interactions
Isothermal titration microcalorimetry (ITC) is an essential tool in drug discovery and the study and regulation of protein interactions. This brochure describes how Malvern MicroCal ITC calorimeters are designed to deliver the exceptional performance and outstanding quality data needed in these application areas.
Biophysical characterization of epigenetic protein interactions with chromatin using isothermal titration calorimetry
In this application note, Malvern Panalytical discuss how isothermal titration calorimetry (ITC) can be used to determine the contribution of individual protein residues or modifications to the total binding affinities of complex substrates. Incorporating binding, thermodynamic and stoichiometry data from ITC, structural studies like NMR and X-ray crystallography, and in vivo studies, will ultimately lead to a complete understanding of these complex interactions.
Practical Tips for MicroCal PEAQ-ITC Experiments
In this application note, practical tips are provided for scientists conducting Isothermal titration calorimetry. These tips pertain to things such as:
- The removal of bubbles
- Residual solution in the ITC cell
- Remaining sample solution in a cell reservoir
- Importance of cleaning the cell reservoir and pipette retaining nut
- Washing and rinsing error troubleshooting
Biophysical Application of Calorimetric Methods to Protein Misfolding And Aggregation Examinations
Unfolded proteins fold into thermodynamically-favorable native structures under the correct physiological conditions for gain of function. This whitepaper describes how differential scanning calorimetry and isothermal titration calorimetry can be used as a powerful method for the thermodynamic investigation of interactions between diverse biomolecules and inorganic compounds. It details some examples from literature of the methods application in studying amyloid fibrillation.
Analysis of Demicellization Data from Isothermal Titration Calorimetry
This whitepaper outlines how two different approaches can be combined to provide a data-analysis strategy of superior reproducibility and without user bias, as the only user input required is the calorimetric dataset itself. Many industrial and biochemical applications depend on the usage of detergents and other surfactants. These compounds are indispensable, in particular, for the extraction, purification, and handling of membrane proteins. Owing to their amphiphilic nature, detergents can provide a membrane-mimetic environment required by integral membrane proteins to retain their native structures and functions in aqueous solution. The CMC is one of the most fundamental characteristics of a surfactant. In aqueous solutions, surfactants form colloidal aggregates, so-called micelles, when their concentration exceeds the CMC. To obtain the CMC in an accurate and precise manner, however, isothermal titration calorimetry (ITC) is the method of choice, which is due to its extraordinary sensitivity, unsurpassed resolution, and high reproducibility.
Malvern Instruments: The Theory and Practice Behind Isothermal Titration Calorimetry
Isothermal titration calorimetry (ITC) is an analytical technique that has become the “gold standard” for studying intermolecular interactions. As its name indicates, it is a titrimetric technique, that is, a volumetric laboratory method for quantitative chemical analysis (traditionally intended to determine the unknown concentration of an identified analyte) where a reagent solution, the titrant, is made to react with a solution of analyte or titrand. This white paper details the use of ITC in biophysical studies.
Addressing the Needs of Drug Discovery with the MicroCal PEAQ-ITC Instruments
Measurements and characterization of binding interactions between proteins and low-molecular weight ligands are fundamental for drug discovery. Among the most recognized challenges in characterizing binding interactions are (1) the need to accurately assess a wide span of binding affinities (KD) and (2) accurately rank and characterize low-molecular weight (LMW) ligands based on affinity, mechanism of action, and energetics of interaction.
Use of the New MicroCal PEAQ-ITC System for Measurement and Characterization of a Broad Range of Protein-LMW Compound Interactions
Measurement and characterization of binding interactions between proteins and low-molecular weight (LMW) ligands are a focus of academic research and drug discovery. Isothermal titration calorimetry (ITC) directly measures heat released or absorbed in a binding event, providing means for studying protein-small molecule interactions in solution without the need for labeling or immobilization. Importantly, ITC is often utilized to characterize differences in entropic and enthalpic contributions to binding of novel ligands.
Interaction analysis masterclass with Malvern Panalytical
This Malvern Panalytical masterclass delves into the intricacies of interaction analysis and explores how ITC and GCI are complementary and orthogonal techniques for all early-stage drug discovery needs. Discover how ITC and GCI technologies can be applied to complex targets and interactions in a series of applications from hit identification to lead-optimization.
Exploiting the biophysical sweet spot in fragment screening
Over the last twenty years, Fragment-based drug design (FBDD) has emerged as a prominent strategy. Yet FBDD is not without challenges, including commercial libraries often hampered by excessively sized fragments. Recently new assaying techniques have emerged that push the boundaries of what’s possible with fragment screening, offering collections specifically tailored to biophysical screening. In this webinar, two respected industry authorities reveal how next-generation FBDD fragment screening can assist biophysical researchers more efficiently identify and prioritize the best fragment hits, compress hit-to-lead campaign times, and reduce research cost and raise lead throughput.
MicroCal PEAQ-ITC - Advanced Isothermal Titration Calorimetry
Learn more about the MicroCal PEAQ-ITC, the most advanced Isothermal Titration Calorimetry system from Malvern Instruments, which offers high quality data, ease of use, and exceptional sensitivity. Suited to life sciences and drug discovery applications, this microcalorimetry instrument is perfect for researchers wishing to study biomolecular interactions.
Join Malvern For Expert Help on Protein Interaction Analysis with Microcal PEAQ-ITC at the 29th Annual Symposium of The Protein Society
Resolving challenges, finding solutions