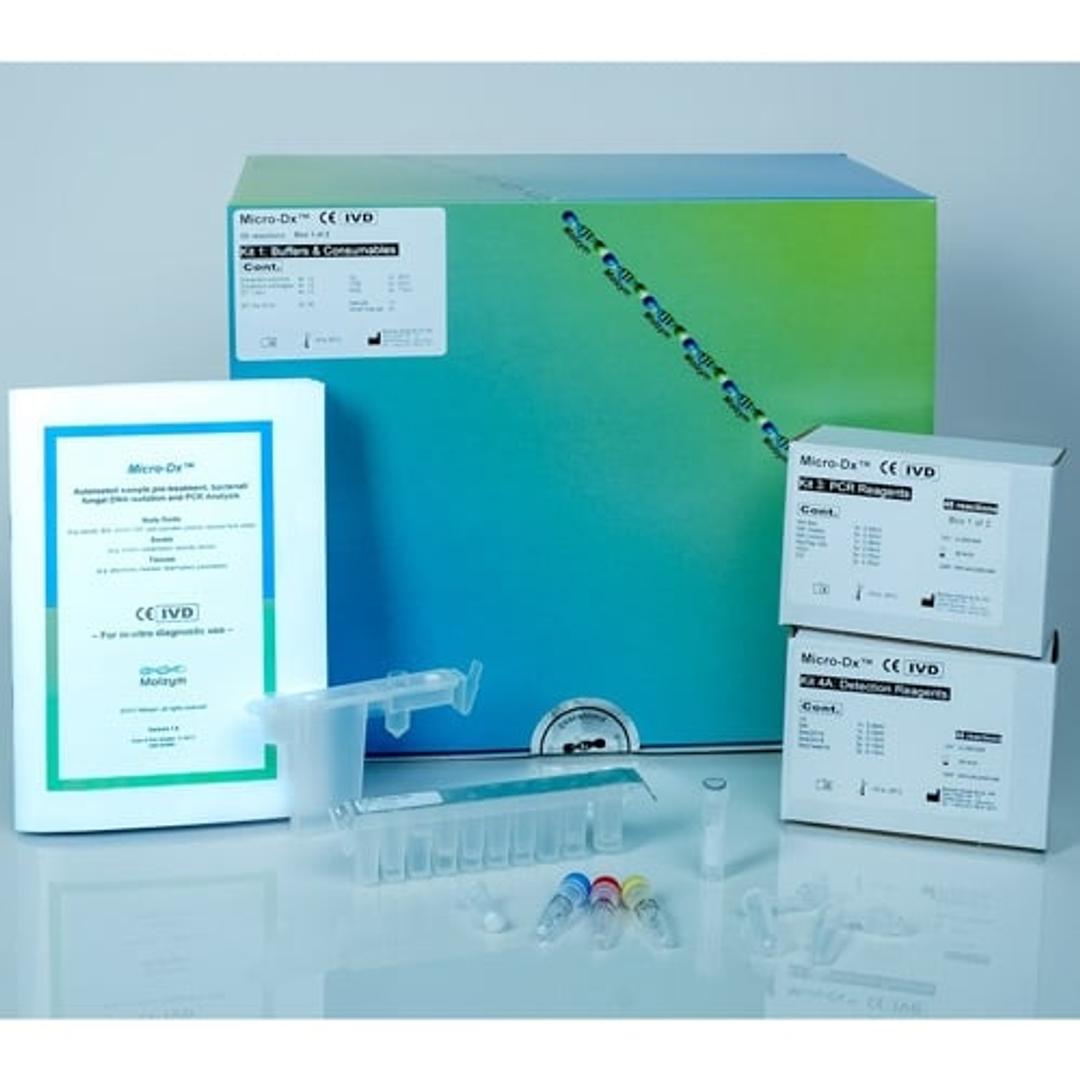
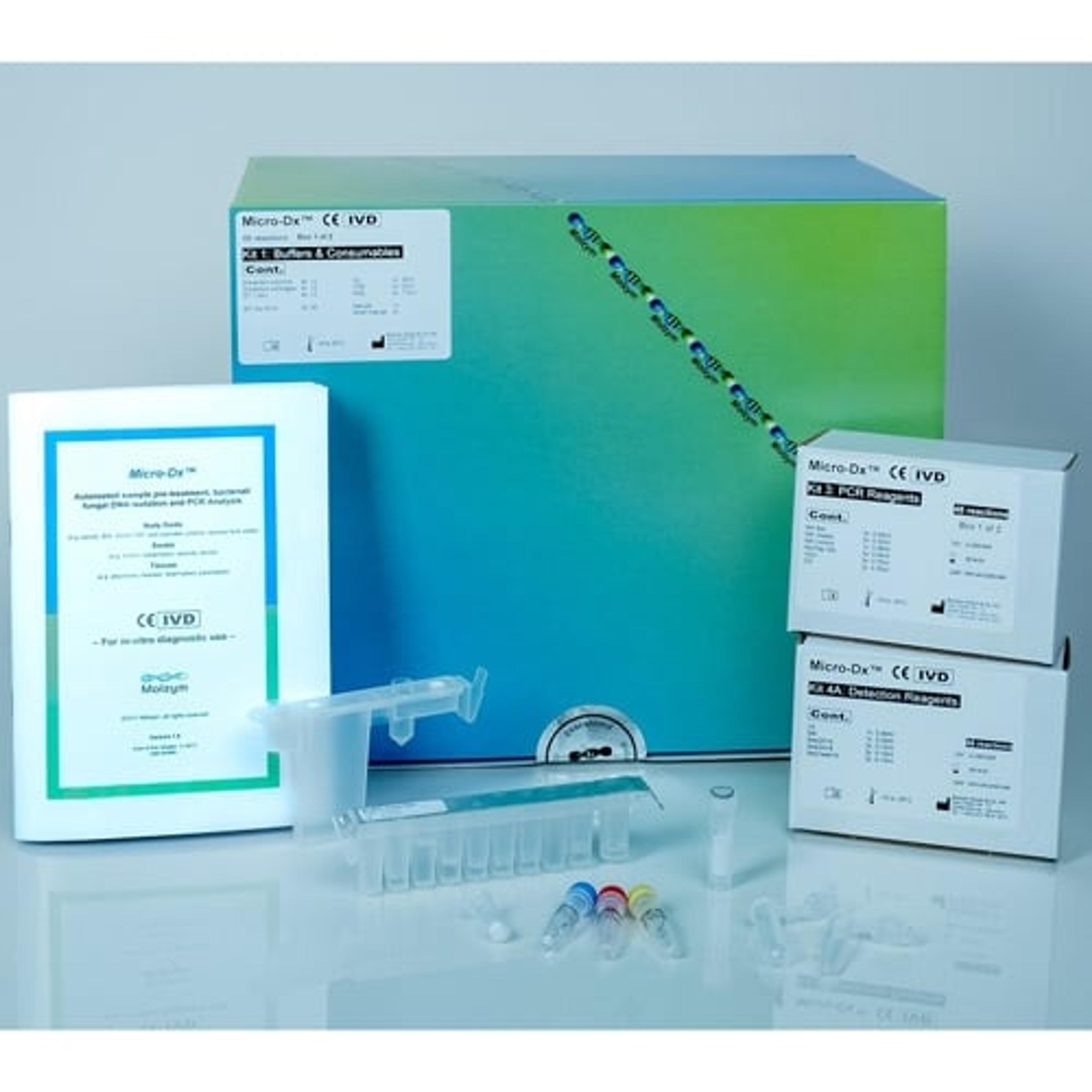
Micro-Dx™ CE IVD
Fully automated pathogen DNA isolation and broad-range PCR analysis from body fluids, swabs and tissues for clinical routine diagnostics

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Micro-Dx™ CE IVD is designed for pathogen detection and identification from body fluids, swabs and tissues independent from culture. The fully automated sample preparation includes host DNA depletion for a more sensitive detection of bacteria and fungi in clinical samples. The broad-range PCR assays deliver first results after approximately four hours. Subsequent Sanger sequencing can identify the pathogens down to species level.
Benefits:
- Reduced hands-on time
- Variety of specimens: body fluids, swabs, tissues
- Flexible: 1- 12 samples
- Increased sensitivity by depletion of host DNA
- DNA-free reagents and consumables
- Broad-range PCR assays for the detection of bacterial and fungal DNA
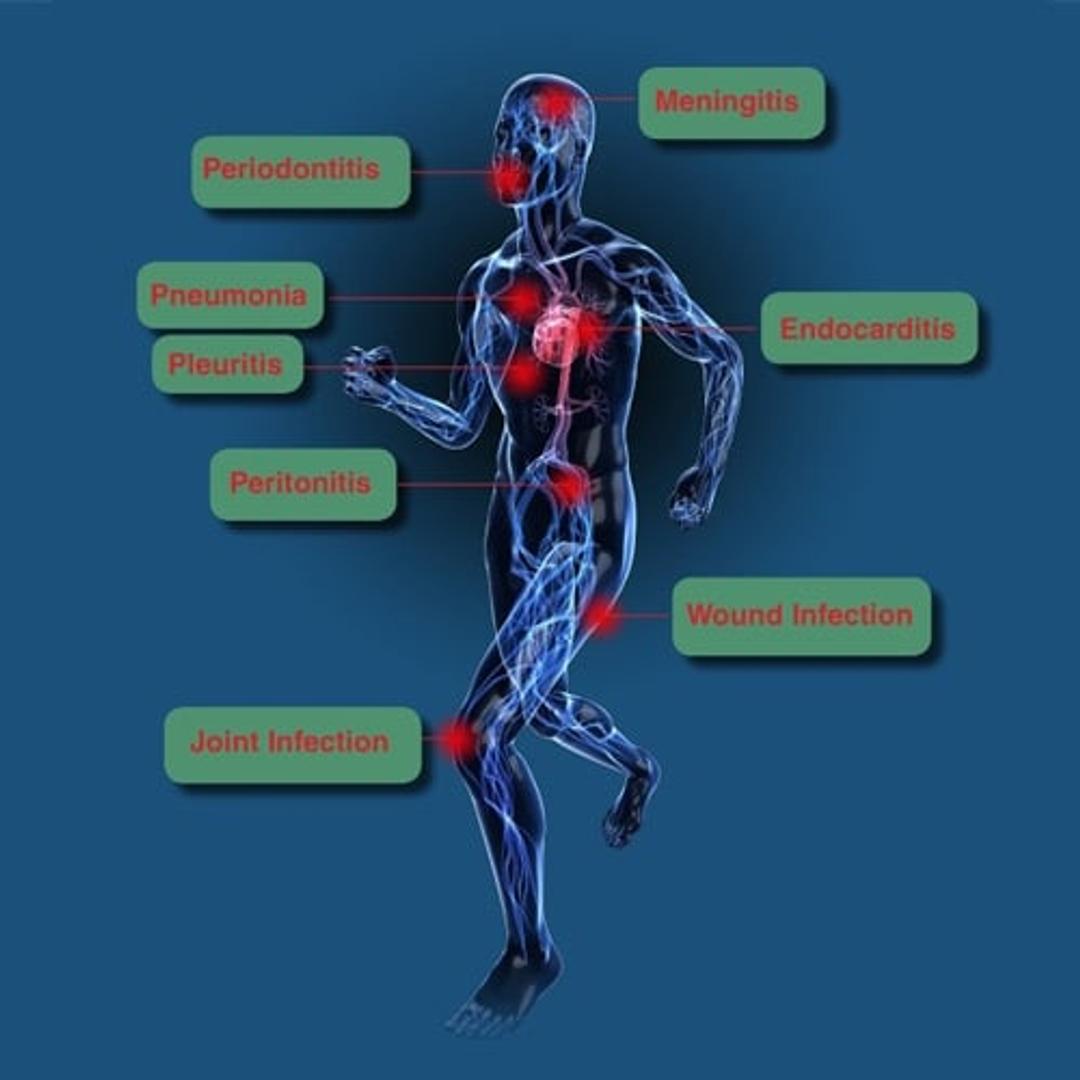
In most samples, host DNA is in vast excess to target microbial DNA which often reduces the sensitivity of molecular analysis significantly. Therefore, a reduction of host DNA leads to an increased sensitivity in the detection of microbial DNA. The DNA-free reagents provided with the kit and the automated DNA isolation commit to the contamination-free processing of the samples. 16S and 18S rDNA-based broad-range PCR assays are used for the sensitive detection of bacterial and fungal DNA, respectively. More than 1.300 different pathogens have already been detected in clinical studies and evaluations. Micro-Dx™ can identify pathogens causing e.g. sepsis, bacterial meningitis, pneumonia, joint and wound infections.
Brochures
Culture-independent molecular detection of pathogens
Molecular analysis of microbes directly from samples can be challenging as the contained human DNA compromises the sensitivity of broad-range assays. Molzym has introduced the Micro-Dx™, a unique CE IVD test for culture-free diagnosis of bacterial and fungal targets directly from samples. It includes an automated protocol for the depletion of human DNA prior to microbial DNA extraction and purification. For the detection of microbial DNA, 16S & 18S rDNA broad-range PCR assays are used, with a first result promised after only four hours. Sanger sequencing and BLAST analysis are applied to identify the pathogens on the species or genus level.
Micro-Dx: Culture-Independent Molecular Diagnosis of Pathogens
Fast and precise culture-independent diagnosis by innovative pathogen enrichment and broad-range PCR and sequencing.
Unlock fast, in-depth pathogen insights with nanopore sequencing
Discover how Molzym's Micro-Dx™ kit, combined with Oxford Nanopore Technology (ONT), enables rapid and comprehensive 16S rRNA-based pathogen identification.
In this technical note, molzym showcases a validated CE IVD-marked workflow for isolating microbial DNA and amplifying bacterial 16S rRNA using broad-range PCR. The study compares multiple bioinformatics approaches for taxonomic classification against known species and Sanger sequencing, providing clear insights into performance and accuracy.
Improving orthopedic diagnostics: The impact of human DNA depletion combined with 16S/18S PCR and sequencing
Identification of the causative agent(s) in bone and joint infections (BJIs) is a crucial but important challenge. Rapid pathogen identification and targeted treatment are critical to prevent serious complications such as bone destruction, joint damage, and sepsis. A considerable number of BJIs are culture-negative or mistakenly identified as aseptic failures, even when diagnostic culture techniques are correctly performed. Molzym presents a diagnostic workflow for the culture-independent detection and identification of bacteria and fungi directly from samples. This workflow offers significant value for the analysis of culture-negative orthopedic samples.
Clinical applications of Molzym’s broad-spectrum molecular diagnostics
Culture diagnosis is a standard method for identifying infectious agents in clinical microbiology, though it is often criticized for its lengthy time to results. Broad-spectrum antibiotics are frequently administered until species identity is confirmed, due to the slow nature of culture. Another limitation is its low positivity rate, often caused by initial antibiotic use or the specific growth requirements of certain microorganisms. Modern molecular methods detect microorganisms by analyzing their DNA, offering speed, sensitivity, and reproducibility without the need for cultivation. However, they are limited in the number of microorganisms they target. Molzym proposes a broad-range approach to overcome this limitation, and presents results from multi-site studies that support its Molecular Diagnostic Test (MMDx) as a precise and rapid tool for identifying pathogens directly from samples. MMDx has the potential to optimize laboratory workflow and provide clinically relevant information to improve patient outcomes.
Broad-range molecular detection of rare and fastidious pathogens
Molzym presents case reports and studies on the diagnosis of various culture-negative bacteria and fungi in which MMDx enabled the identification of, for example, Candidatus Neoehrlichia mikurensis, Malassezia restricta, Bartonella spp., and Tropheryma whipplei, as well as other fastidious or anaerobic, slow-growing or non-cultivable microorganisms. Download this application note to find out how you can utilize the MMDx broad-range diagnostic workflow in your research.
Eubacterial and Panfungal PCR Diagnosis Using Automated DNA Extraction
Automation in routine molecular pathogen testing is crucial for the provision of timely and accurate results. Handling errors with resulting false findings can be greatly reduced by robotic solutions. Micro-Dx™ is the new kit developed for the in vitro diagnosis of microbial pathogens without the need for cultivation. This application note demonstrates a comprehensive clinical evaluation of the system from a multi-site study.
Rare Pathogen Identification with Molzym's 16S rRNA Gene Diagnosis
In this white paper, Molzym discusses its approach to tackling the current challenges with identifying rare pathogens in clinical diagnostics. With the aim to support tailored antibiotic regimens for patients with critical conditions, the method described, which incorporates 16S rDNA PCR and sequencing analysis, should deliver precise strain identification within hours.
Enhanced Diagnostics in Orthopedic Infections: Overcoming the Limitations of Culture with Molecular Testing
Accurate diagnosis of orthopedic infections is crucial for successful patient treatment. Bone and joint infections (BJI) are particularly challenging, with up to 50% of cases reported as 'culture negative'.
Marina Linow, product manager, Molzym GmbH & Co. KG, will explore studies comparing traditional culture-based methods with advanced molecular techniques to enhance diagnostic accuracy. She will examine the limitations of conventional cultures, as well as the factors influencing molecular testing and their impact on the accuracy and efficiency of culture-independent molecular diagnostics.
Key learning objectives
- Understand the constraints of traditional culture methods
- Identify the variables that affect the accuracy and efficiency of culture-independent molecular tests
- Understand the diagnostic impact of human DNA depletion for culture-independent infection diagnosis
Who should attend?
- Lab directors, lab managers, quality managers
- Medical laboratory scientists and technicians
- Molecular microbiologists
- Clinical lab scientists
- Clinicians, infectious disease Specialists, pathologist, and medical doctor
- Researchers, microbiologists, and biotechnologists
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes
9 top resources for your COVID-19 & infectious disease research
Exclusive interviews, new methods, free downloads, and much more to help you advance your work