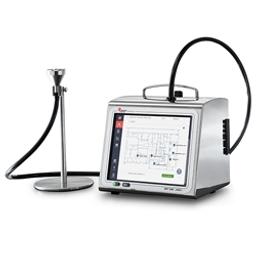
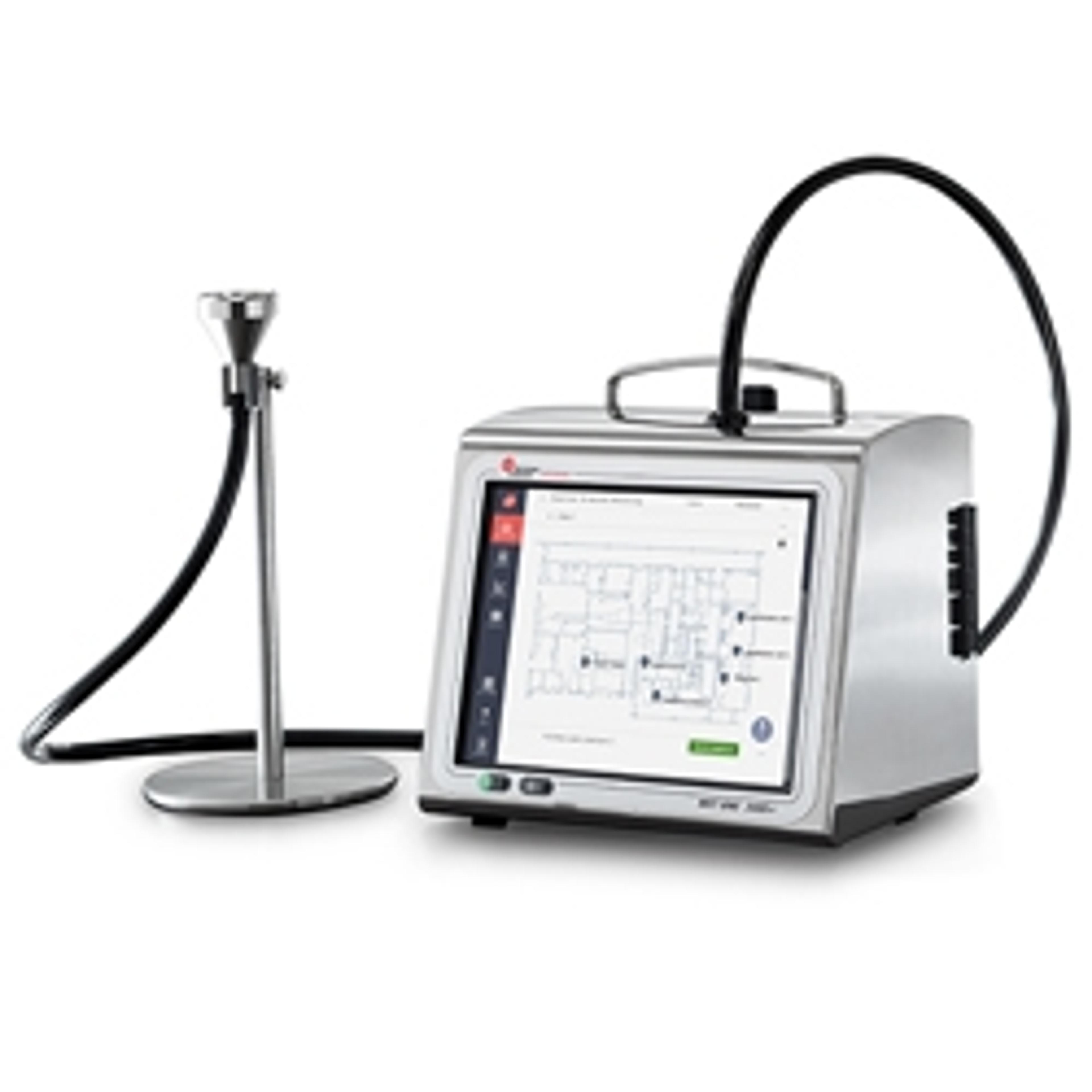
MET ONE 3400+ Series Cleanroom Routine Environmental Air Particle Counter
Our new MET ONE 3400+ Series Portable Airborne Particle Counter allows users to load their routine environmental monitoring SOP sampling map and sampling configuration for each location directly into the counter, so that the SOP becomes an interactive sampling map directly on the counter screen to guide users doing daily sampling. This helps reduce training requirements and sampling data errors/omissions.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Great results from instrument
Particle sizes
Giving perfect scenario of sample and analysis perfectly
Review Date: 18 Jun 2022 | Beckman Coulter Life Sciences
Our new MET ONE 3400+ Series Portable Airborne Particle Counter allows users to load their routine environmental monitoring SOP sampling map and sampling configuration for each location directly into the counter, so that the SOP becomes an interactive sampling map directly on the counter screen to guide users doing daily sampling. This helps reduce training requirements and sampling data errors/omissions.
Accessed remotely via a web browser, features such as review/approve workflow, SOP version control and electronic record export are all in the counter – there’s no external software needed. Secure on-board searchable/filterable Audit Trail provides fast reporting during audits combined with Microsoft Active Directory for User Name and Password control for log-on and electronic signatures to support 21 CFR Part 11 compliance.
Customized Interactive Electronic SOP Maps
Using a web-browser interface, load your SOP map into the counter, mark sample locations, and define SOP sampling requirements for each.
Interactive Tracking
Onscreen instructions help operators navigate your map and perform sampling according to the SOP. Location names turn green after sampling, making it easy to track progress at a glance.
Electronic Records Straight from the Counter
The MET ONE 3400+ can provide electronic records straight from the counter via USB, wired Ethernet or built-in WiFi.
Web-browser SOP Version Control and Review & Approve Workflow
Managers can remotely review daily monitoring progress including all sample results via a web-browser. Remote SOP configuration and version control is also available for administrators, as well as remote review & approve, electronic signature sign-off and electronic record export for supervisors – all via web-browser.
Brochures
MET ONE 3400+: Automate environmental monitoring for GMP cleanroom compliance with one easy decision
In this brochure, Beckman Coulter introduces the MET ONE 3400+, a particle counter that promises to help you manage accurate cleanroom monitoring data via safe and secure internal software and web access—there’s no external software needed.
MET ONE HHPC+ Series handheld particle counters
In this application note, Beckman Coulter Life Sciences highlights the MET ONE HHPC+ Series handheld particle counter designed for routine monitoring in cleanrooms and controlled environments. Its high-resolution display can be customized to show relevant data clearly, and its lightweight design enables single-handed operation, making it suitable for troubleshooting HEPA filter leaks in tight spaces. Additionally, it allows for quick validation of cleanroom classifications, provides visual particle trending for process profiling, and offers easy data transfer via USB memory stick and ethernet for efficient analysis outside of the cleanroom environment.
Optimizing workflow efficiency of cleanroom routine environmental monitoring
In this application note, Beckman Coulter Life Sciences presents the MET ONE 3400 portable air particle counter to streamline biopharmaceutical environmental monitoring. Failure to execute monitoring programs properly can lead to product wastage or regulatory issues. Root causes often include user errors, which can be addressed through automation and instrument-based software assistance. The MET ONE 3400's Simply Paperless system ensures data integrity by providing password-protected logins, legible electronic records, and contemporaneous recording of data. By generating secure pdfs and enabling easy data export, the system minimizes errors associated with manual transcription and data handling.
GMP cleanrooms classification and routine environmental monitoring
Designing and validating your Cleanroom Routine Environmental Monitoring and Classification SOPs is time consuming and complex. This application note discusses the differences between GMP Cleanroom Classification and Routine Environmental Monitoring and explains how Beckman Coulter can help.
Ensure compliance and patient safety using total particle air counting in sterile compounding facilities
April 17, 2025 at 16:00 BST / 17:00 CEST / 11:00 EDT / 08:00 PDT
With the critical role that sterile compounding plays in patient care, ensuring compliance with USP <797> standards and mitigating risks from airborne particulate matter is essential for all sterile compounding operations — including 503A pharmacies, hospitals, infusion centers, and 503B outsourcing facilities. For this reason it is crucial to have adequate implementation to uphold aseptic standards, reduce risks, and protect patients.
In this educational webinar, Dr. Amy Summers, 503A & 503B Compounding Consultant at Restore Health, will give a comprehensive overview of the importance of total particle air counting. Hear real-life case studies that highlight best practices that you can implement to ensure you meet regulatory requirements and maintain a sterile environment.
Key learning objectives:
- The why: Why is total particle air counting crucial for sterile compounding facilities? Understand its role in contamination control, risk reduction, and patient safety as outlined in USP <797>.
- The how: Learn how to use total air particle counters, including best practices for accurate air sampling in ISO-classified areas (ISO 5, 7, and 8).
- Avoiding pitfalls: Learn how to avoid common errors and challenges in air counting, such as incorrect sampling, misinterpretation of data, and maintenance oversights.
- Real-world case studies: Hear examples of sterile compounding facilities implementing robust particle monitoring programs, including lessons learned and benefits achieved.
- Regulatory alignment: Understand how to align particle monitoring programs with the updated USP <797> requirements, focusing on maintaining ISO air quality classifications and proper documentation.
Who should attend
- Pharmacists and pharmacy technicians
- Compounding supervisors
- Quality and compliance professionals in 503A/503B facilities, hospitals, and infusion centers
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.
Annex 1 revision at a glance
In this video, Beckman Coulter Life Sciences highlights the Annex 1 revision document. The topics include main principles, quality risk management and contamination control strategy, and the creation of the framework around the processes. Furthermore, the video also highlights realization of the processes, environmental and process monitoring and quality control and testing requirements.
7 essential resources in pharmaceutical quality assurance
Explore the latest technologies, free downloads, and expert insights that can help your laboratory achieve compliance successfully
Advances in materials characterization, from graphene production and correlative imaging to transmission Raman spectroscopy
Explore cutting-edge technologies and the latest resources to support your materials characterization