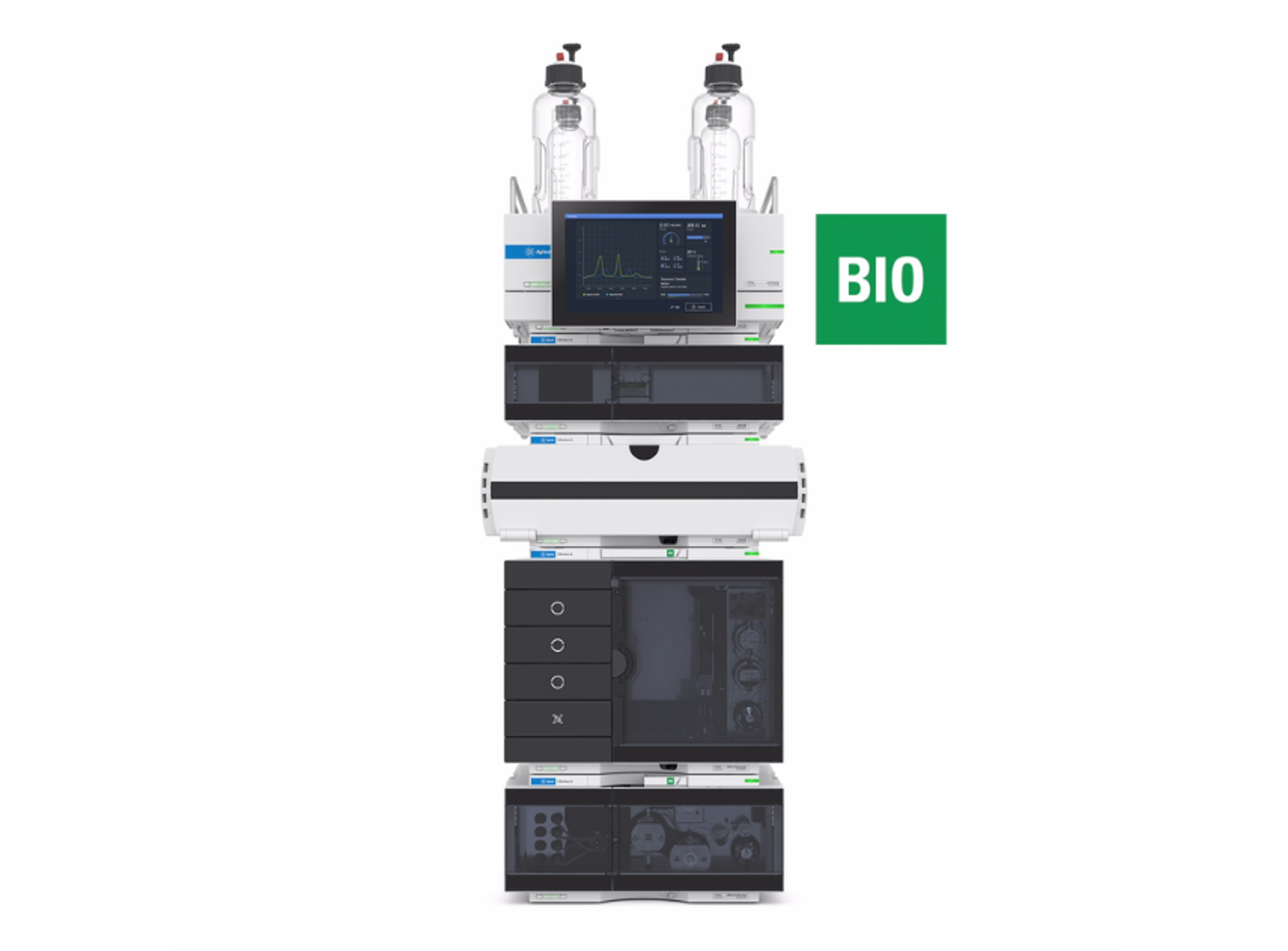
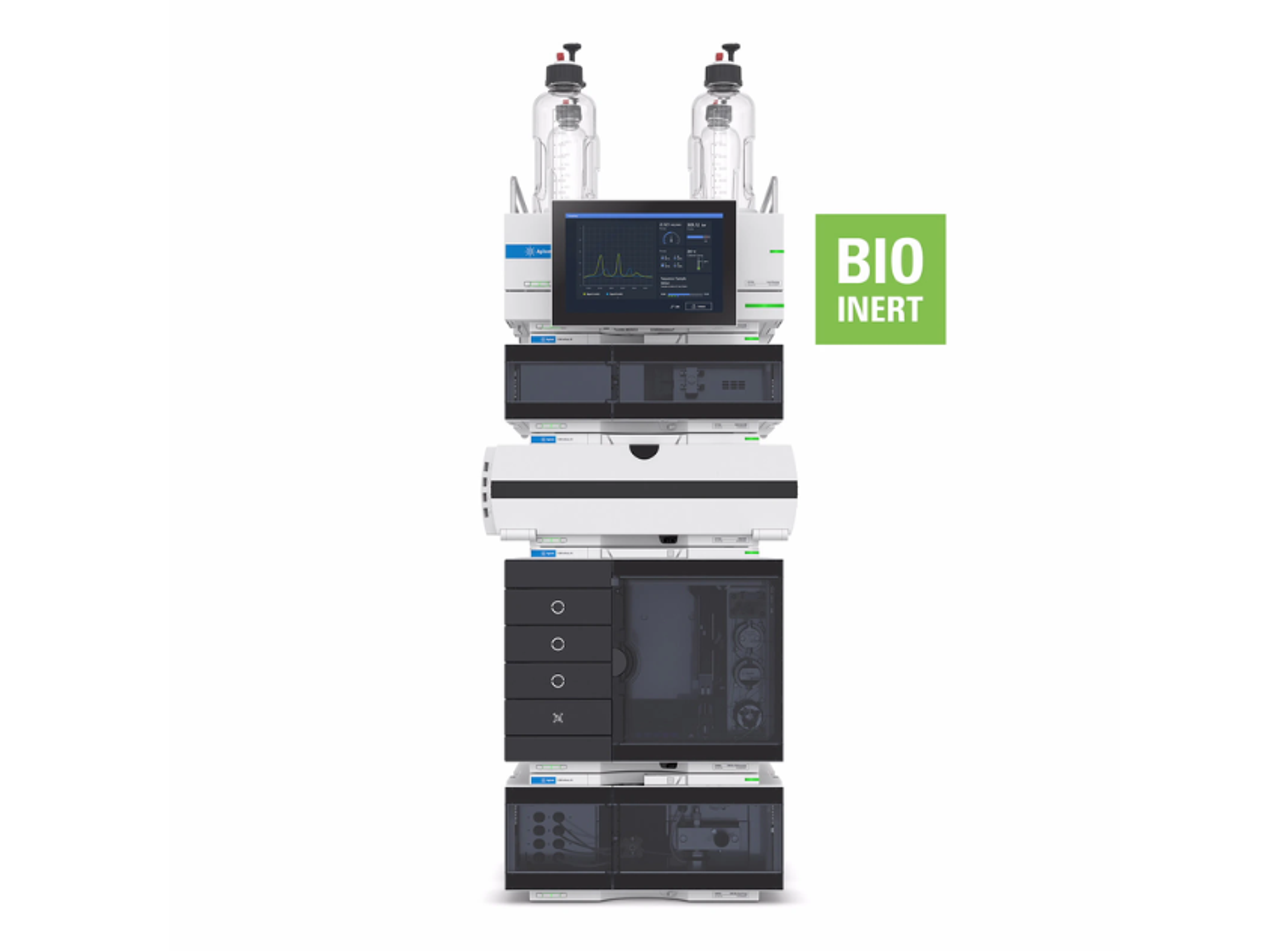
Agilent 1290 Infinity II Bio LC System
The 1290 Infinity II Bio LC System is a UHPLC, binary or quaternary ,- consisting of biocompatible material for use in biopharma (e.g., critical quality attributes) and other applications utilizing high salt and extreme pH conditions. Biocompatibility ensures the integrity of your biomolecules and robustness of the system.
Great results!!
peptides, nucleic acid and sugar
Agilent 1290 Infinity II Bio LC could be more effective in analysis of peptide and nucleic acid to reduce the analysis time and running costs. Recently, we use it by remote access from other office and home.
Review Date: 10 May 2022 | Agilent Technologies
Great!
Analytical chemistry
Product is very easy to use, which helps us a lot.
Review Date: 7 Apr 2021 | Agilent Technologies
Efficient detection and analysis.
Analytical chemistry, compounds detection and sepration
The instrument gives an excellent result and is efficient.
Review Date: 7 Apr 2021 | Agilent Technologies
Top choice, great results!
Pharmaceuticals
This system is the premium choice and yields excellent results.
Review Date: 7 Apr 2021 | Agilent Technologies
Greatly improves work efficiency.
Pharmaceuticals
Powerful software, easy to use, timely and thoughtful after-sales service.
Review Date: 7 Apr 2021 | Agilent Technologies
I'd buy it again.
Oligonucleotides
I haven't used a lot of the 1290 Bio yet, but I've confirmed that it gives the same results as the general 1290. I'll have to try using it more, but I think it performs better for robustness and convenience in bio applications so far. It can be connected with the existing ECM server, so there is no difficulty in using it.
Review Date: 7 Apr 2021 | Agilent Technologies
I can't work without this great partner now.
Pharma, Biopharma
Our lab has had a 1290 system for more than two years. It brings extra impressive robustness, and useability to us even for entry level scientist.
Review Date: 7 Apr 2021 | Agilent Technologies
Great performance, improves work efficiency.
Biopharma
Especially good for CQA in biopharma
Review Date: 7 Apr 2021 | Agilent Technologies
Great results.
Analytical chemistry
Great product.
Review Date: 7 Apr 2021 | Agilent Technologies
This is the best bioinert UHPLC system available.
Analytical Chemistryc
I have had the pleasure of running two different modes of separation with the newly launched Agilent 1290 Bio UHPLC system. This instrument has been long awaited for many years and it actually comes to industry a bit late but it was worth the wait. The ratings for ease of use, after sales care and value for money go hand in hand with the current Agilent platforms that I have been using and been accustomed to for over 16 years. This new line of Bioinert UHPLC will blow the other vendors away. It truly is the best bioinert system out on the market. For the smaller companies out there, this would be the instrument of choice!
Review Date: 24 Dec 2020 | Agilent Technologies
The 1290 Infinity II Bio LC System is a UHPLC, binary or quaternary ,- consisting of biocompatible material for use in biopharma (e.g., critical quality attributes) and other applications utilizing high salt and extreme pH conditions. Biocompatibility ensures the integrity of your biomolecules and robustness of the system.
Based on proven 1290 Infinity II LC technology, the system offers easy method transfer, also from legacy instrumentation, and saves time and effort spent on training. It provides highest resolution and lowest dispersion at pressures up to 1300 bar. A variety of bio flow cells are available for sensitive UV detection.
Features
- Biocompatible solvent and sample flow path ensure integrity of biomolecules and minimize unwanted surface interaction
- Different bio accessories include a range of bio heat exchangers, bio capillary kits, bio loops, and analytical heads to cover all the different application needs for instrument versatility and efficiency
- Flexibility for all applications - through wide power, temperature, an automatically scalable injection range, and gradient options (binary and quaternary solvent mixing)
- Lowest delay volume throughout the system enables highest chromatographic resolution and lowest dispersion
- High salt tolerance and wide pH range offer enhanced flexibility and robustness for increased instrument uptime
- Based on the proven 1290 Infinity II technology for easy method transfer, also from legacy instrumentation, and less training effort
- Shallow microplate drawers take a maximum load of 6,144 samples for unmatched sample capacity
- Power range combines ultrahigh pressure up to 1300 bar and high analytical flow rates up to 5 mL/min for maximum UHPLC performance
- A wide range of sensitive optical detection capabilities with various flow cells for VWD, DAD, FLD, Bio MDS, or LC/MS detection for highest adaptability
Separation of organic acids on an Agilent InfinityLab Poroshell 120 Aq-C18 column
In this application note, an InfinityLab Poroshell 120 Aq-C18 column is used for the analysis of organic acids under a highly aqueous mobile phase. Two mobile-phase additives were investigated, and the method was optimized separately. Organic acids are an important group of components in many food samples, including fruit drinks, wine, and other aqueous samples. Download this free application note to discover more, including the results of these analyses.
Automated switching between peptide and glycan mapping with the Agilent 1290 Infinity II LC System
In this application note, Agilent showcases the new 1290 Infinity II Bio LC as a novel platform for peptide mapping. Exploiting the high-precision, binary Agilent 1290 Infinity II Bio High-Speed Pump and a biocompatible, iron-free flow path, the system is especially suited to biomolecules like peptides, proteins, and metabolites.
Analysis of lipid nanoparticle composition
In this application note, Agilent presents liquid chromatographic method development for the analysis of the lipid components of patisaran (trade name Onpattro) in a quaternary setup.
Robust and reliable peptide mapping
Peptide mapping is the gold standard for elucidating the primary structure of monoclonal antibodies and the key to successful peptide mapping is a robust and reliable LC system for high-quality peptide separation. In this application note, Agilent Technologies presents the Agilent 1290 Infinity II Bio LC as the system of choice for peptide mapping.
In-depth peptide mapping with iterative MS/MS acquisition on the Agilent 6545XT AdvanceBio LC/Q TOF
Therapeutic monoclonal antibodies (mAbs) represent one of the fastest growing classes of protein-based drugs in the biopharmaceutical industry. Here, Agilent Technologies presents an integrated workflow for peptide mapping of a mAb, from automated sample preparation to data analysis, featuring an Agilent AssayMAP Bravo liquid-handling robot, the Agilent 1290 Infinity II LC system, the Agilent 6545XT AdvanceBio Q-TOF, and the Agilent MassHunter BioConfirm B.09 software.
N-glycan analysis: Better together
In this brochure, Agilent Technologies introduces the Agilent and ProZyme sample preparation workflows that are designed to help push your glycoprotein analysis to new levels of performance.
Refining charge variant analysis of mAbs with the Agilent 1290 Infinity II Bio LC System
Charge variant analysis is a demanding application for applied liquid chromatography systems due to the use of highly corrosive buffer salts in combination with very shallow gradients for optimal separation. This application note demonstrates the analysis of charge variants for trastuzumab and the NISTmAb reference standard.
Elevate your mAb aggregate analysis with the Agilent 1290 Infinity II Bio LC System
Modern columns for size exclusion chromatography (SEC) separation of proteins comprise material with sub-2 μm particles for optimum resolution. This application note demonstrates the superior resolution in SEC protein separation made possible by the Agilent 1290 Infinity II Bio LC System.
Seamless method transfer to the Agilent 1290 Infinity II Bio LC System
Peptide mapping requires reliable and robust methods with high precision for analyzing the primary structure and post-translational modifications (PTMs) of biopharmaceuticals. This application note shows that method transfer can be easy and convenient with the new Agilent 1290 Infinity II Bio LC System.
From Start to Finish: How Agilent Can Support Better Decisions in IVT mRNA-Based Biopharmaceutical Analysis
Jan 21st 2025 - 16:00 GMT / 17:00 CET / 11:00 EST / 8:00 PST
Development and production of IVT mRNA vaccines require measurements of Critical Quality Attributes (CQAs), such as identity, purity, quality, and quantity. Orthogonal methods developed for such purposes are outlined in the USP guidelines for mRNA vaccines.
Agilent Technologies provides many analytical solutions that are used by leading pharmaceutical companies in early-stage drug development through production quality control (QC) and final product release. These systems help users save time and money with optimized workflows, streamlined processes, and fast time-to-results.
Whitney Pike, M.Sc, Application Scientist and Dr. Sonja Schneider, Manager, Application Development, Agilent Technologies, will discuss how automated electrophoresis and LC/MS systems solutions from Agilent provide reliable insights for IVT mRNA workflows at many steps, from QC of the initial plasmid DNA, throughout the IVT process, to the finished mRNA drug product.
Key learning objectives
- Explore QC throughout the entire IVT mRNA workflow, including data from various nucleic acids and LNP analyses
- Discover how Agilent instruments/technologies, such as the Fragment Analyzer and LC/MS systems, can be used for different CQAs
- Learn how Agilent is working together with its partners to provide reliable insights for all aspects of the IVT mRNA workflow
Who should attend?
- Researchers, lab managers, lab directors, QA/QC managers performing IVT mRNA analysis in biotherapeutics research and production
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
For Research Use Only. Not for use in diagnostic procedures. PR7001-2498
The benefits of dedicated HPLC instrumentation and columns for analysis of biomolecules
In this webinar, Andrew Coffey and Cornelia Vad, Agilent Technologies, demonstrate how biologics differ from small molecules, and how the InfinityLab Bio LC Solutions from Agilent can help you make an assured start in biopharma.
Enhance your chromatography knowledge with these top trending resources
Discover the latest technologies, application eBooks, and expert insights that can help your laboratory optimize its separations
HPLC: 5 top resources to maximize accurate data generation
From video interviews with top scientists to our interactive application guide, explore this collection of resources to help hone your technique for optimum results
Interactive Biopharmaceutical HPLC Analysis Content Hub launched
Advance your biopharma research through HPLC with this first-of-its-kind application-focused resource
Meet the winners of the 2021 Scientists’ Choice Awards for Drug Discovery & Development
Scientific, technological and communications excellence has been celebrated at the Drug Discovery & Development Webinar Series, with Waters, BioTek and PHCbi among those recognized
7 lab products advancing drug discovery
Take a look at what other researchers and scientists are saying about cutting-edge technologies in drug discovery