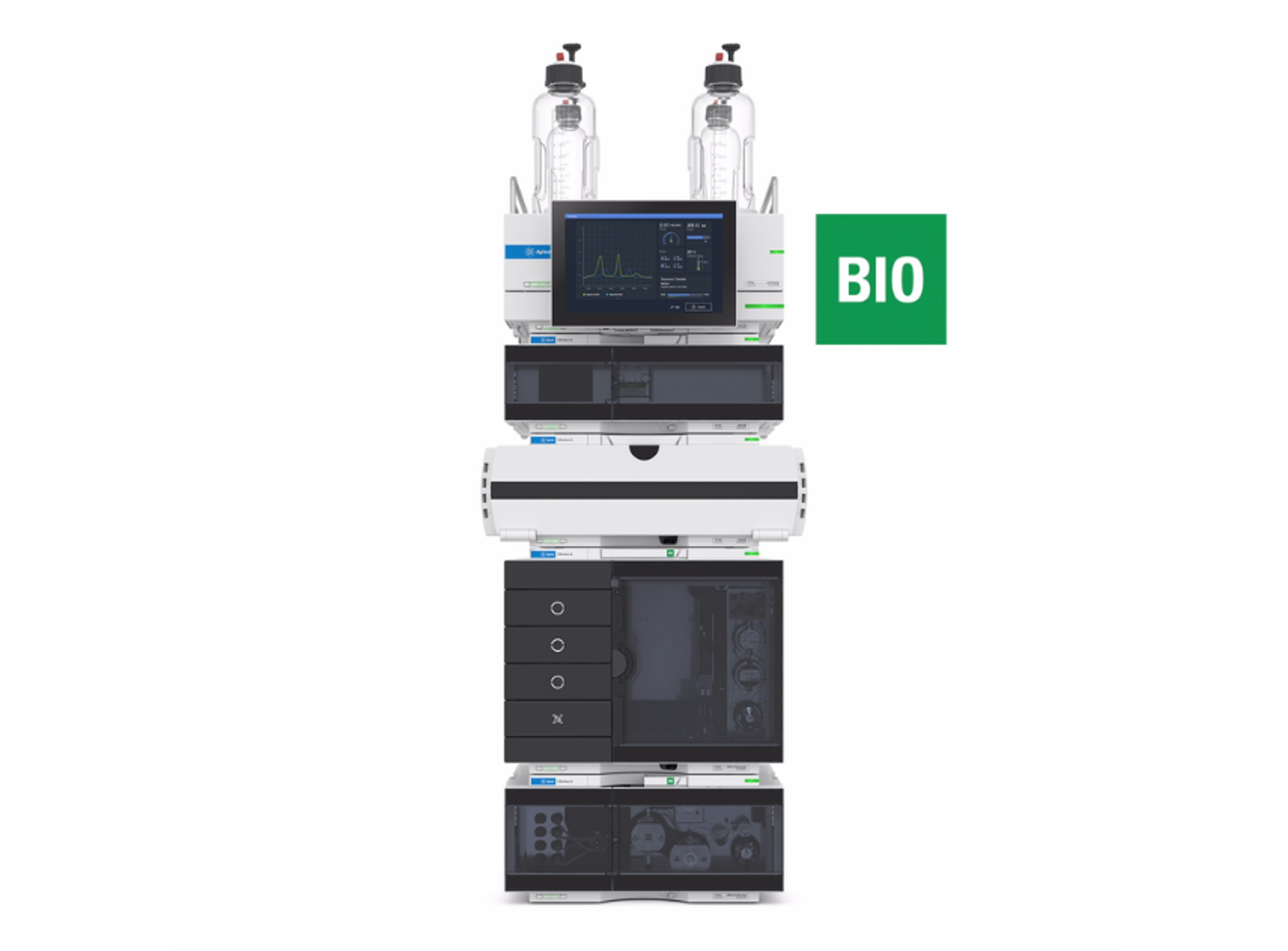
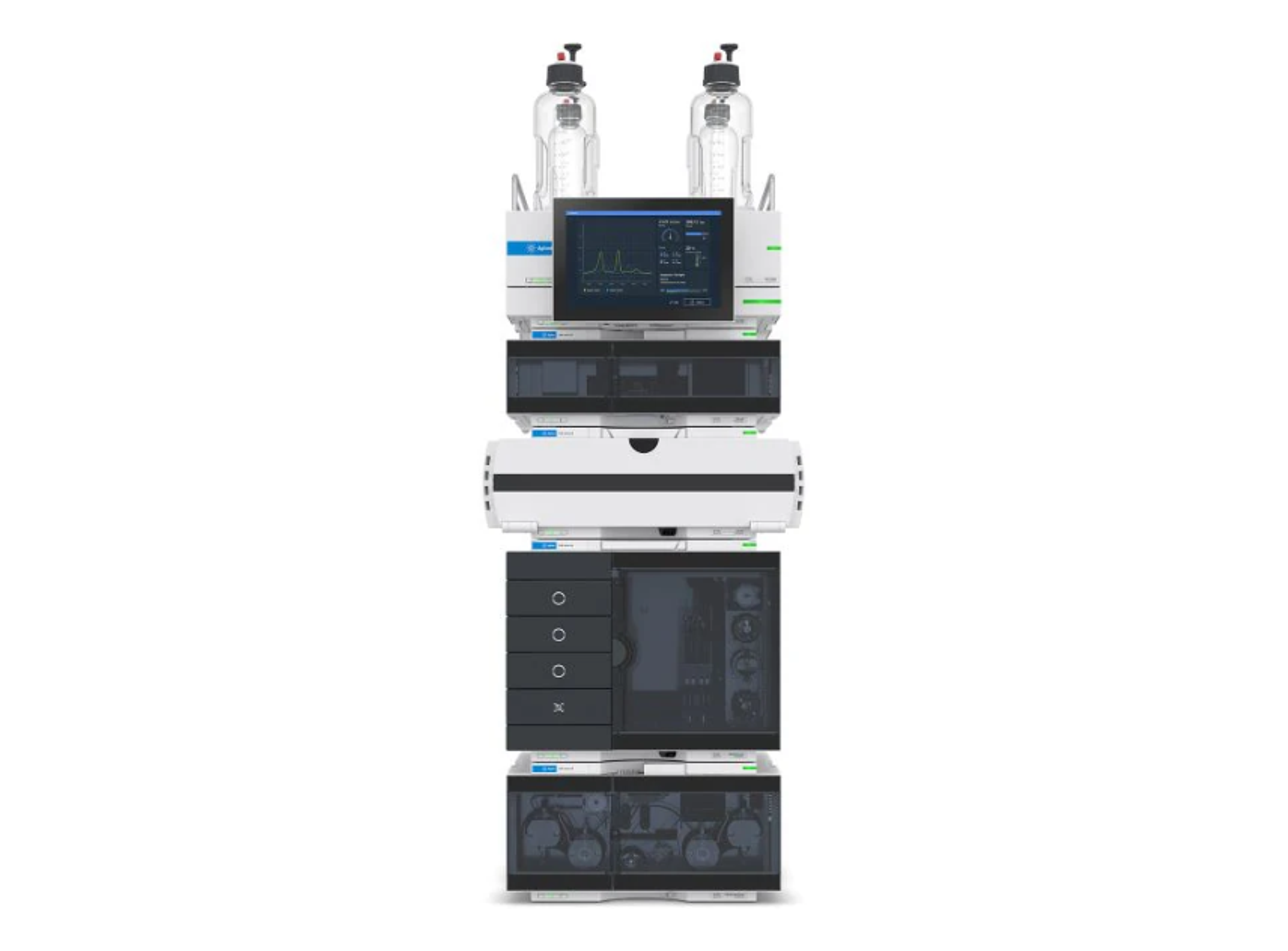
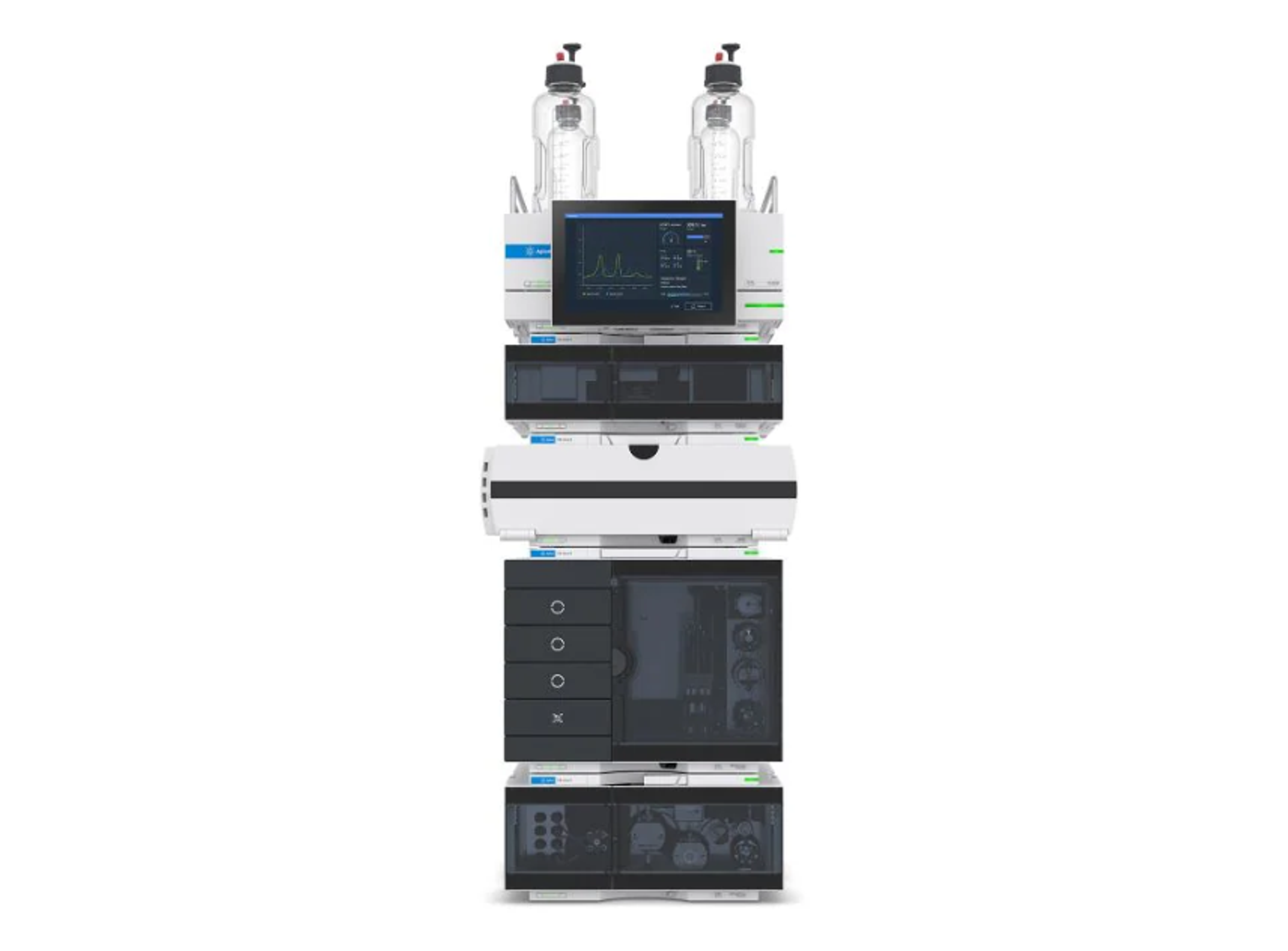
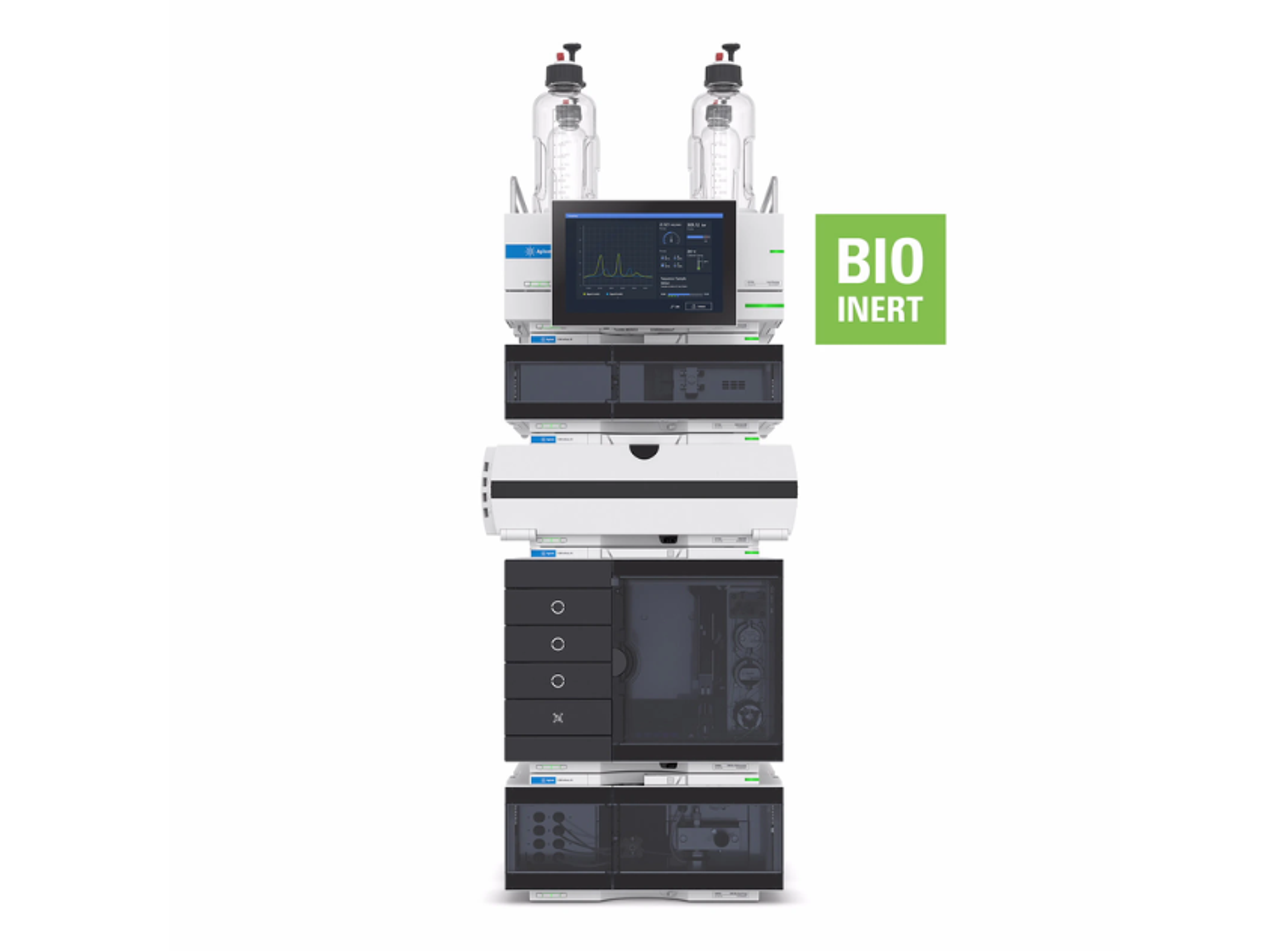
Agilent 1260 Infinity III LC System
The 1260 Infinity III LC System is a robust analytical high-performance liquid chromatography (HPLC) system that offers the widest choice of modules for analytical HPLC and entry-level UHPLC analysis. This HPLC instrument delivers the performance, reliability, and robustness you need for highest confidence in your daily HPLC results.
Building on the history of the Agilent 1100, 1200, and 1260 Infinity II series analytical HPLC systems, the 1260 Infinity III LC System delivers robust HPLC results while providing several ease-of-use features, making it the standard system for a reliable routine HPLC analysis. Mix and match new modules with existing HPLC instrumentation to maximize uptime and minimize disruption. Agilent Instrument Control Framework (ICF) enables smooth control of Agilent LC instrumentation through third-party chromatography data systems. The 1260 Infinity III LC System puts you on the fast track to efficiency, optimizing speed and resolution for analysis by HPLC.
Brochures
Elevate your Infinity II LC to Infinity III LC
The Agilent Infinity II LC Series has been a trusted solution for over a decade, consistently delivering reliable analytical results. Building on this proven foundation, the Agilent 1260/1290 Infinity III LC Series introduces enhanced automation, system health indicators, and guided assistance to simplify routine operations and significantly reduce operator training and troubleshooting time. Designed with full backward compatibility, the Infinity III LC Series enables seamless upgrades without compromising existing methods or results from Infinity II LC systems.
Download this resource to explore what’s new in the Agilent Infinity III LC Series and learn how you can future-proof your Infinity II LC investment with minimal disruption.
Eliminate errors with automation and stress-free sample tracking
In a typical laboratory, quality control mishaps are inevitable during sample preparation. For example, a sample going missing or a sample being miss-identified will result in downstream effects including the loss of significant time, money and samples. Therefore, advances in digitalization have been shown to reduce human error as manual input and transcription is eliminated from the process. Consequently, this lowers the likelihood of obstacles such as these, to improve data accuracy and process efficiency.
Join Dr. Kelsey Mato, Product Marketing at Agilent Technologies, to hear about a new technology, linking samples and software for unprecedented control, to give you confidence in your laboratory workflow.
Key learning objectives:
- Understand the long-term risks and costs of manual sample tracking
- Learn how taking steps toward digitalization can reduce errors in sample management
- Find a new solution for seamlessly linking samples and software
Who should attend?
Lab managers, and experienced and inexperienced lab technicians.
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.