4200 TapeStation Instrument
The Agilent 4200 TapeStation system is an established automated electrophoresis tool for DNA and RNA sample quality control. Fully automated sample processing enables the unattended analysis of size, concentration and integrity. Choose any sample number between 1 and 96 and analyze at constant cost per sample.
Very easy to use
Life sciences
Fast and reliable quality control of DNA and RNA samples.
Review Date: 16 Jul 2024 | Agilent Technologies
Best machine for quick checking of samples!
Analysis of bad quality DNA samples
The Agilent Tapestation has truly changed my way of doing research. I use this machine almost weekly, to check my samples either before or after analysis. It is very easy and quick to use.
Review Date: 18 Nov 2023 | Agilent Technologies
Easy to use
Library size range
Instrument is easy to use and quick to get results. It takes a few practices to use Tapestation.
Review Date: 12 May 2023 | Agilent Technologies
Great product, who works on a tight schedule and have shortage of time
Molecular Biology
Ease of use, value of money, cool design, and most important effective results in a very short time. Minimum hands-on for reagent prep. All it taken few minutes and within a 30 min. you can see the results of your 4 samples.
Review Date: 2 Dec 2022 | Agilent Technologies
Excellent and reliable results!
Analyze sample quality
The instrument is very easy to use and provides sufficient data to proceed with our experiments. The instrument demonstrates high quality and accuracy.
Review Date: 10 Dec 2021 | Agilent Technologies
A "Must-have!"
Analysis of DNA fragment size and quantity
Always a dependable instrument for many on the team. Definitely deserves a spot in our lab!
Review Date: 17 Nov 2021 | Agilent Technologies
Great value
Life science
Quick and easy
Review Date: 17 Nov 2021 | Agilent Technologies
Easy to use with shorter run time compared to traditional agarose gel method
Quality control testings DNA and RNA
The instrument is very easy to use and it is compatible with both DNA and RNA tapes. We always use it to check for quality of DNA and RNA before proceeding to downstream assays. Saves time and consistent results
Review Date: 17 Nov 2021 | Agilent Technologies
Saves time.
4C-seq library size
This machine saves time (time is money). I used to run a gel for our lib size. I started using this machine, I can do so much more in less time.
Review Date: 17 Nov 2021 | Agilent Technologies
This is a fantastic instrument to add to our NGS suite.
DNA analysis
The TapeStation is a great upgrade to the Bioanalyzer. It is much easier to use, with relatively little chance for user error. The resolution isn't quite as fine as the Bioanalyzer, but the sensitivity, speed and high-throughput ability of the TapeStation make for a great improvement to our lab.
Review Date: 27 Sept 2021 | Agilent Technologies
The Agilent 4200 TapeStation system is an established automated electrophoresis tool for DNA and RNA sample quality control. Fully automated sample processing enables the unattended analysis of size, concentration and integrity. Choose any sample number between 1 and 96 and analyze at constant cost per sample.
The TapeStation system provides a complete solution for true end-to-end sample quality control within any next-generation sequencing (NGS) or Biobank workflow. This covers the full DNA and RNA range and offers high levels of flexibility in a simplified workflow with ready-to-use ScreenTapes.
Features:
- Simplify your workflow with fully automated sample processing and ready-to-use ScreenTape technology.
- Any sample number between 1 and 96 can be analyzed, with constant costs per sample providing scalable throughput.
- Easily switch between DNA and RNA ScreenTape assays for excellent flexibility.
- Results are obtained quickly
- in as little as 1 to 2 minutes per sample, or less than 90 minutes for 96 samples.
- Achieve user-independent results with minimal manual intervention and excellent reproducibility for sizing, concentration and integrity assessment.
- Requires as little as 1 to 2 µL of DNA or RNA samples, even for high sensitivity analysis.
- Rely on the integrity standards for RNA (RNA integrity number equivalent, RINe) and genomic DNA (DNA Integrity Number, DIN).
- Carryover is eliminated as the ScreenTape device analyzes each sample in a separate lane with individual loading tips for each.
Brochures
Upgrade your sample quality control workflow
In this flyer, discover Agilent Technologies' portfolio of automated electrophoresis solutions that enable laboratories to easily and effectively analyze nucleic acids for a wide range of applications.
Trade in your bioanalyzer instrument
In this flyer, find out how you can trade in your Bioanalyzer system with Agilent Technologies for a limited time and receive credit toward an Agilent TapeStation, Fragment Analyzer, or ProteoAnalyzer system.
Tumor genomic profiling with Agilent SureSelect cancer assays
Agilent SureSelect cancer assays are a portfolio of targeted resequencing assays based on next-generation sequencing technology (NGS), which can be utilized in comprehensive genomic profiling (CGP) to advance precision oncology. SureSelect cancer assays enable you to detect key classes of somatic variants and assess immuno-oncology biomarkers and homologous recombination deficiency. Explore the benefits such as error-correcting molecular barcodes, convenient enzymatic fragmentation, and minimum sample input; plus, discover how the workflow solution can be configured to meet the specific needs of your laboratory.
Agilent automated electrophoresis portfolio brochure
Whether performing low- or ultra-high-throughput testing, Agilent Technologies' automated electrophoresis instrument portfolio delivers reliable, objective assessments of sample integrity, concentration, and fragment size. Explore the complete portfolio of automated electrophoresis instruments, application-specific assays, intuitive software, and expert technical services that provide flexible solutions for robust and objective sample quality assessment.
Use of the Agilent 4200 TapeStation System for sample quality control in the whole-genome sequencing workflow
Explore how the Genomics and Proteomics Core Facility of the German Cancer Research Center has successfully integrated the Agilent Genomic DNA and Agilent D1000 ScreenTape assays using the Agilent 4200 TapeStation system as a QC tool into its whole exome sequencing workflow.
Discover how using the 4200 TapeStation system has enabled the Facility to optimize the sequencing library preparation process from the beginning to the end with a final QC of finished libraries to ensure valuable sequencing results.
Quality control of NGS libraries with daisy chains
The Agilent automated electrophoresis solutions offer fast and reliable quality control of next-generation sequencing (NGS) libraries. The Bioanalyzer, TapeStation, and Fragment Analyzer systems with dedicated high sensitivity assays allow unambiguous detection of daisy chains and accurate sizing of the target library. In this application note, Agilent Technologies provides a recommendation for which assay to employ to reliably visualize daisy chain products in NGS libraries. Furthermore, it emphasizes the consistency between the instruments and reproducibility of analysis confirmed by results on a set of two-fold serial dilutions.
Sample measurement and analysis best practices for Agilent TapeStation Systems
In this application note, Agilent Technologies outlines best practices for achieving reliable quantification and sizing results using the assays available for the 4150 and 4200 TapeStation systems.
FFPE RNA quality assessment with the Agilent Bioanalyzer and TapeStation Systems
In this application note, Agilent Technologies highlights the capabilities of both the Agilent Bioanalyzer and TapeStation systems in analyzing FFPE RNA samples using the DV200 score. The results provide a comparison between the DV200 scores from each system.
Quantitative analysis with the Agilent TapeStation Systems
In this application note, Agilent Technologies systematically evaluates the quantitative performance of the TapeStation systems and compares it to the Bioanalyzer system, NanoDrop, and Qubit.
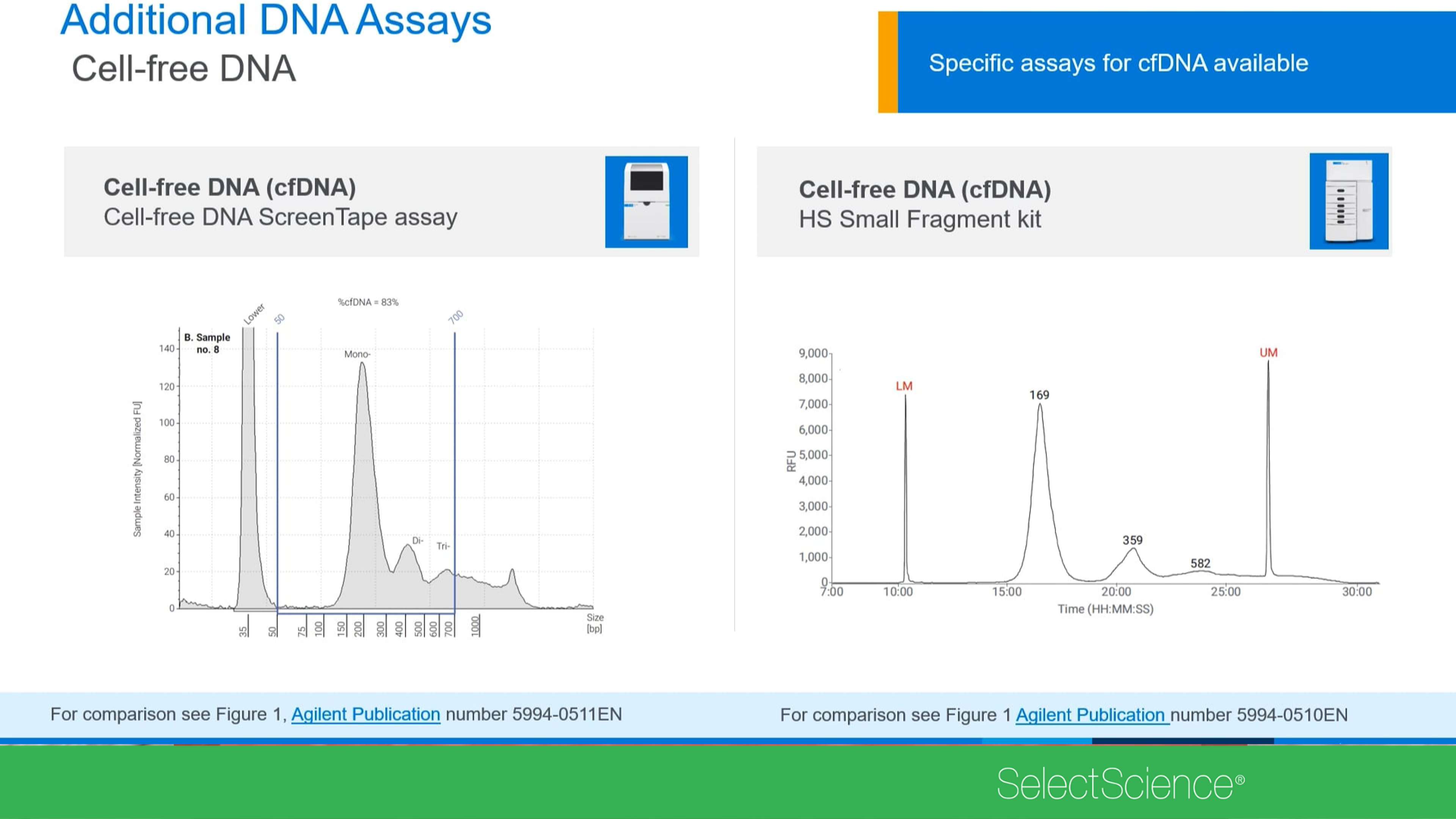
Performance characteristics of the cell-free DNA ScreenTape assay
In this application note, Agilent Technologies discusses the performance of the cell-free DNA ScreenTape assay by evaluating sensitivity, accuracy, and precision of quantification and sizing while exploring the performance of the new quality metric %cfDNA. The performance of the assay was compared on the 4150 and the 4200 TapeStation systems to demonstrate a seamless transition between the two instruments.
Quality control of cell-free DNA samples analyzed with next-generation sequencing
Analysis of cfDNA can introduce challenges due to its low concentration and possible contamination from high-molecular-weight (HMW) DNA, both necessitating reliable quality control. In this application note, Agilent Technologies discusses how to assess the quality of cell-free DNA samples analyzed with next-generation sequencing (NGS). The Agilent TapeStation system is a vital quality control tool in NGS workflows. The TapeStation systems and cell-free DNA ScreenTape assay provide a %cfDNA quality metric for determining the quality of input cfDNA for downstream processes.
Quality control of genomic DNA for The French Kidney Disease Study by the Biobanque de Picardie
The French Chronic Kidney Disease – Renal Epidemiology and Information Network (CKD-REIN) project is a prospective cohort study that was established to identify the determinants, biomarkers, and practice patterns associated with chronic kidney disease outcomes. Whole blood samples of the cohort study were processed into buffy coats followed by genomic DNA (gDNA) extraction and quality control (QC) by the Biobanque de Picardie.
In this application note, Agilent Technologies demonstrates the use of the Agilent 4200 TapeStation system and the Genomic DNA ScreenTape assay as an optimal QC tool to monitor the integrity of gDNA extracted from buffy coats stored frozen for up to five years. The note also describes how the objective quality metric of the TapeStation system, the DNA integrity number (DIN), was applied to evaluate the impact of a delay in blood processing on gDNA quality. The QC steps with the DIN quality metric allowed the Biobanque de Picardie to confirm the stability of the biospecimen processing workflow and verify that all the CKD-REIN samples were high quality and suitable for genome-wide association studies.
Agilent Automated Electrophoresis compendium
Automated electrophoresis is a powerful tool for microbial, fungal, and viral nucleic acid analysis. The Agilent Fragment Analyzer, Agilent TapeStation, and Agilent Bioanalyzer systems provide a fast and effective method for sample quality control as part of the workflow for genomic analysis applications such as next-generation sequencing (NGS) or new method development. Download this Agilent compendium to learn the numerous ways that Agilent electrophoresis systems are utilized for the analysis of infectious diseases
From Start to Finish: How Agilent Can Support Better Decisions in IVT mRNA-Based Biopharmaceutical Analysis
Jan 21st 2025 - 16:00 GMT / 17:00 CET / 11:00 EST / 8:00 PST
Development and production of IVT mRNA vaccines require measurements of Critical Quality Attributes (CQAs), such as identity, purity, quality, and quantity. Orthogonal methods developed for such purposes are outlined in the USP guidelines for mRNA vaccines.
Agilent Technologies provides many analytical solutions that are used by leading pharmaceutical companies in early-stage drug development through production quality control (QC) and final product release. These systems help users save time and money with optimized workflows, streamlined processes, and fast time-to-results.
Whitney Pike, M.Sc, Application Scientist and Dr. Sonja Schneider, Manager, Application Development, Agilent Technologies, will discuss how automated electrophoresis and LC/MS systems solutions from Agilent provide reliable insights for IVT mRNA workflows at many steps, from QC of the initial plasmid DNA, throughout the IVT process, to the finished mRNA drug product.
Key learning objectives
- Explore QC throughout the entire IVT mRNA workflow, including data from various nucleic acids and LNP analyses
- Discover how Agilent instruments/technologies, such as the Fragment Analyzer and LC/MS systems, can be used for different CQAs
- Learn how Agilent is working together with its partners to provide reliable insights for all aspects of the IVT mRNA workflow
Who should attend?
- Researchers, lab managers, lab directors, QA/QC managers performing IVT mRNA analysis in biotherapeutics research and production
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
For Research Use Only. Not for use in diagnostic procedures. PR7001-2498
Is Sample Integrity Assessment for Long-Read Sequencing Technologies Essential?
Exploring Key Topics in Sample Quality Control for NGS Workflows
Next-generation sequencing (NGS) technologies enable deeper insights into different areas of research. In this latest webinar series, researchers from the European Molecular Biology Laboratory (EMBL), Quantabio LLC,, and Agilent Technologies discuss the importance of input sample quality control in whole genome sequencing (WGS), panel-based targeted sequencing and long-read sequencing workflows in order to maximize sequencing output.
Sample Integrity Assessment for Long-read Sequencing Technologies, Is It Essential?
Next generation sequencing (NGS) has evolved rapidly in the past decade creating opportunities for new technologies to emerge, ultimately making this technology more robust. Among these, particularly long-read sequencing has experienced an increase of popularity in the research community due to improvements in the overall stability and the quality of generated data.
However, the value of long-read sequencing data is critically dependent on the quality of the input material used for a library preparation. Therefore, reliable and accurate quality control (QC) is essential for the success of these sequencing technologies. At EMBL, the Agilent Femto Pulse system is a fundamental part of the daily routines for processing LRS samples. It allows researchers to verify the integrity and fragment size distribution of nucleic acid samples.
This webinar will highlight the importance of Femto Pulse in GeneCore workflows and present data that emphasize the significance of sample quality control for success of EMBL’s undertakings.
Key learning objectives:
- How stringent QC of incoming samples helps identify and prevent potential problems that could negatively impact their performance in library preparation and sequencing
- How ensuring the accurate size and quantity of the finished library is essential for maximizing sequencing output.
- How the Agilent Femto Pulse system is used in GeneCore workflows.
Who should attend:
- NGS and genomics researchers
- Molecular biologists
- Cancer researchers
- Sequencing lab managers and technicians
Certificate of attendance
- All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
Streamlined DNA Library Preparation Solutions and Guidelines for Robust Quality Control of PCR-free WGS Libraries
Exploring Key Topics in Sample Quality Control for NGS Workflows
Next-generation sequencing (NGS) technologies enable deeper insights into different areas of research. In this latest webinar series, researchers from the European Molecular Biology Laboratory (EMBL), Quantabio LLC,, and Agilent Technologies discuss the importance of input sample quality control in whole genome sequencing (WGS), panel-based targeted sequencing and long-read sequencing workflows in order to maximize sequencing output.
Streamlined DNA Library Preparation Solutions and Guidelines for Robust Quality Control of PCR-free WGS Libraries
Whole genome sequencing (WGS) and panel-based targeted sequencing provide vital genetic information for various applications. The quality of sequencing data hinges on the input library, traditionally prepared using PCR amplification. PCR-free WGS library preparation has become a critical alternative, eliminating amplification-induced issues and preserving the genome's true representation. This is especially important for accurate mutational analysis and disease diagnostics, enhancing the detection of structural variants and GC-rich regions. Quality control (QC) of PCR-free libraries presents a challenge when using electrophoresis. This typically results in inaccurate library size estimations due to the structural features of Y-shaped adapters commonly used during library preparation. In this webinar, we describe a method to accurately determine the size of PCR-free libraries by adding a short PCR amplification step prior to automated DNA fragment analysis.
Key learning objectives:
- Understand why PCR-free WGS library preparation is essential for improving the reliability and efficiency of genetic analyses across various fields
- Discover how sparQ DNA Frag and DNA Library Prep Kits can be utilized to generate PCR-free libraries for DNA library preparation
- Learn how to modify PCR-free libraries for proper QC utilizing the Agilent TapeStation system
Who should attend:
- NGS and genomics researchers
- Molecular biologists
- Cancer researchers
- Sequencing lab managers and technicians
Certificate of attendance
- All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
Quality matters: Tackling high molecular weight DNA contamination in cell-free DNA for optimal liquid biopsy NGS
Tuesday, June 24, 2025 at 16:00 BST / 17:00 CEST / 11:00 EDT / 08:00 PDT
Cell-free DNA (cfDNA) has become an essential tool in clinical research and serves as valuable biomarker in liquid biopsy applications. However, cfDNA presents a challenge for Next-Generation Sequencing (NGS) workflows due to its low yield, complex fragmentation pattern and potential presence of contaminating high molecular weight (HMW) DNA.
In this webinar, our expert speaker will discuss reliable pre-analytical quality control using the Agilent Cell-free DNA ScreenTape assay and the TapeStation systems, emphasizing their relevance to NGS-based liquid biopsy applications.
Key learning objectives
- Understand the challenges associated with cfDNA extraction and HMW DNA contamination.
- Learn about the impact of HMW DNA on cfDNA quality and NGS results.
- Explore how to ensure pre-analytical quality control of cfDNA samples.
- Discover the unique benefits of the Agilent Cell-free DNA ScreenTape assay for obtaining the full picture of your cfDNA quality.
Who should attend?
NGS and genomics researchers, molecular biologists, researchers in cancer diagnostics and liquid biopsy, and sequencing lab managers and technicians
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.
For Research Use Only. Not for use in diagnostic procedures. PR7001-4363
How automation and integrated sample QC enhance a targeted long-read-sequencing workflow
Tuesday, November 4, at 16:00 GMT | 17:00 CET | 11:00 EST | 8:00 PST
As the demand for greater accuracy and efficiency in genetic analysis grows, new approaches are needed to overcome the blind spots of conventional sequencing methods. This webinar will showcase a fully automatable workflow that integrates Agilent SureSelect hybrid capture, Agilent automated electrophoresis systems for sample quality control with PacBio HiFi long-read sequencing.
In addition to discussing workflow, we’ll share case studies where long reads play a fundamental role in identifying genetic variants that may be missed by conventional sequencing in research studies.
Join us to learn how targeted long-read sequencing:
- Focuses on genes and regions of interest in genetic research, while reducing costs and boosting sequencing depth at key loci.
- Overcomes the limitations of short-read technologies, including challenges with pseudogenes, homologous loci, structural variants, complex transcript isoforms, splicing events, and difficult regions such as repetitive or GC-rich sequences.
- Enhances both analytical performance and laboratory efficiency through automation.
Key learning objectives:
- Gain practical insights into implementing an automated targeted long-read sequencing workflow in the laboratory.
- Identify the genetic mutation types best suited for targeted long-read sequencing approaches.
- Understand the analytical advantages of long-read sequencing compared with conventional short-read methods.
Who should attend?
Lab directors, molecular pathologists, research scientists, clinical lab technicians, and anyone running or interested in running NGS workflows.
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.
For Research Use Only. Not for use in diagnostic procedures. PR7003-294
Equivalent performance: Bioanalyzer and TapeStation systems
In this video, Agilent Technologies compares the Agilent Bioanalyzer and the Agilent TapeStation systems, analyzing genomic DNA and RNA.
Ensuring quality control in next-generation sequencing
Explore the various systems from Agilent Technologies including the Bioanalyzer, TapeStation, and Fragment Analyzer, that can address challenges in next-generation sequencing (NGS) quality control. Gain top tips on how you can improve your workflow and transform your NGS applications with reliable quality metrics.
Exploring cell-free DNA and small RNA analysis techniques
Discover how you can sensitively determine sample quality using the Bioanalyzer, TapeStation and Fragment Analyzer from Agilent Technologies. These automated electrophoresis systems can be used for cell-free DNA (cfDNA) and small RNA quality control and analysis. Delivering highly accurate and precise analytical evaluation, learn how these systems fit perfectly into next-generation sequencing (NGS), biobank or vaccine development workflows for low to high sample throughput.
Sample QC control in multiple workflows
Discover Agilent’s automated electrophoresis solutions designed to help ensure experimental success through accurate sample quality assessment. With a focus on NGS and protein QC, these solutions offer flexible throughput and high-resolution performance to support and streamline your workflow.
Ensure Sample Quality Control with Agilent's TapeStation Systems
Dr. Rainer Nitsche, product manager at Agilent Technologies, gives an overview of the benefits of Agilent’s TapeStation platform for end-to-end QC within next-generation sequencing (NGS) workflows. With ready-to-use ScreenTape consumables, discrete ScreenTape lanes and fast results, the TapeStation automated electrophoresis system delivers high-quality DNA and RNA sample QC.
Automated Metabolomics Workflows to be Showcased by Agilent at SLAS
Agilent set to display a brand-new metabolomics-specific sample preparation solution