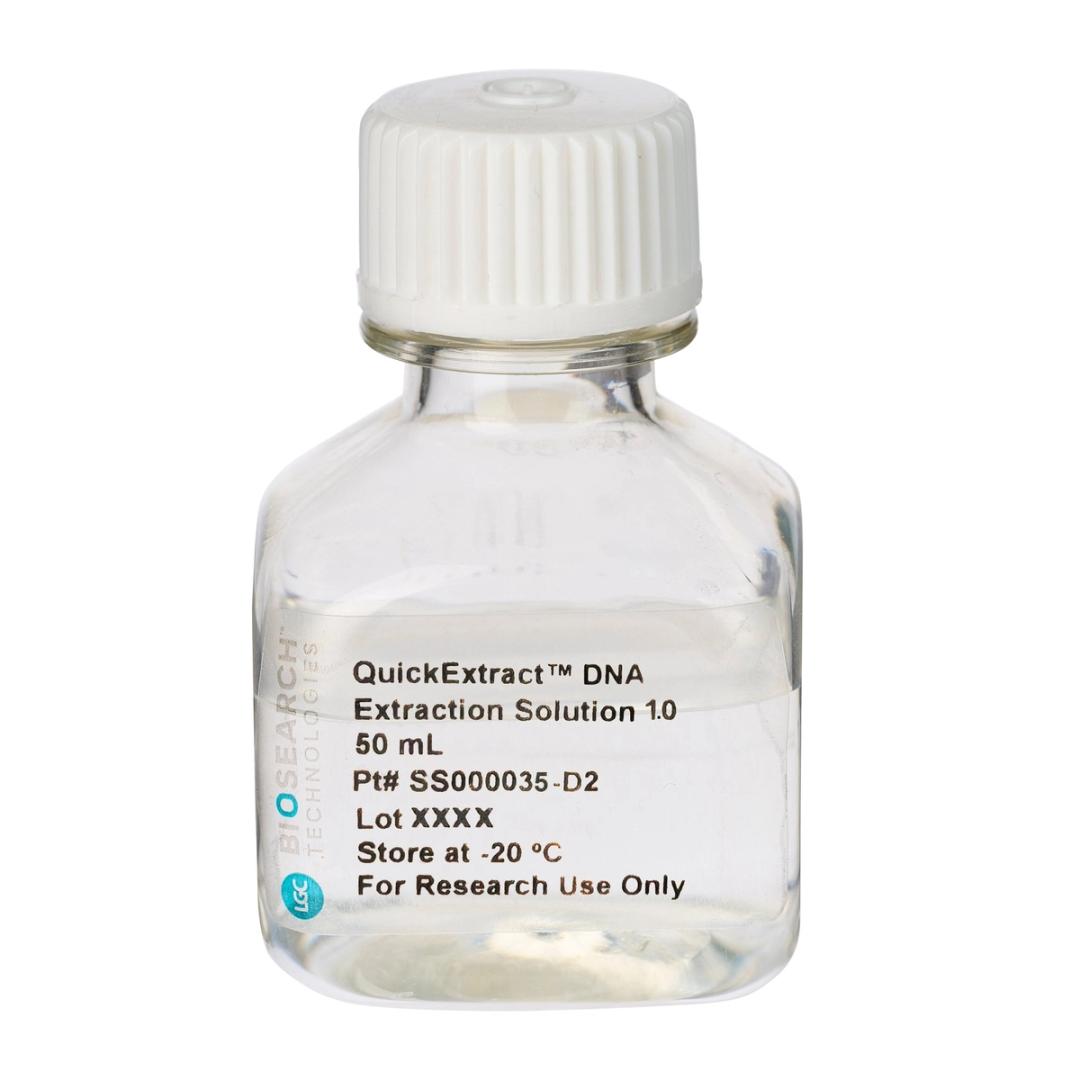
QuickExtract™ DNA Extraction Solution
The QuickExtract™ DNA Extraction Solution can be used to rapidly and efficiently extract PCR-ready genomic DNA from almost any sample type using a simple, one-tube protocol that takes only 3-8 minutes, depending on the sample. The extracted DNA is suitable for PCR analyses, such as genomic, transgenic, or viral DNA screening in animals, or for genetic or environmental research and screening in humans and other organisms.
Simple, rapid extraction of PCR-ready DNA for screening and genotyping type applications
- Fast: 8 minute extraction protocol for most sample types
- Simple: No centrifugation steps of spin columns used to help increase yields
- Automation-friendly: Simple protocol integrated easily into automated workflows
- Safe: Uses only non-toxic reagents
Simplifying the journey from molecular diagnostic assay development to commercialization
Molecular diagnostics (MDx) is rapidly advancing, with a growing array of DNA- and RNA-based tests now available to physicians, pharmacists, geneticists, research scientists, and other healthcare professionals.
As labs work with human and microbial genes, test developers must comply with clinical guidelines and regulations. Both established companies and start-ups face challenges, including meeting regulatory standards, ensuring quality, and managing supply chain issues. Early planning and robust partnerships are key to overcoming these obstacles.
This essential guide explores these challenges through detailed case studies. It covers everything from early development and assay formulation, to supply chain security, offering expert insights on successfully bringing PCR, next-generation sequencing (NGS), and other nucleic acid-based diagnostic tests to market.
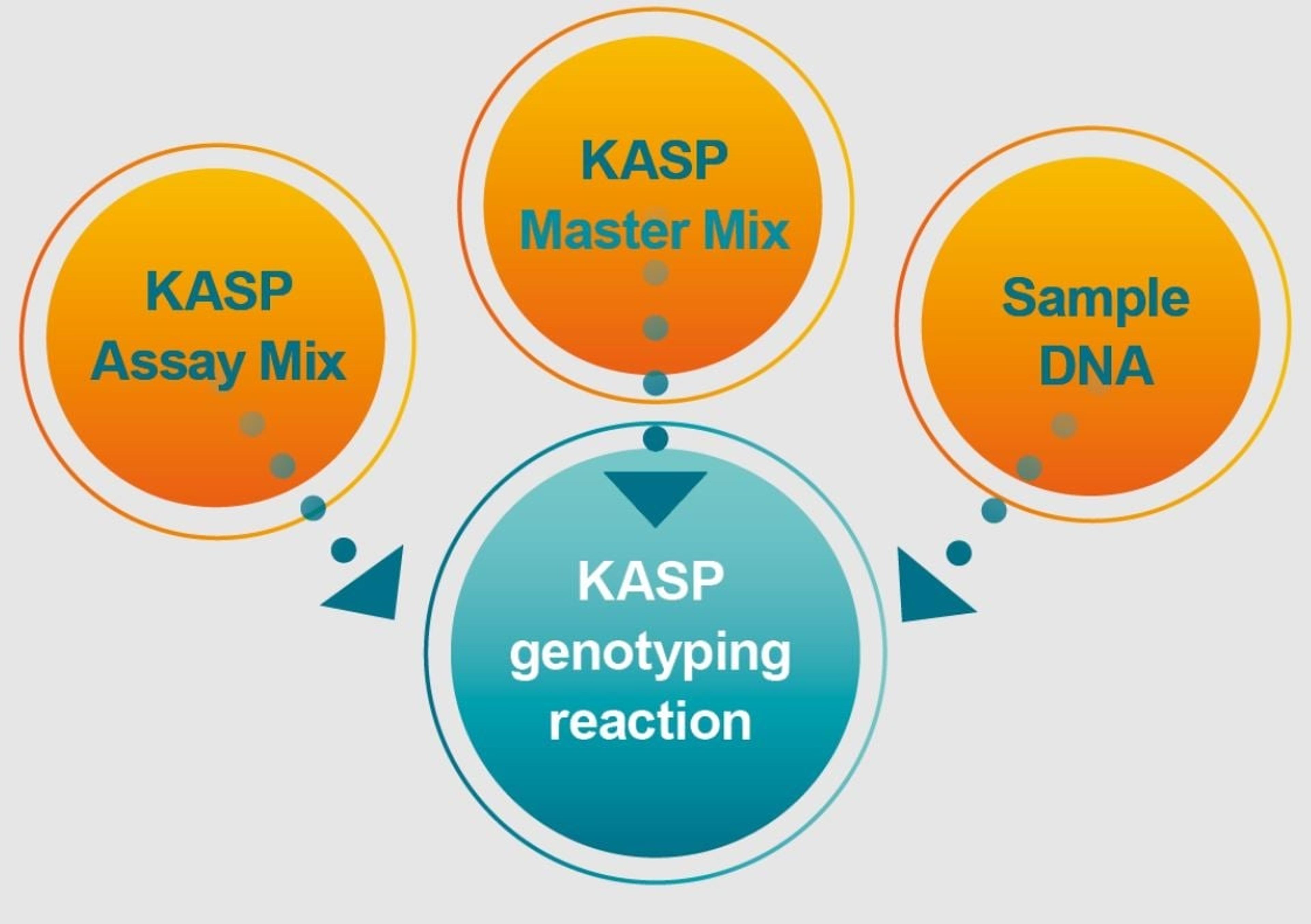
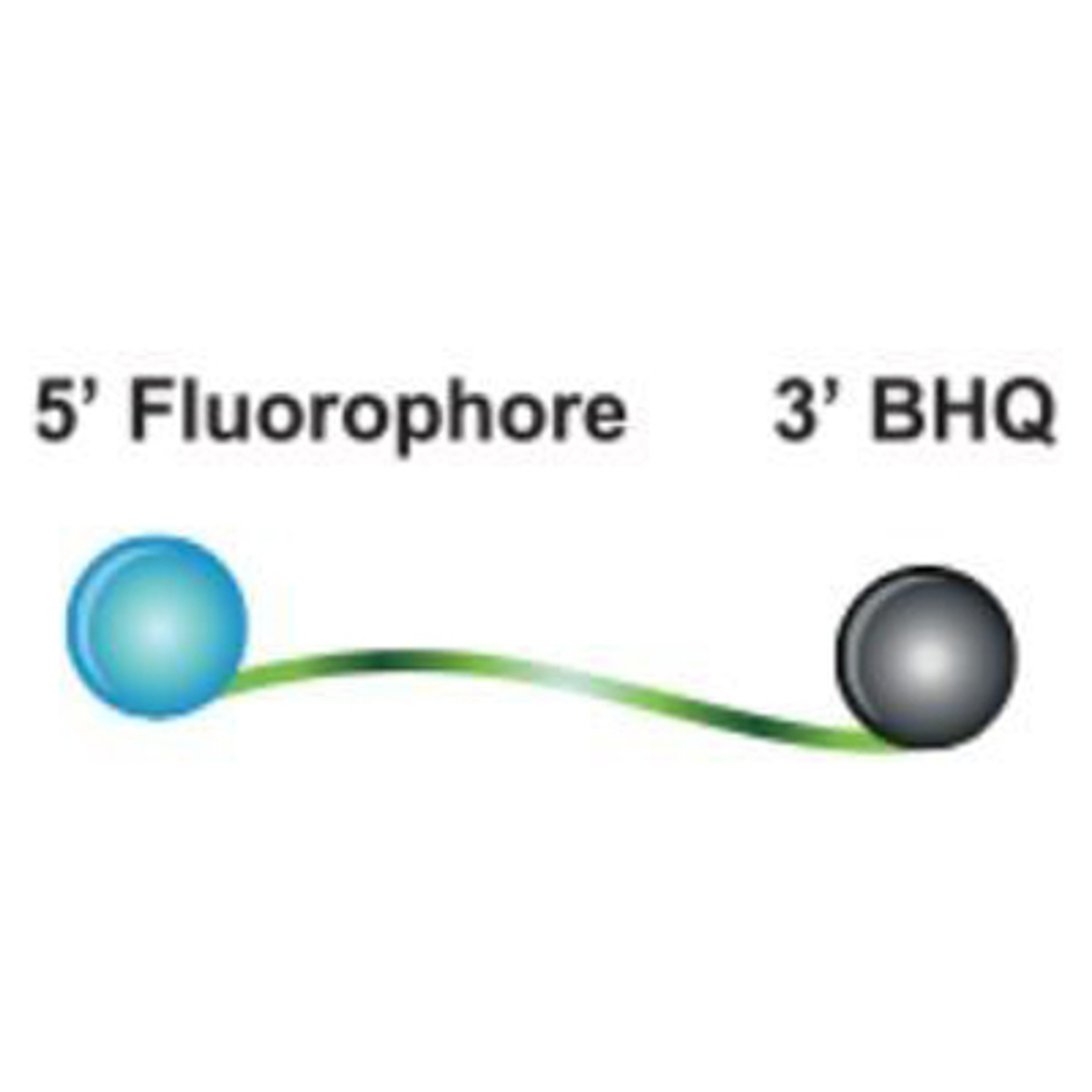
Nucleic acid extraction and purification for molecular diagnostics
Molecular testing protocols require reliable and high-quality isolation of genetic material from diverse sample types. In this infographic, LGC Biosearch Technologies explores how its portfolio of products and services can support your molecular testing workflows and shares how to select the best kits for your sample type and downstream requirements.
Tackling HPV detection and the global cervical cancer prevalence problem
Explore the concerted effort healthcare professionals, pharma, and biotech are making to combat cervical cancer through the development and implementation of new HPV assays
SARS-CoV-2 variants: Are Ct values an unused piece of the profiling puzzle?
Infectious disease scientist Dr. Padmapriya Banada explores the potential of cycle threshold values and reveals her melt-based assay alternative