iCell Cardiomyocytes
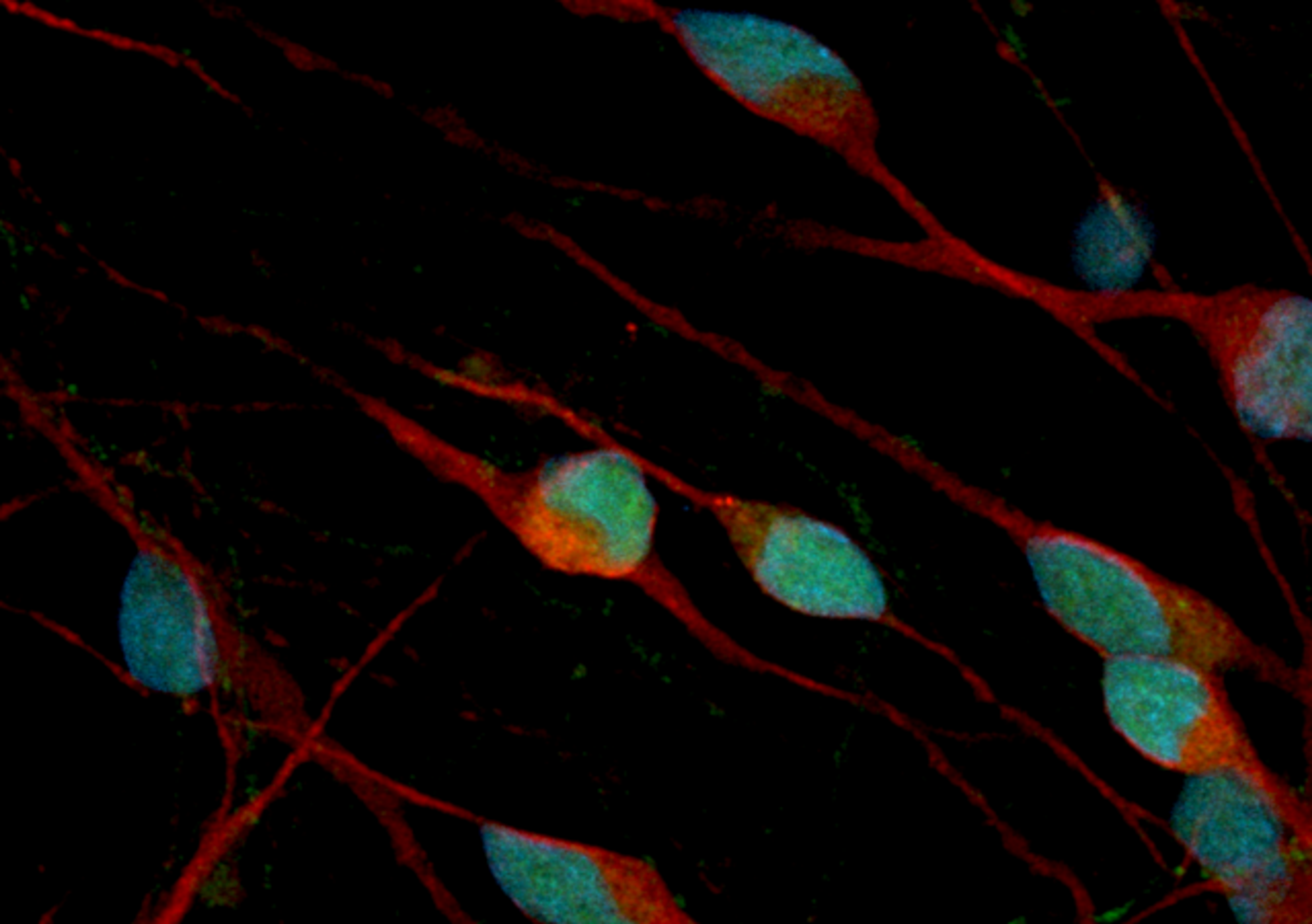
iCell® Cardiomyocytes, human induced pluripotent stem (iPS) cell-derived cardiomyocytes, aid drug discovery and improve the predictability of drug efficacy and toxicity screens, weeding out ineffective and potentially toxic compounds early in the pharmaceutical pipeline process before significant time and resources have been invested. iCell Cardiomyocytes are a mixture of spontaneously electrically active atrial, nodal, and ve…

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
iCell® Cardiomyocytes, human induced pluripotent stem (iPS) cell-derived cardiomyocytes, aid drug discovery and improve the predictability of drug efficacy and toxicity screens, weeding out ineffective and potentially toxic compounds early in the pharmaceutical pipeline process before significant time and resources have been invested.
iCell Cardiomyocytes are a mixture of spontaneously electrically active atrial, nodal, and ventricular-like myocytes that possess typical electrophysiological characteristics and exhibit expected electrophysiological and biochemical responses upon exposure to exogenous agents. Thus, these cells are a reliable source of human cardiomyocytes suitable for use in targeted drug discovery, toxicity testing, and other life science research.
iCell Cardiomyocytes are shipped as cryopreserved suspensions of dissociated cells with specifically formulated culture media for optimal cell performance. Once thawed, iCell Cardiomyocytes remain viable in culture for up to two weeks, allowing for acute and chronic studies.
iCell Cardiomyocytes Benefits:
- Human Cells - Saves valuable time, resources, and compound.
- Highly Pure Cell Population - Provides cardiac-specific response to reference molecules.
- Homogenous and Reproducible
- Fully Functional Model
- Acute and Longer-term Testing - Remain viable in culture for up to two weeks.
- iPS Cell-derived
iCell Cardiomyocytes Applications:
- Cell-based Assays - Cell viability, Apoptosis, ATP production, Oxidative stress, Mitochondrial dysfunction.
- Electophysiological Applications - Conventional patch clamp recording, Microelectrode assay (MEA) recording.
iPSC-Derived Cardiomyocytes and Luciferase Reporters: A Robust Reporting Platform for Monitoring Cardioprotection and Pathway Biology in Endogenous Human Tissue Cells
This application note aims to assess several properties of iCell Cardiomyocytes including transfection efficiency, feasibility of using luciferase reporter vectors, reporter activity, and the induction and suppression of ARE-driven gene expression.
Implementation of Human iPSC-Derived Cell Types into High Throughput Screening Workflows
This poster presents examples of assay miniaturization, transfection optimization, and high content imaging-based phenotypic assays to demonstrate the impact of iCell products in the drug discovery space. iCell Cardiomyocytes were used for disease modeling.
Multi-Parameter In Vitro Assessment of Compound Effects on Cardiomyocyte Physiology Using Induced Pluripotent Stem Cells (iPSC)
This poster describes methods for measuring the impact of drug candidates on the beating rate of human iPSC derived cardiomyocytes using fast kinetic fluorescence imaging.
Application of Human iPS Cell-Derived Models for Highly Predictive Toxicity Screening
This poster presents the development of an industrial-scale manufacturing platform for the production of terminally-differentiated, human iPS cell-derived tissue types (e.g. neurons, cardiomyocytes, and hepatocytes) that are highly pure (>95%) and exhibit normal genotypic, phenotypic, and functional characteristics of native cells.
Applications Development at CDI: Improving Workflows, Pushing Biology, and Enabling Screening
This poster highlights some of the on-going application development projects at CDI. These include general workflow improvements, phenotypic modeling of cardiac hypertrophy by high content screening (HCS) assay in 384-well format, modulating neuronal activity on multi-electrode arrays (MEA), and investigation of the bioenergetics of hepatotoxicity.
Human iPSC-Derived Cells for Modelling Cellular Bioenergetics: Building a Metabolic Profile Using the XF Mito Stress Test
In this study, the XF96 Extracellular Flux Analyzer was used to determine whether the iCell human cells represent a physiologically relevant cell model to study cellular metabolism. Results show that iCell Cardiomyocytes, iCell Neurons, and iCell Hepatocytes, offer a powerful tool to investigate bioenergetics in human cells.
Optical Measurements of Electrical Activity from hiPSC-Derived Cardiomyocytes is a Robust and High-Throughput Method for Measuring NCE-Effects on the Cardiac Action Potential
This poster presents data characterizing a novel optical platform, used in conjunction with Cellular Dynamics iCell® Cardiomyocytes, which offers a solution that overcomes the bottlenecks of low throughput and suitable tissue cells associated with traditional cardiac AP studies. This study investigates the effects of a range of pharmacological compounds on the cardiac AP, and demonstrates the suitability of these technologies for assessing NCE-mediated pro-arrhythmogenicity.
Advancements in the Use of iPS Cell-Derived Systems for In Vitro Disease Modeling and Phenotypic Screening
This poster demonstrates the impact of iCell products in the drug discovery and development space. Examples of assay miniaturization, transfection optimization, and high content imaging-based phenotypic assays are presented.
iCell® Cardiomyocytes: Human iPSC-derived Cardiomyocytes for Fully Functional and Comprehensive Assessments of NCE-mediated Effects
iCell® Cardiomyocytes are derived from human induced pluripotent stem cells (hiPSCs), are fully functional, surmount many of the hurdles associated with traditional models, and exhibit provide relevant drug induced effects. This poster explores the suitability of iCell® Cardiomyocytes for assessing the arrhythmogenic potential of NCEs.
iCell™ Cardiomyocytes: Assaying Cytoxicity
This application note describes the use of the Promega MultiTox-Fluor Multiplex Cytotoxicity Assay to perform a cell viability and cytotoxicity assay on iCell Cardiomyocytes. It is demonstrated that the Promega MultiTox-Fluor Multiplex Cytotoxicity Assay can be used to reliably assess drug compound induced general cytotoxic effects on iCell Cardiomyocytes independent of cell number.
Roberto Iacone, Roche, Discusses the Use of Stem Cells in Diabetic Cardiomyopathy Research
Dr. Iacone describes the incorporation of iPSC-derived iCell® and MyCell® Cardiomyocytes into their research program in diabetic cardiomyopathy associated with Type II diabetes and how these cells may enable development of personalized medicine.
Advancing Heart Research with iCell® Cardiomyocytes from Cellular Dynamics
Prof. Ulrich Broeckel, Medical College of Wisconsin, discusses the advantages of incorporating Cellular Dynamics' iCell® Cardiomyocytes into his research of left ventricular hypertrophy.
Cellular Dynamics Product Tour
Emile Nuwaysir, Chief Operating Officer of Cellular Dynamics International, introduces the range of iCell® products available from CDI. Watch this video to see time lapse footage of the cells growing.
Cellular Dynamics: Beating iCell® Cardiomyocytes
This video presents a synchronously beating monolayer of cardiomyocytes (heart cells) derived from induced pluripotent stem cells.
Cellular Dynamics: Handling iCell® Cardiomyocytes
Watch this training video from Cellular Dymanics International to learn about the storage, thawing, seeding, plating and maintainance of iCell Cardiomyocytes prior to performing you intended assay. Follow these techniques to maximize cell viability and ensure your success.