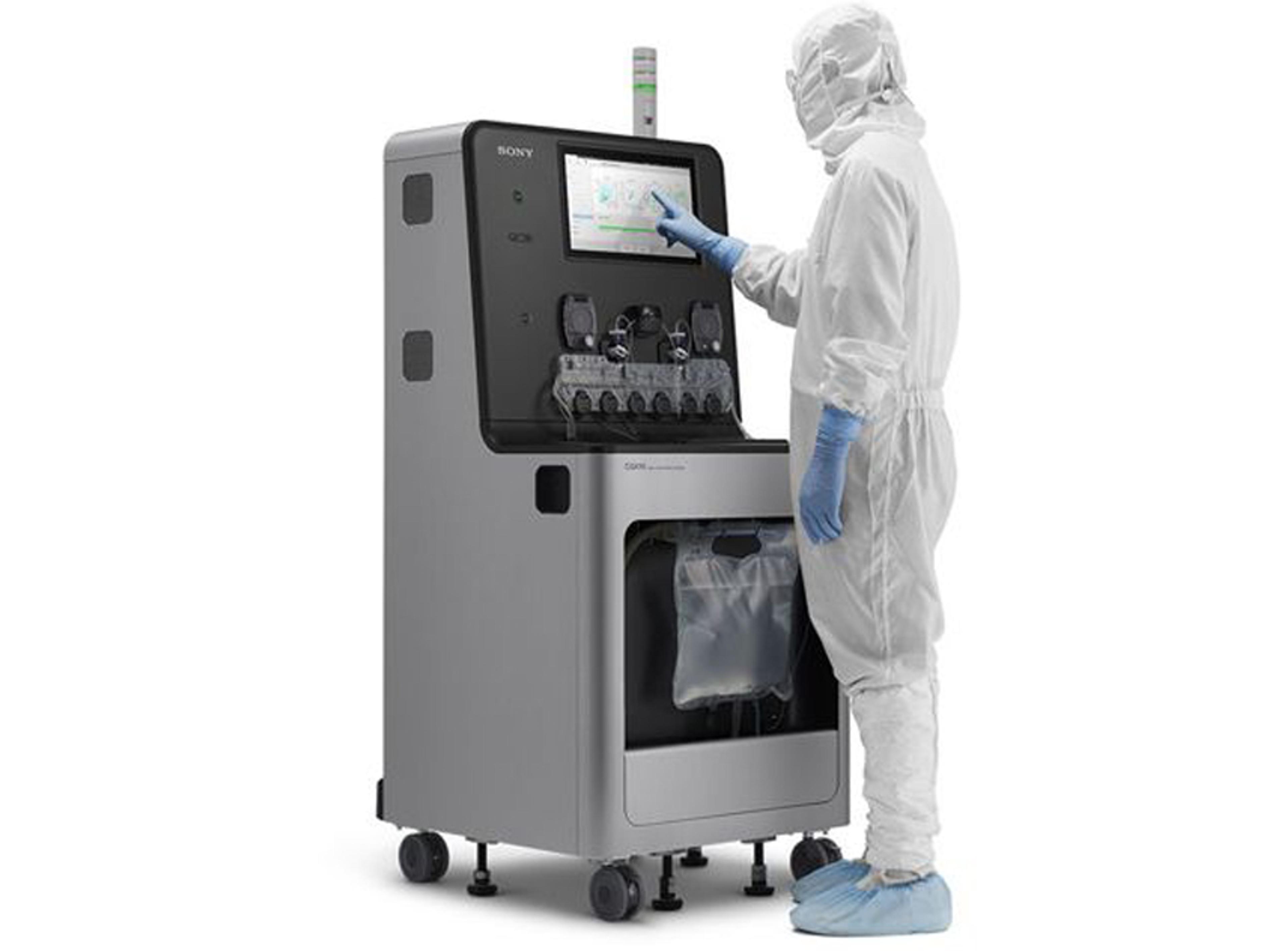
CGX10 Cell Isolation System
The CGX10 Cell Isolation System is a fully closed cell isolation system for GMP-compliant cell production and cell sorting applications. The innovative system design provides superior cell isolation along with ease of operation, with unparalleled flexibility that enables a smooth transition from process development to cell production.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Progress in the area of cell and gene therapies has accelerated significantly in recent times. Flow cytometry-based cell sorting has been used to sort cells by desired phenotype based on the presence of one or more biomarkers. Sorted cells have been expanded ex vivo and reinfused into patients as cell-based therapies. The ability to use multiple biomarkers is important. It enables identification and isolation of specific immune cells possessing the desired phenotype. Multi-marker-based cell isolation not only delivers a higher purity of isolated cells, but also enables selection to eliminate non-target cells, or therapeutically ineffective cells, for example, over-differentiated T cells.
However, the use of traditional flow cytometers has been limited to research laboratories and preclinical and early phase studies. The complex design of conventional flow cytometers and their usability issues makes them poorly suited to a cell production environment that is GMP compliant. Certain conventional flow cytometers have also been known to shear or cause electrically-induced stress or damage to sorted cells.
The CGX10 Cell Isolation System has been developed with the goal of enabling multi-marker selection to isolate target cells while ensuring high cell viability. The system delivers a user experience that is suited to the GMP compliant, cell production environment, and transcends the complexity and subjectivity of conventional sorter platforms.
The CGX10 Cell Isolation System and related products are intended for use by trained laboratory technicians in research, process development, or manufacturing environments all related to ATMP/regenerative medicine, including cell and gene therapy. The CGX10 instrument and related products are for ex vivo cell separation processing only, and are not intended for therapeutic, diagnostic, or human in vivo applications. Any clinical application of the cells is exclusively within the responsibility of the user of the CGX10 instrument and related products. For the manufacturing and use of cells in humans, regulations must be followed. The CGX10 Cell Isolation System and related products are not sold as medical devices.
The CGX10 Cell Isolation System is classified as a Class 1 laser product.
Brochures
CGX10 Cell Isolation System: Cell isolation you can count on
In this product brochure, Sony Biotechnology describes the CGX10 Cell Isolation System, a fully closed cell isolation system for GMP-compliant cell production and cell sorting applications.
CGX10 Cell Isolation System
In this application note, Sony Biotechnology describes the technical specifications of its CGX10 Cell Isolation System, a fully closed system for GMP-compliant cell production and cell sorting applications.
Isolation of highly pure and viable T and NK cells using the CGX10 Cell Isolation System
In this application note, Sony Biotechnology describes the use of the CGX10 Cell Isolation System, a closed system suitable for GMP–compliant cell separation, for large-scale enrichment and isolation of T and NK cells for cell therapy.
GMP-compliant cell sorting by flow cytometry
Jerry Barnhart and Marc-Aurele Brun of Sony Biotechnology discuss the role of flow cytometry in cell and gene therapy research, and how multiparametric approaches can address key challenges with rare population sorting. Here, they highlight how the CGX10 Cell Isolation System is designed for automated and GMP-compliant cell sorting at high speeds, purity, and flexibility.
Explore full insights from Jerry and Marc in the editorial article.
Multiparametric selection enables GMP-ready cell production
Learn about the development of the GMP-ready CGX10 Cell Isolation System and how it can transform cell and gene therapy workflows