Neurodegenerative Diseases: Latest Advances in Technology and Research
A mass spectrometry breath test, artificial intelligence algorithms and novel anti-inflammatory compounds among the innovations that could pave the way for better diagnoses and treatment
25 Oct 2019
In 2016, a study published in the Lancet found that nearly 44 million individuals worldwide were living with dementia – more than double the figures reported in 1990. Dementia is a symptom of neurodegenerative disease, an umbrella term to describe the irreversible and progressive damage to neurons in the brain, of which there are several disease types – each characterized by their origin in the brain. They include: Alzheimer’s, Parkinson’s, Huntington’s, motor neurone and prion disease, as well as spinal muscular atrophy and ataxias, among others. Interestingly, the 2016 study found that nearly two thirds of those diagnosed with dementia are female.
Whilst increasing prevalence can be largely attributed to an ageing population and improved diagnostic regimens, the past two decades have seen few successes in challenging this global disease affliction. Some of the greatest roadblocks lie in identifying early disease biomarkers and developing physiologically relevant assay formats. In this special editorial feature, we present some of the latest technologies and research advancing the field. From new methods to track plaque formation in real time, to the promise of a mass spectrometry diagnostic breath test, read on to discover what the future holds for neurodegenerative diseases.
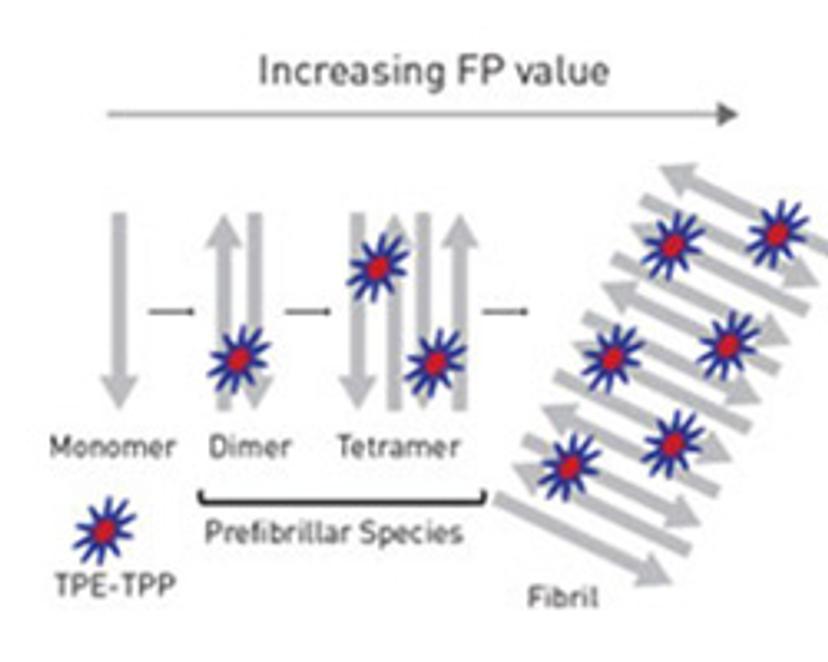
Unravelling β-Amyloid Aggregation
The misfolding of cytoplasmic proteins into insoluble fibrillar deposits is characteristic of many neurodegenerative diseases and considered to be the leading cause of neuronal cell death. Protein fibrils activate a whole host of downstream pathophysiological pathways, including oxidative stress, membrane depolarization and inflammatory signaling. The study of these progressive changes in protein structure – notably, β-Amyloid and Tau proteins – has undergone a recent resurgence, yet the highly complex nature of aggregation makes them difficult to study. There is a lot of interest, therefore, in the production of probes for real-time in vitro analysis of fibril self-assembly, where previous methods have been limited to the study of FFPE tissue with already established protein plaques.
BMG Labtech has now developed a novel probe for the monitoring of prefibrillar aggregation of proteins. Its technique utilizes fluorescence polarization (FP) technology, which involves measuring the rotation of molecules in solution by polarized light. The rate at which fluorophore-protein complexes rotate is dependent on the mass, enabling the identification of proteins at different stages of assembly. Download this white paper to understand why the technique is thought to be superior to thioflavin T for amyloid biosensing.

Neuroinflammation: The Real Cause of Plaque Formation?
Recent findings have revealed the innate cerebral immune cells, microglia, as potential mediators – or even initiators – of degenerative disease. Pharmaceutical company INmune Bio Inc. has translated this knowledge into developing a novel therapy for Alzheimer’s disease. Its new compound has a unique activity: it sequesters the immune-mediating protein TNF that, in turn, normalizes microglia and the synaptic activity of neurones. Clinical trials are due to start this month, following a $1 million investment from the Alzheimer’s Association.
The company is also due to implement some exciting technology for monitoring patient response: a VOC-based mass spec breath test. Find out more about this potentially paradigm-changing approach in this exclusive interview with Dr. C.J.Barnum, Director of Neuroscience at INmune Bio.
Improving Quality and Quantity of Viable Cells
Understanding the molecular basis to neuropathology relies on the analysis of physiologically relevant models, including primary human or mouse tissue. For single-cell analysis, intact viable cells must be prepared in solution. Disruption of cell health and integrity during this process, however, may introduce subcellular remnants and DNA debris into the solution, distorting the quality and reliability of data produced. Furthermore, inadequate cell separation reduces data quality where conglomerates of cells remain.
In this video, Andreas Bosio, from Miltenyi Biotec, describes an automated brain tissue dissociation protocol for effective removal of debris and ethryocytes, and high-quality isolation of oligodendrocytes, neurons, astrocytes and microglia using the GentleMACS Dissociator and magnetic isolation technology.
Understanding Cell Behavior in Exquisite Detail
Much of our understanding of cell behavior and function come from post-mortem analyses and fixed-cell staining. Whilst this reveals the presence or absence of important cell molecules, it fails to permit the evaluation of cell-to-cell interactions, changes in protein expression over time, and other dynamic processes. As technology has advanced, live-cell analysis has become an integral tool in drug discovery and translational research, particularly as 3D culture technologies have improved as well.
Experienced neuroscientist Susana Alcantara recently delivered a webinar, now available to watch on demand, in which she describes how the latest high-content technology has enabled her to gain greater biological insight into neuro-toxicity, neuro-degeneration and neuro-inflammation. Presenting recently published data, Alcantara reveals how to conduct automated assays with stem cell and primary neuronal cultures.
AI and the Alzheimer’s Algorithm
To date, most diagnostic tests for dementia are based on non-discrete memory and function tests. However, at the point of clinical presentation of these symptoms, neuronal protein plaques are significant and irrevocable. Identifying biomarkers indicative of earlier-stage disease, therefore, could help to identify druggable protein targets to halt disease progression. Thus far, progress has been limited, owed largely to the multi-faceted and heterogeneous nature of neurodegenerative disease that could be mediated by multiple, interacting factors.
To address this unmet need, scientists at the University of Southern California harnessed the power of machine learning to expose patterns of biomarker expression in Alzheimer’s patients. Computer analysis of vast quantities of data was able to decipher distinct pathological algorithms. Find out more about this exciting work in this article.
Center Stage for Microglia
Microglia are the immune cells of the brain, and their dysfunctional activation is associated with neurodegeneration. Their roles in potentiating disease aren’t entirely clear, since there is abundant evidence to suggest they can be both neuroprotective and neurotoxic. In the healthy brain, microglia ‘sense’ their surroundings, and up or down-regulate protein expression levels to maintain immunological balance. Therefore, much like the immune system is circumvented in cancer, it is likely that microglial function becomes defective during disease, eventually reprogrammed to foster progression. For example, clearance of Amyloid β species by microglia is defective in dementia, and microglial cells have been demonstrated to actively secrete toxins to injure neuronal synapses.
More research is required to unravel their disease-mediating effect. Yet, limitations exist in cell line accessibility and the physiological variations associated with those available. Axol Bioscience has recently launched a novel iPSC-derived microglial cell line that overcomes many of these caveats. Find out more in this article.

Cannabinoids in Dementia
A leading UK university, King’s College London, has entered an exciting endeavor with Alzheimer’s Research UK to investigate the efficacy of cannabinoid compounds in dementia patients. The trial, termed the STAND (Sativex® for the Treatment of AgitatioN in Dementia) trial, will look at alleviating the psychiatric symptoms of dementia, which many consider to be the most devasting.
Administered as a mouth spray, Sativex® contains a 1:1 ratio of two main cannabinoids found in the cannabis plant and has an already-established pharmacokinetic and safety profile in patients. You can read more about this collaboration in this article.
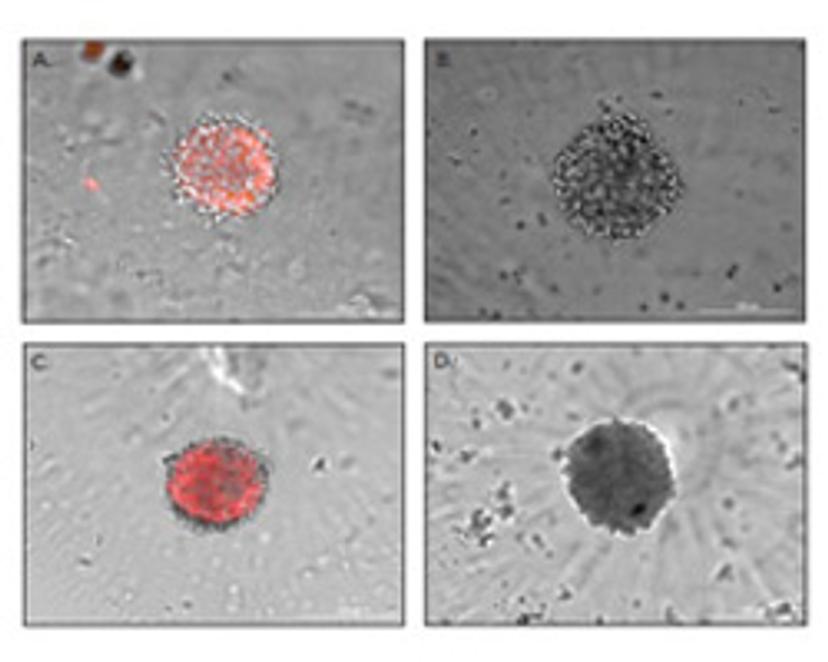
Reproducible Neurosphere Formation
Animal models are an important part of pre-clinical research but can be time and cost-intensive. 3D cell culture models, therefore, are becoming increasingly popular in drug discovery. They permit a more biologically-relevant response whilst enabling high-throughput workflows to be implemented.
Using ultra-low attachment spheroid microplates, this white paper demonstrates a method for reproducible neurosphere generation from human neural stem cells, for drug compound or neurotoxicant testing.



