Why consider an FDA-cleared device for your genetic tests and molecular diagnostics
24 Feb 2026
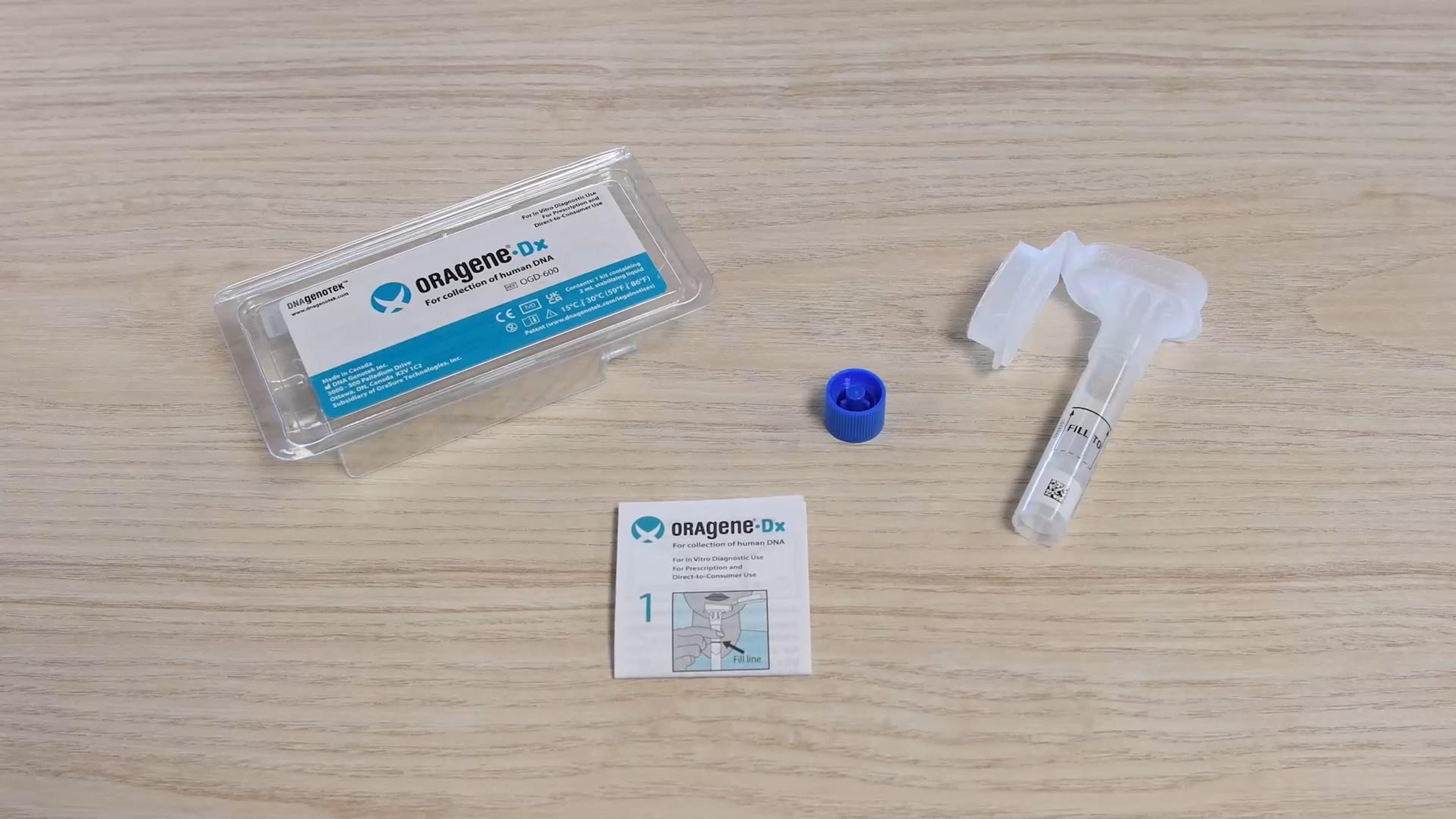
The Oragene™•Dx and ORAcollect™•Dx devices have US FDA 510(k) general clearance for prescription and over-the-counter (direct-to-consumer) uses. This designation allows this device to be used by commercial partners and legally marketed with their therapeutics or devices in conjunction with their product's intended use. Discover how an FDA-cleared device may offer advantages such as:
- Validated performance and ease of use
- Reduced timeline to validate your Laboratory Developed Assay (LDT)
- Cost-effective market path for your genetic test
- Reduced regulatory burden when filing an FDA submission for your assay
- Customization offerings for your test (branding and fulfillment services)
About the company

DNA Genotek Inc.
OraSure Technologies, Inc., with its subsidiaries, DNA Genotek and Sherlock Biosciences, is a leader in the development, manufacture, and distribution of rapid diagnostic tests and sample collection and stabilization devices to discover and detect medical conditions.
DNA Genotek Inc. is a subsidiary of OraSure Technologies, Inc. and provides collection and stabilization devices for biological sample types, such as saliva, blood, stool, and urine. Our product lines include the Oragene™•Dx and ORAcollect™•DNA/RNA devices. Saliva collection can be done by either a healthcare professional at point-of-care or using at home DNA collection kits.
The Oragene™•Dx and ORAcollect™•Dx saliva collection devices have U.S. FDA 510(k) general clearance. Leveraging the 510(k) clearance allows molecular test manufacturers to immediately use these saliva collection devices in either a prescription or direct-to-consumer model.
*Some DNA Genotek products may not be available in all geographic regions. Customers must review the labels (RUO or IVD) of sample collection devices and ensure compliance for their intended use.