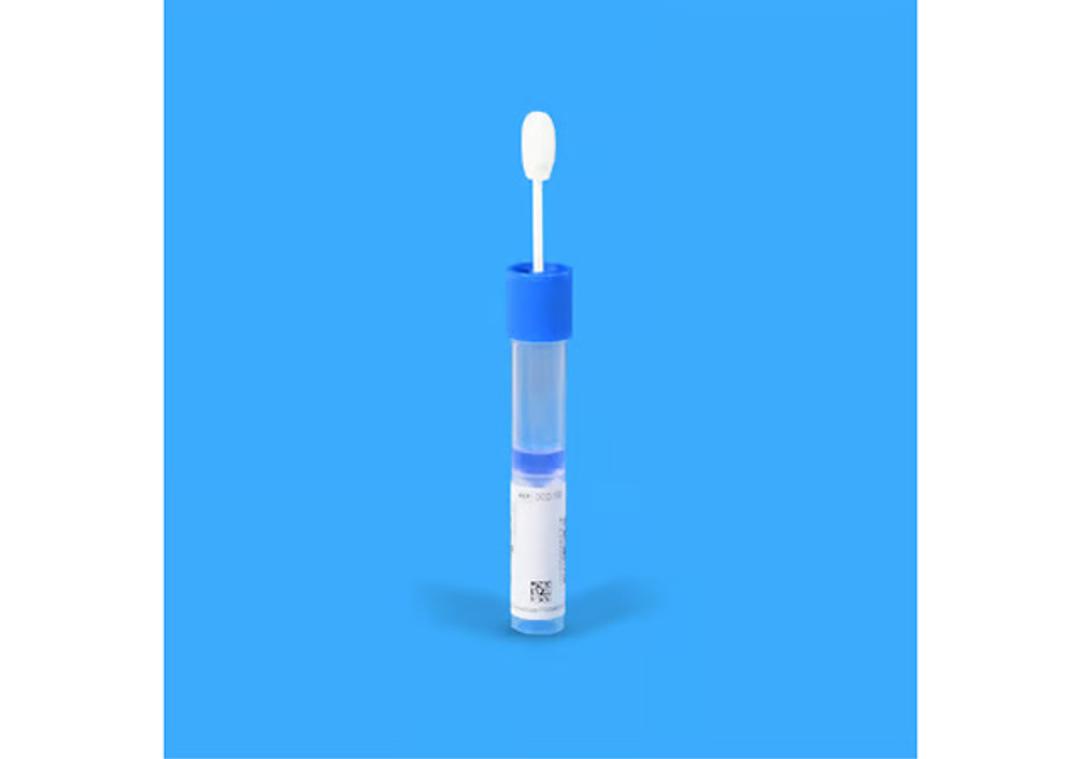
ORAcollect™•Dx saliva collection device
Collect high-quality DNA samples for molecular diagnostics using an FDA cleared device.
- U.S. FDA 510(k) general clearance for prescription (Rx) and direct-to consumer (DTC) uses
- Painless, minimally-invasive and easy-to-use DNA sample device for self collection
- Unlike buccal swabs, collected samples are liquid-based, bacteriostatic and integrate easily in existing lab workflows
Device / Assay manufacturers must validate the use of this saliva collection device for use with their device.
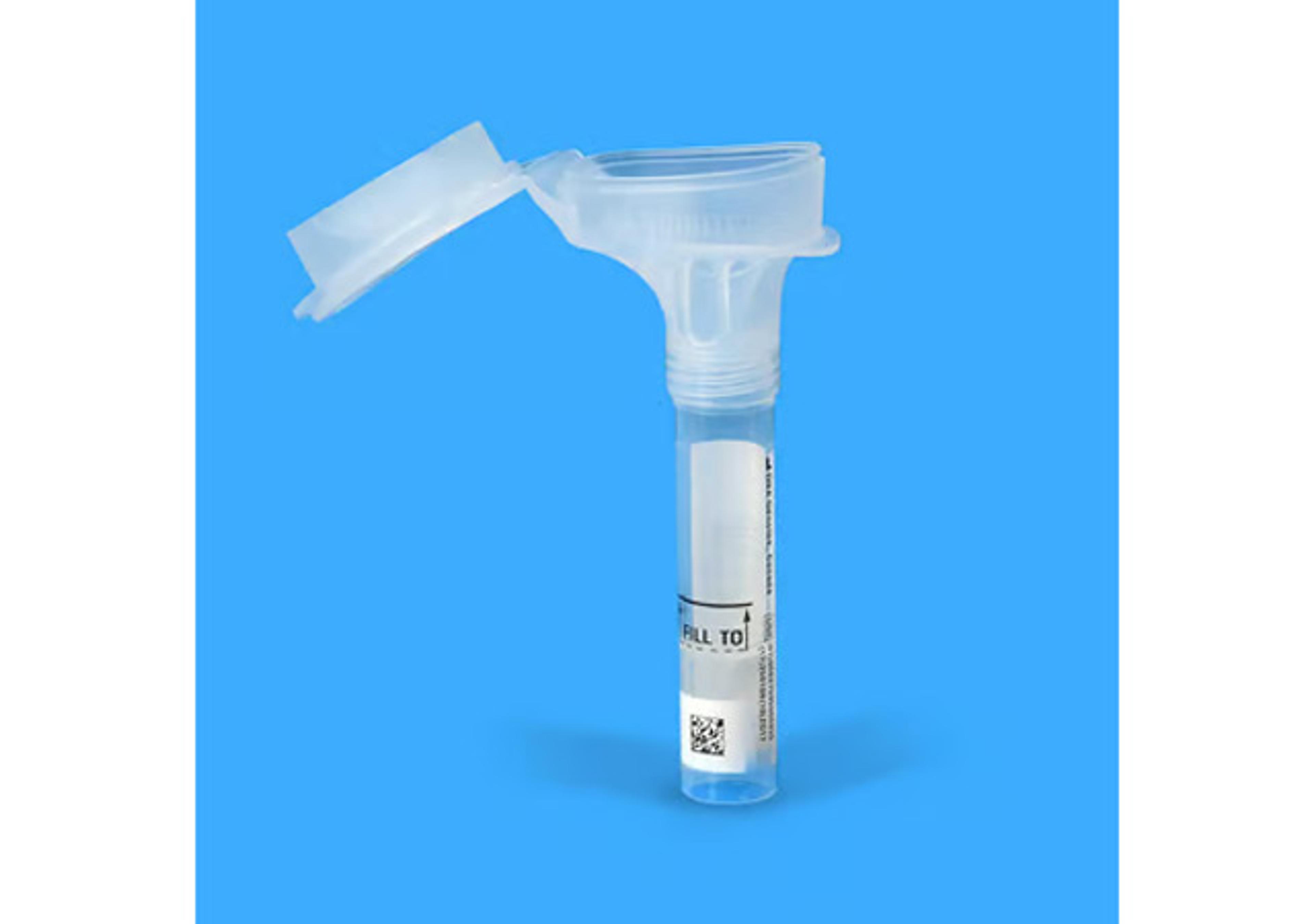
Sample collection instructions for the ORAcollect™•Dx device
Learn how to properly use the ORAcollect™•Dx in vitro diagnostic device for safe, reliable oral DNA sample collection. This step-by-step demonstration walks users through preparation, collection, stabilization, and transport procedures. FDA-cleared for prescription and over-the-counter use, ORAcollect™•Dx offers a simple, non-invasive solution for collecting high-quality human DNA from oral samples while ensuring compliance with biological specimen transport regulations.
Why consider an FDA-cleared device for your genetic tests and molecular diagnostics
The Oragene™•Dx and ORAcollect™•Dx devices have US FDA 510(k) general clearance for prescription and over-the-counter (direct-to-consumer) uses. This designation allows this device to be used by commercial partners and legally marketed with their therapeutics or devices in conjunction with their product's intended use. Discover how an FDA-cleared device may offer advantages such as:
- Validated performance and ease of use
- Reduced timeline to validate your Laboratory Developed Assay (LDT)
- Cost-effective market path for your genetic test
- Reduced regulatory burden when filing an FDA submission for your assay
- Customization offerings for your test (branding and fulfillment services)