Streptavidin from Streptomyces avidinii
essentially salt-free, lyophilized powder, ≥13 units/mg protein

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
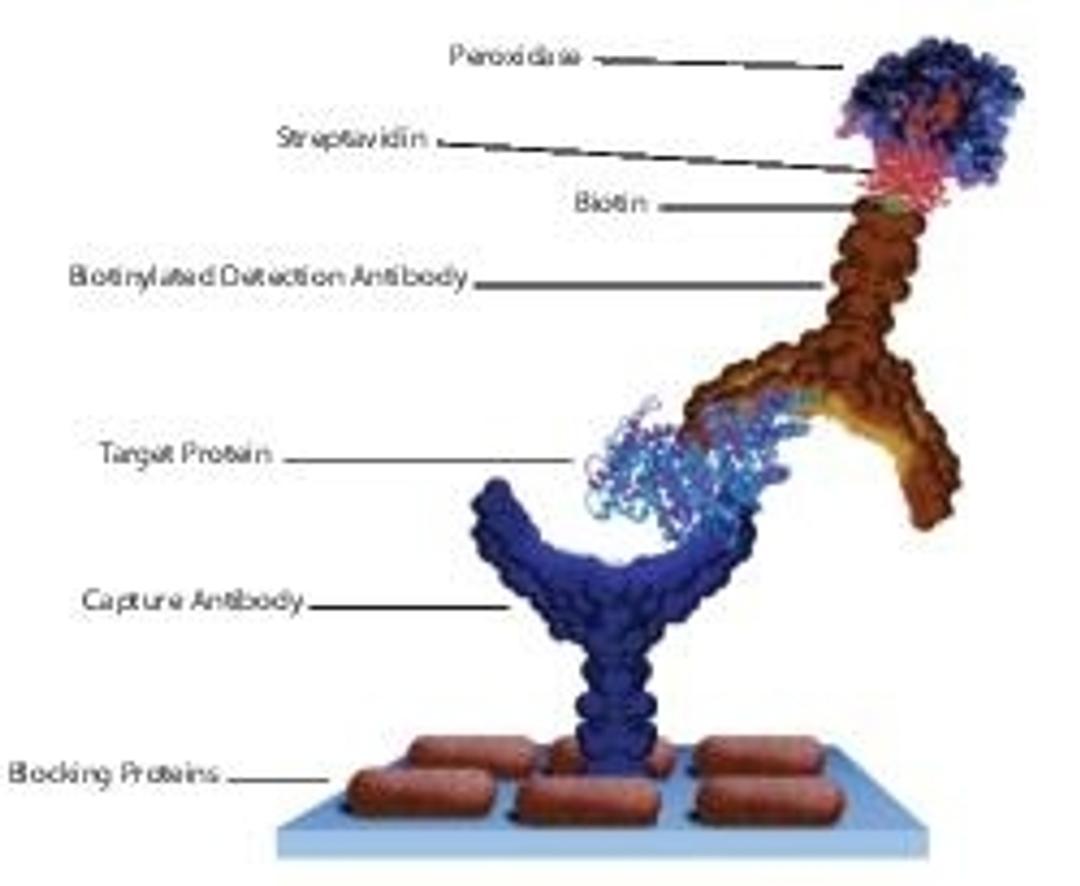
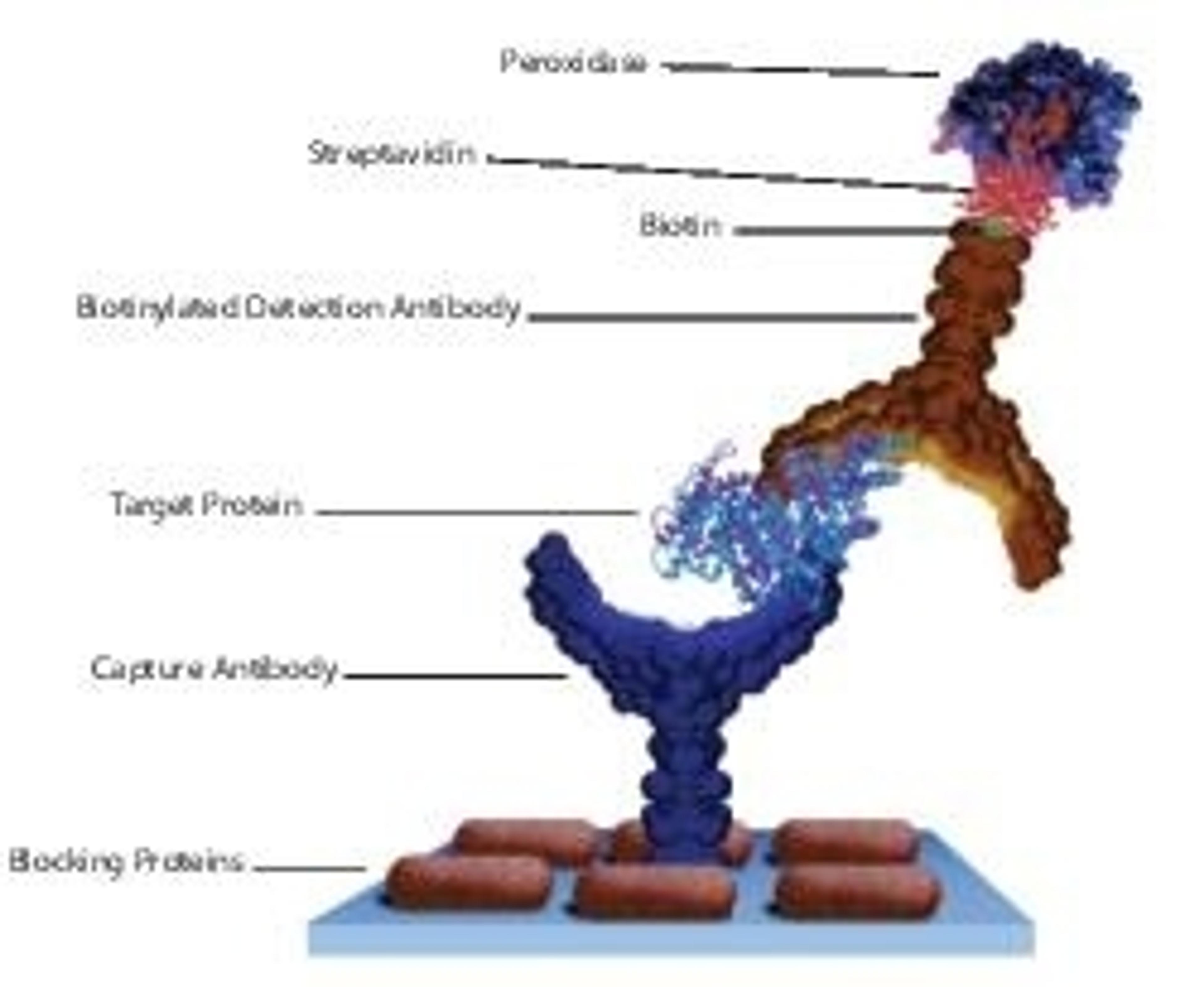
Streptavidin derives its name from its bacterial source Streptomyces avidinii and from the hen egg-white protein, avidin, which has high affinity to biotin. Its homologous core shares 33% sequence similarity with avidin, as well as sharing a common tetrameric structure. It is a crystalline tetrameric protein, with a molecular weight of 4*15000Da. It binds four molecules of biotin. Streptavidin lacks carbohydrate and sulfur-containing amino acids.
Streptavidin is a crystalline protein isolated from the bacterium Streptomyces avidinii and possesses biotin-binding ability. It has four binding sites for biotin like avidin and binds four molecules of biotin. Streptavidin is now considered as a new type of biotin-binding protein.