SepsiTest™-UMD CE IVD
Microbial DNA enrichment, isolation and PCR detection

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
SepsiTest™-UMD is a CE IVD-marked kit for the culture-independent detection of bacteria and fungi in clinical samples. The detection and identification of bacteria and fungi is based on broad-range 16S and 18S rRNA gene PCR followed by Sanger sequencing. The sensitivity of molecular analysis is often limited due to the vast excess of human DNA compared to the low bacterial and fungal load. The human DNA depletion prior to the microbial DNA isolation overcomes this limitation. Reagent-borne contaminations are another risk counteracted with guaranteed DNA-free reagents and consumables provided in SepsiTest™-UMD. The amplification with 40 PCR cycles allows the sensitive detection of even minute amounts of bacterial and fungal DNA and provides a first result after approximately four hours. PCR-positive samples are analysed with Sanger sequencing to identify the pathogens. Diverse specimens can be analysed with SepsiTest™-UMD, including different body fluids, swabs and tissue biopsies.
Benefits:
- Variety of specimens: body fluids, swabs, tissues
- Flexible amount of samples
- Increased sensitivity by depletion of host DNA
- Contamination-free reagents and consumables
- Broad-range 16S and 18S rDNA PCR assays
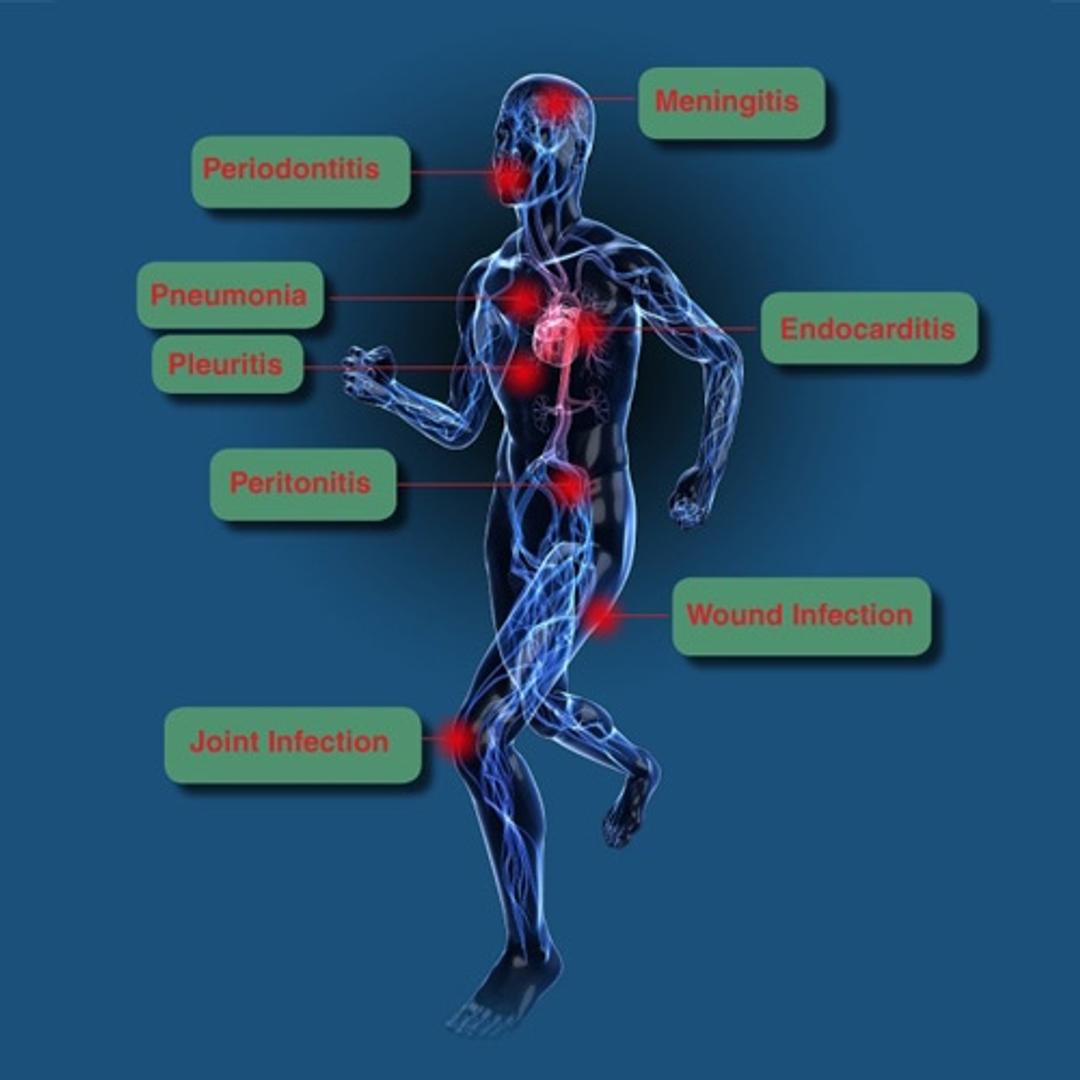
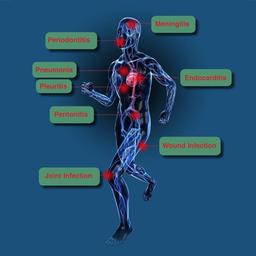
SepsiTest™-UMD is particularly suited for the detection of pathogens in samples from patients pretreated with antibiotics and for fastidious organisms negative in culture. More than 1.300 pathogens have been identified with SepsiTest™-UMD so far, among others, pathogens causing sepsis, bacterial meningitis, pneumonia, joint and wound infections.
Brochures
Broad-Range MDx: SepsiTestTM-UMD
Universal Pathogen Identification - SepsiTest™-UMD is a precise culture-independent routine diagnosis to address non-growing pathogens under antibiotic treatment and rare pathogens, including anaerobic and fastidious organisms.
Broad-range molecular detection of rare and fastidious pathogens
Molzym presents case reports and studies on the diagnosis of various culture-negative bacteria and fungi in which MMDx enabled the identification of, for example, Candidatus Neoehrlichia mikurensis, Malassezia restricta, Bartonella spp., and Tropheryma whipplei, as well as other fastidious or anaerobic, slow-growing or non-cultivable microorganisms. Download this application note to find out how you can utilize the MMDx broad-range diagnostic workflow in your research.
Molzym announces FDA Breakthrough Device Designation for its molecular diagnostic test system
FDA Breakthrough Device Designation for Sepsis, Joint & Implant Infections, Infective Endocarditis, Bacterial Meningitis
Latest Advances in Infectious Disease Diagnostics: SelectScience Special Feature
From rare pathogens to pandemic prevention, explore the latest techniques and technologies that are shaping today’s diagnosis and treatment of infectious diseases