ioSkeletal Myocytes DMD Exon Deletion disease models - Human iPSC-derived model of Duchenne muscular dystrophy
ioSkeletal Myocytes DMD Exon Deletion disease models are built to help improve the translatability of data generated during the discovery of new candidate drugs for Duchenne muscular dystrophy.
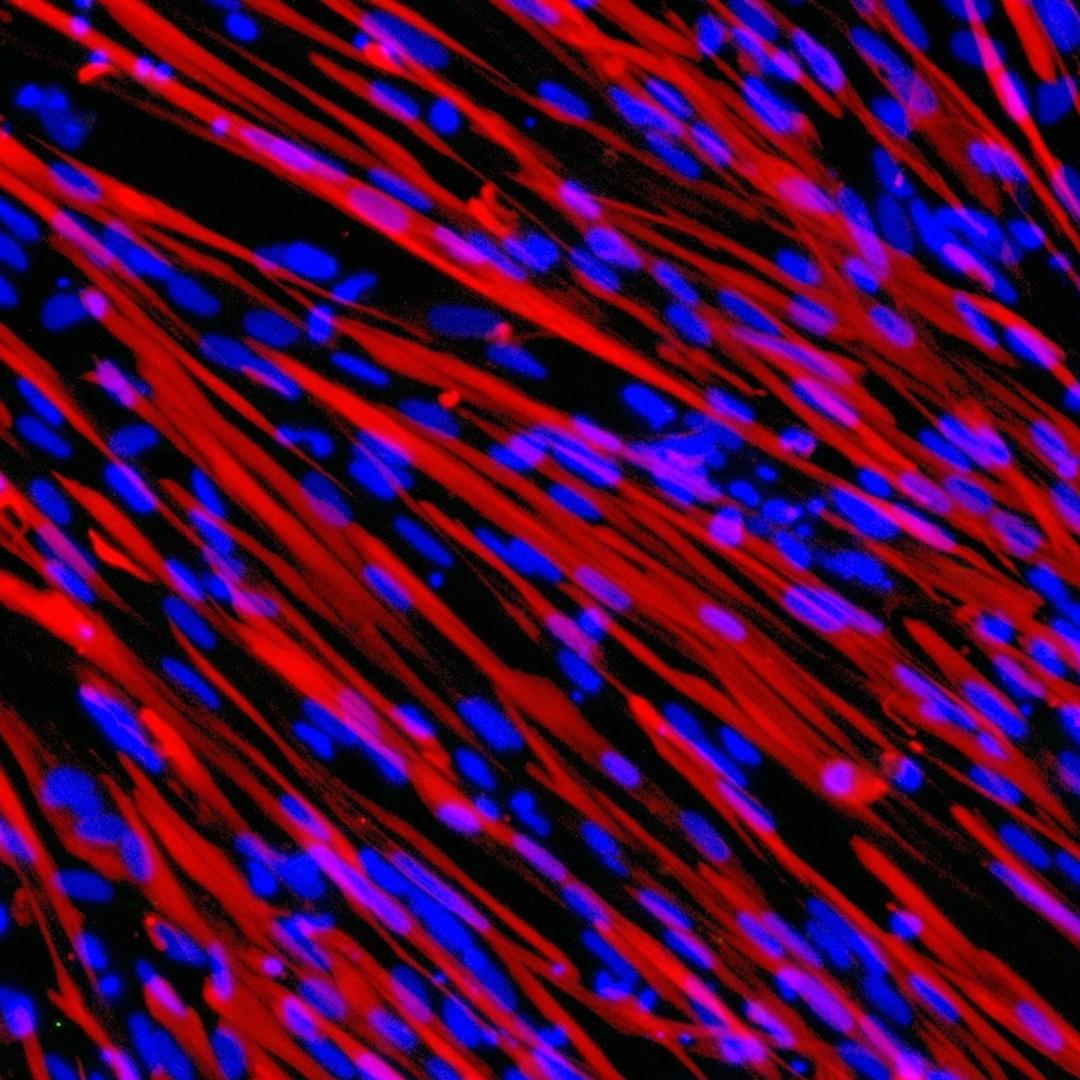
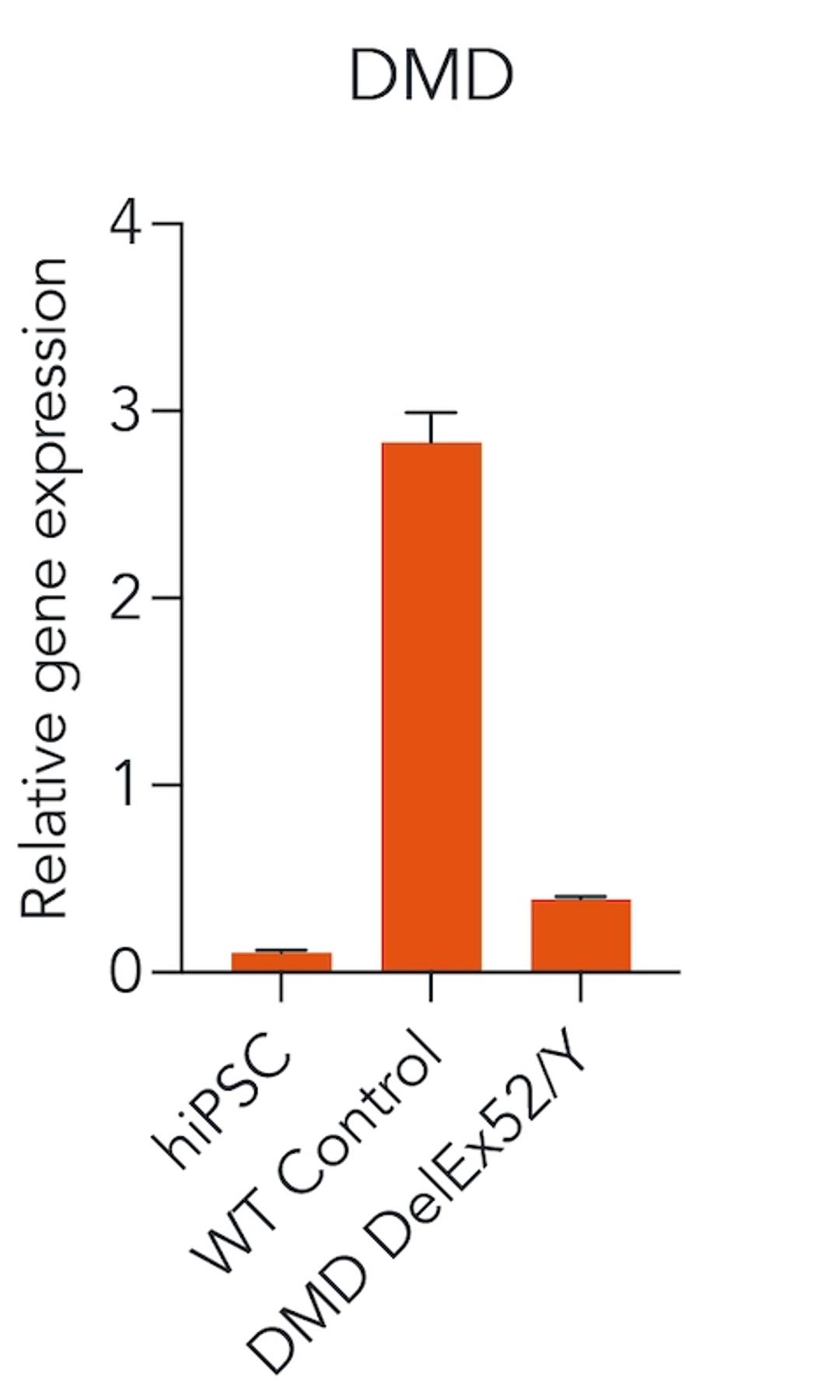
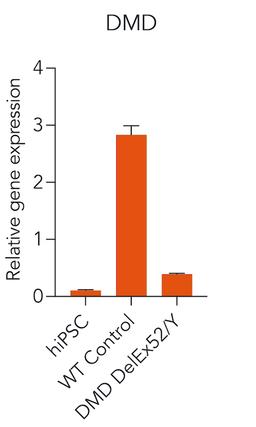
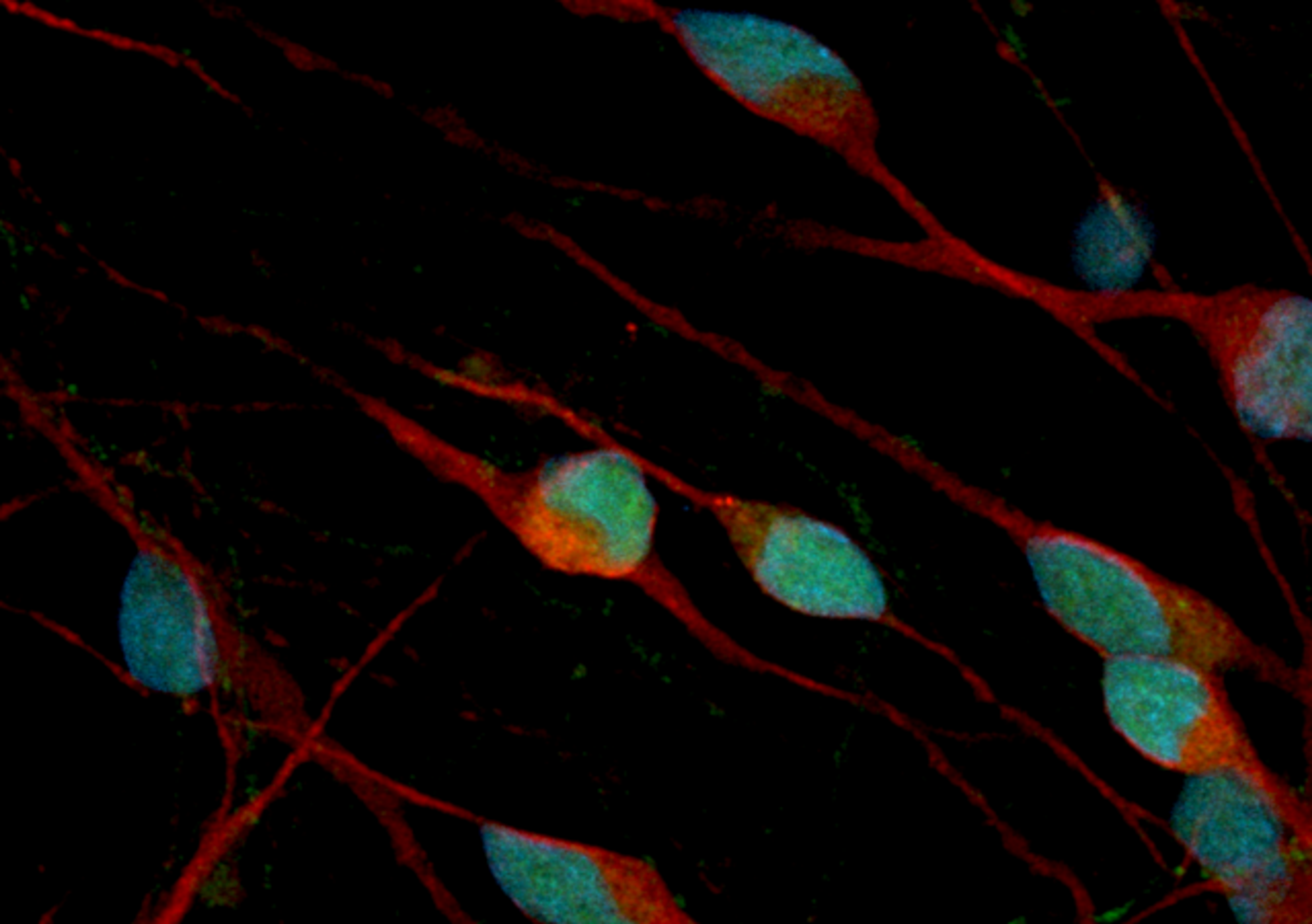
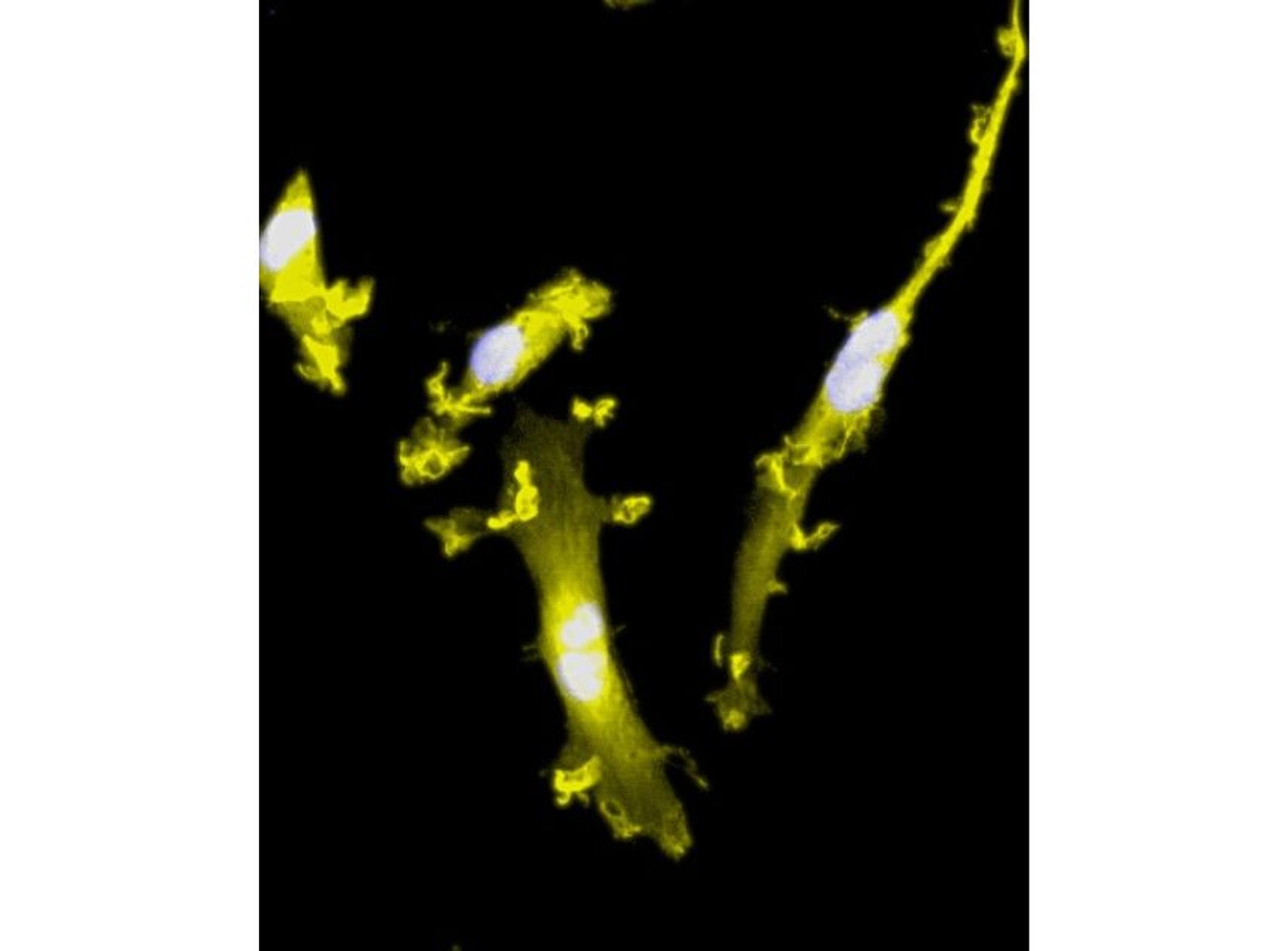
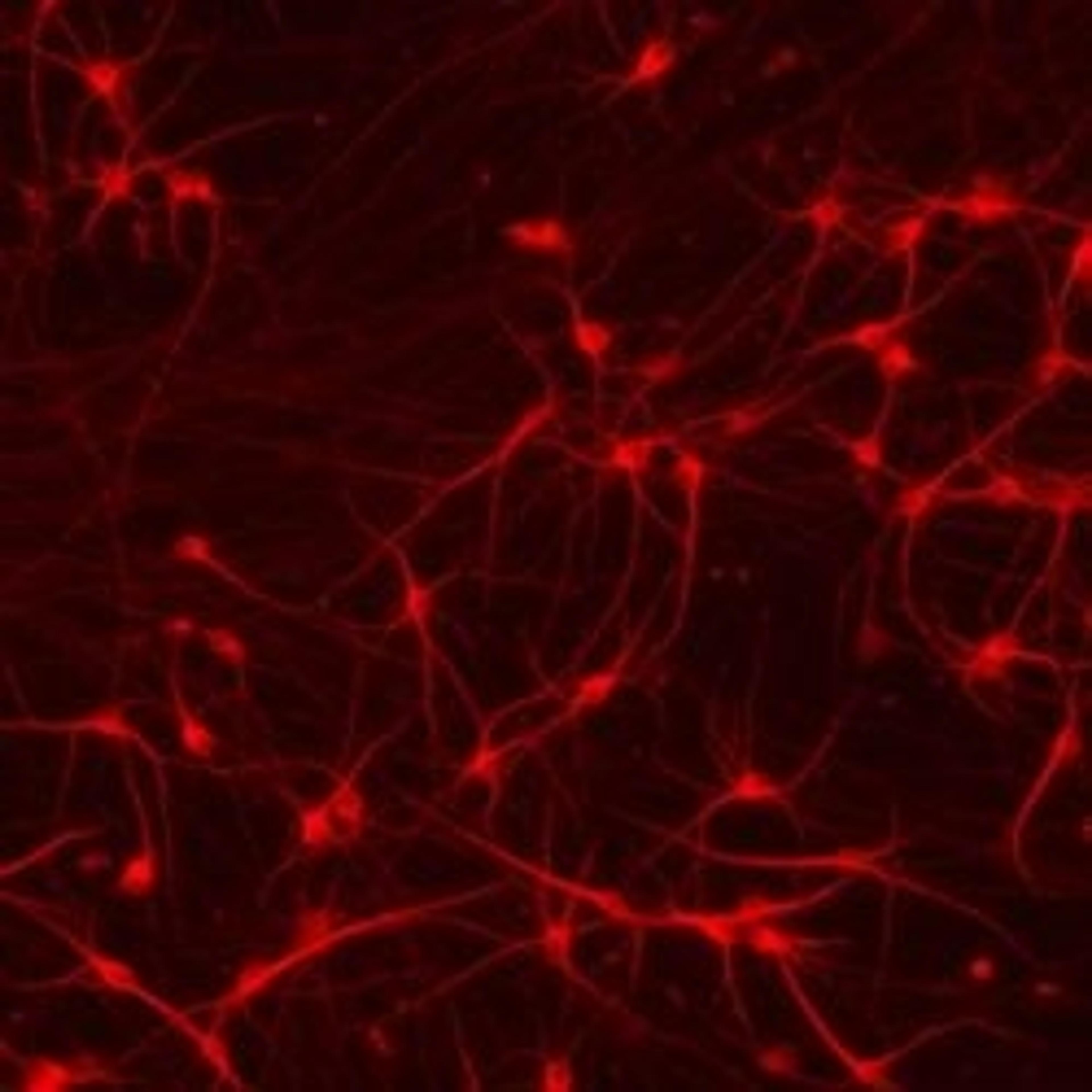
ioSkeletal Myocytes DMD Exon Deletion disease models are first-of-their-kind human iPSC-derived skeletal muscle cells containing single exon deletions in the DMD gene. The cells lack dystrophin expression, as observed in Duchenne muscular dystrophy patients. Independent partners have successfully restored dystrophin expression in these cellswith antisense oligonucleotide-mediated exon skipping, as part of their drug discovery program. Scientists are enabled to study the exon deletion’s impact on human muscle biology and investigate methods of dystrophin restoration in a human and disease-relevant context, leading to more translatable data.
As the ioSkeletal Myocyte DMD Exon Deletion disease models are powered by opti-ox they have consistency built in. The cells can also be paired with an ioWild Type isogenic control, so any observed differences can be confidently attributed to the exon deletion in the DMD gene. These features enable researchers to simplify and standardise their cell culture. This supports the scale-up of cell-based assays to 384-well multi-plate formats, ultimately accelerating the identification of potential drug candidates.
Key features
- Disease-related phenotype: Immunocytochemistry shows a lack of dystrophin in the DMD Exon 44 Deletion cells, and dystrophin restoration has been demonstrated by ASO exon skipping.
- Consistent: Our platform ensures consistency, scalability, and reproducibility, overcoming the challenges associated with the use of primary muscle cells and immortalised cell lines.
- Make true comparisons: Pair the DMD disease model cells with the genetically matched wild-type skeletal muscle cells to study the impact of the deletion, or test methods for dystrophin restoration.
Applications
- ASO-mediated exon skipping
- Muscular dystrophy research
- Dystrophin restoration
- Muscle disease modelling
Tackling the reproducibility crisis with standardized human cells
Tuesday, April 29, 2025 at 16:00 BST / 17:00 CEST / 11:00 EDT / 08:00 PDT
Join experts in human iPSC-derived cells from academia and drug discovery as they discuss the impact of standardization on scientific progress.
The absence of standardized reagents creates barriers to reliable experimental comparisons, slowing research, drug development, and translational efforts. Across human cell models, including primary cells, cell lines, and patient-derived iPSC-derived cells, biological variability and reproducibility challenges persist.
In this expert panel, we will explore how standardizing human iPSC-derived cells can enhance consistency, reproducibility, and regulatory compliance, ultimately enabling more robust and impactful research.
Key learning objectives
- Explore the challenges posed by variability in human cell models
- Understand why a lack of standardized cell models impacts reproducibility, and how implementing it can improve research across academia and industry
- Gain insights on the standardization of human iPSC-derived cells from leaders in academic research and drug discovery
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.
bit.bio launches two new human cell disease models to advance discovery and development of treatments for Duchenne muscular dystrophy (DMD)
Models will be used to investigate, screen for and develop therapies for the restoration of dystrophin, the protein that is missing in DMD patients