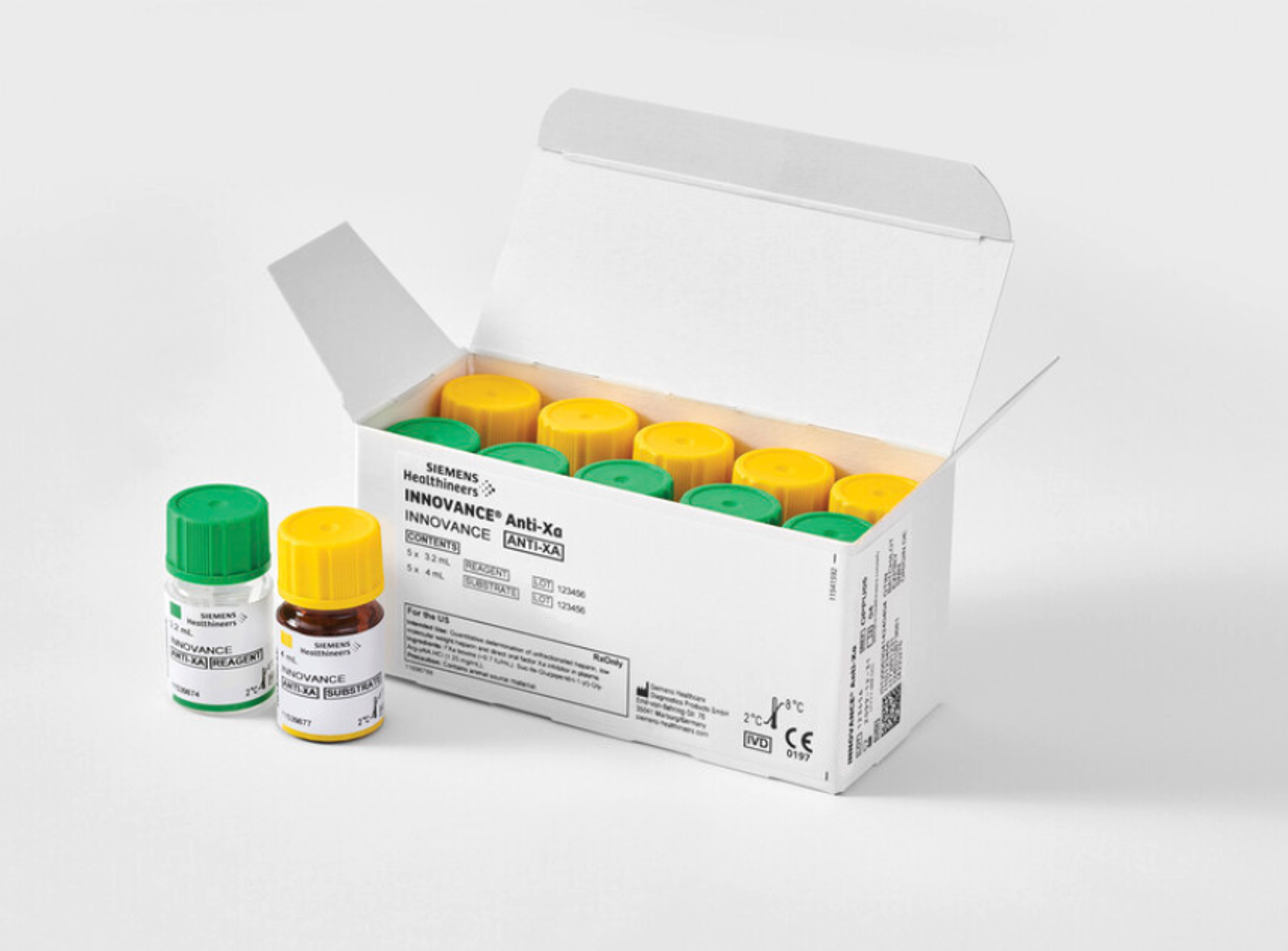
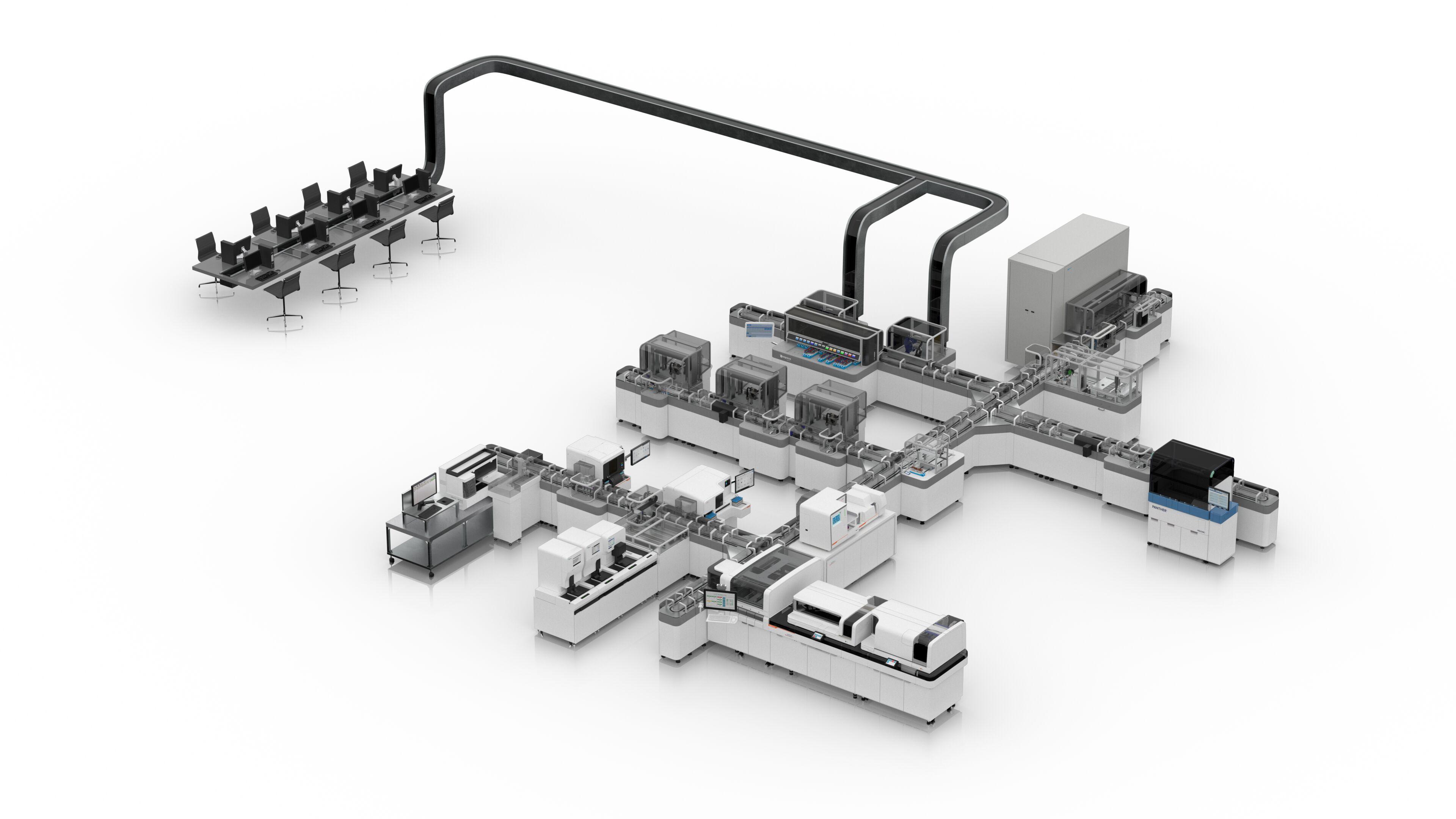
Atellica CI Analyzer
Designed to deliver equivalent results across an entire network, the Atellica CI Analyzer brings the power and possibility of the central lab to spoke and STAT laboratories.
Atellica CI Analyzer brings the power and possibility of the central lab to your entire network, standardizing experiences, and optimizing laboratory space, and maximizing testing throughput for every clinical environment. Not available for sale in the U.S. Product availability varies by country.
Top 5 ways to cope with limited staffing and improve job satisfaction in the clinical lab
A landmark survey of laboratory professionals revealed that 85.3% feel overwhelmed at work as of December 2018. In this case study, Siemens Healthineers discusses the survey results and how technology is helping to solve burnout in the clinical lab.
Optimize Testing to Support the Global Hepatitis Elimination Initiative
The Coalition for Global Hepatitis Elimination (CGHE) is dedicated to accelerating the eradication of hepatitis B and C worldwide. Operating under the Task Force for Global Health, CGHE collaborates with various partners to enhance the capacity of national and sub-national elimination programs.
During this webinar, attendees will learn about the CGHE's objectives, plan, and updated status. Current challenges in testing, including accessibility and accuracy will also be examined.
Key learning objectives
- Identify key challenges and barriers in global hepatitis elimination efforts and explore strategies to overcome them
- Discuss the Coalition for Global Hepatitis Elimination (CGHE)—its mission, programs, and impact on advancing elimination goals
- Explain how improvements in laboratory workflow efficiencies in diagnostics play an essential role in initiatives such as hepatitis elimination
Who should attend?
Healthcare professionals, clinical staff, lab technicians, pathologists, lab directors, laboratorians, researchers, and academics
Accreditation statement
SelectScience® is approved as a provider of continuing education programs in the clinical laboratory sciences by the ASCLS P.A.C.E.® Program. Attendees of these webinars may be entitled to 1.0 ACCENT Continuing Education Credits from the ADLM. This activity was planned in accordance with ACCENT® Standards and Policies. Verification of Participation certificates are provided to registered participants based on completion of the activity, in its entirety, and the activity evaluation. For questions regarding continuing education, please email education@myadlm.org
Precision in Practice: Using Metrics to Elevate Laboratory Quality
WEBINAR SERIES: Quality Data, Informed Action: Metrics and Assay Performance Amplified by Quality Indicators
Part 1: Precision in Practice: Using Metrics to Elevate Laboratory Quality
October 24, 2025 - 15:00 BST, 16:00 CEST, 10:00 EDT, 07:00 PDT
Discover how leveraging available data and tools can significantly enhance your lab's efficiency by reducing unnecessary materials, time, and effort. By gaining a deep understanding of assay performance and making informed decisions about allowable variations, you can streamline workflows and align your quality management with risk-based, patient focused regulatory guidelines.
This session addresses the challenges associated with multianalyzer systems, the critical role of robust QC practices, and the practical application of total allowable error and sigma metrics. You’ll also explore actionable guidelines for lot-to-lot comparisons and learn how to utilize automated tools to streamline and improve QC processes. Apply these theoretical concepts pragmatically to elevate laboratory practices to ensure accurate, reliable test results.
Key Topics:
- Analyzer concordance: Practical strategies for managing multiple analyzers within a single lab or across networks. Learn to set realistic performance expectations and conduct effective intra-lab comparisons to optimize assay consistency.
- Quality control frequency: Understand QC frequency from a risk management perspective. Discover how to select appropriate QC rules and frequencies, and apply standardized rules and targets consistently across analyzers.
- New reagent lot management: Best practices for lot-to-lot comparison. Learn when additional testing on new reagent lots is necessary and when it can be avoided to conserve resources without compromising quality.
We’ll also explore the role of automation in QC processes, including scheduling QC checks based on patient sample volumes and utilizing built-in evaluation tools for assay performance studies. Leveraging these automated features can help your lab stay ahead of evolving best practices and regulatory requirements while improving overall operational efficiency.
Key Learning Objectives:
- Understand the difficulties laboratories face in balancing increasingly stringent regulatory requirements for risk-based quality management with the reality of tightening budgets and resource constraints.
- Recognize how detailed knowledge of assay performance metrics supports the development of quality assurance programs that are sustainable, practical, and aligned with real-world lab operations.
- Learn strategies to effectively manage analyzer concordance, determine optimal QC frequency, and assess new reagent lots by utilizing advanced, built-in analyzer technologies and automated tools.
Who Should Attend?
Healthcare professionals, clinical staff, lab technicians, pathologists, lab directors, quality manager, laboratorians, researchers and academics.
Sharon Bracken summarizes key priorities for Siemens Healthineers at ADLM 2023
Sharon Bracken, President of Diagnostics at Siemens Healthineers, discusses the newly FDA-cleared CI Analyzer that streamlines the hub-and-spoke model for high-volume tests to help meet post-pandemic testing demands. Bracken also discusses an exciting new partnership with Scopio Lab solutions, and the focus of Siemens Healthineers' on advancing automation, consolidation and efficiency in diagnostic testing labs.
This video was filmed at the 2023 ADLM Annual Scientific Meeting and Clinical Lab Expo
Improving laboratory performance with data-driven quality management
In this SelectScience webinar, experts explain how defining total allowable error and applying sigma metrics can guide smarter QC decisions and streamline laboratory workflows
Siemens Healthineers debunks myths around clinical lab automation
Discover how Siemens Healthineers is breaking down barriers and making automation more accessible, efficient, and beneficial for clinical labs of all sizes
Discussing diagnostic trends and innovations after ADLM 2024
Discover how sustainability, efficiency, automation and other trends are impacting diagnostic innovation and improving healthcare outcomes
Clinical Diagnostics Scientists’ Choice Awards winners announced at ADLM 2024
Thermo Fisher Scientific, Beckman Coulter Clinical Diagnostics, and LGC Clinical Diagnostics among those recognized
Siemens Healthineers offers new way to detect heart failure
NT‑proBNPII Assay now part of Atellica cardiac testing menu
Siemens Healthineers launches Atellica CI Analyzer
The new compact testing system is designed to tackle a range of challenges faced by today's laboratories