ultraDAWN™
A breakthrough in process analytical technology for production of nanoparticles, biopharmaceuticals and polymers

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
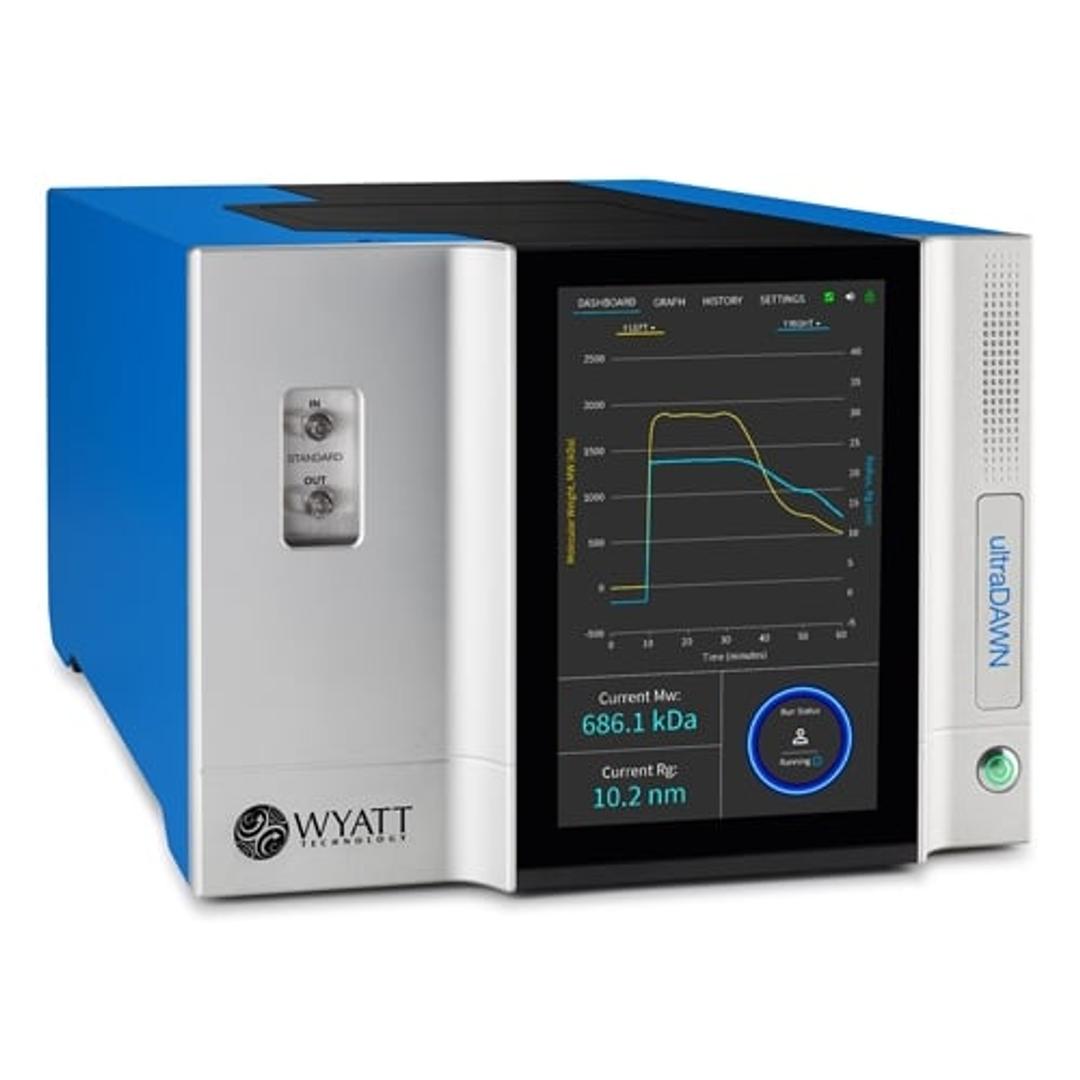
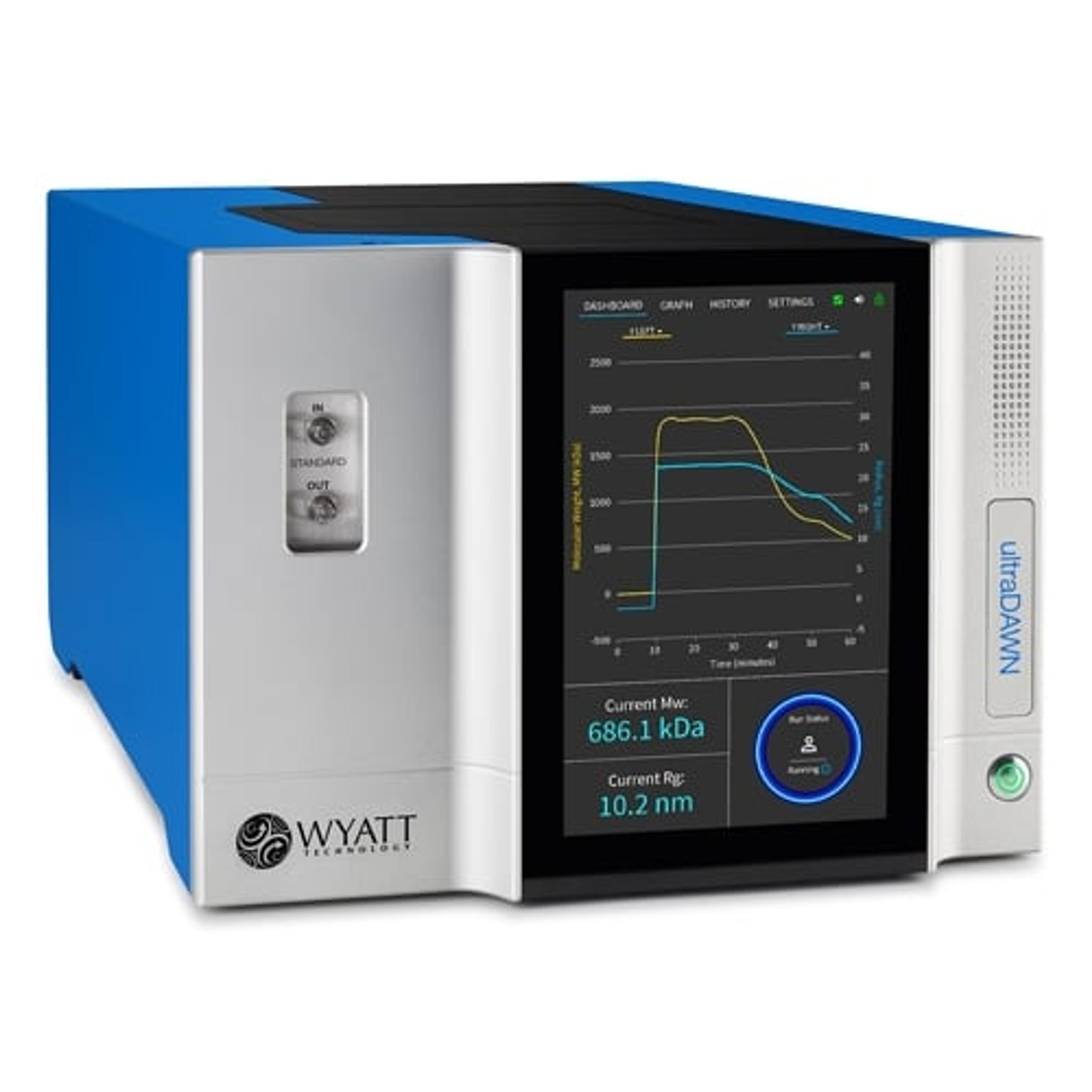
The ultraDAWN is a unique multi-angle light scattering (MALS) instrument, designed for monitoring key attributes of macromolecules and nanoparticles such as proteins, biopolymers, gene vectors and nanodrug-delivery vehicles. With its accompanying OBSERVER™ software, ultraDAWN calculates and reports molar mass, size, particle concentration or payload, in real time, up to 300 times per minute.
- Process development: accelerate PD with immediate feedback on product quality when changing conditions such as flow rates, column loading, resin, buffer conditions or process duration
- Production: monitor and control the process based on direct measurements of product attributes, without invoking process models; obtain immediate data on product quality for confidence in sending material to the next unit operation.
OBSERVER can generate a trigger for controlling the process based on allowed values of a product attribute like particle size or gene vector payload. In addition, the real-time data may be fed back to the process controller for control purposes and saved for post-process analytics.
ultraDAWN determines:
- Molecular weight from 103 to 109 g/mol
- Radius from 10 to 250 nm
- Particle concentration for the above size range
- Full-total capsid ratio (Vg/Cp), full and empty titer and size of small viral vectors like AAV
- Product aggregation via increases in molar mass or size
- Presence of impurities via distinct molecular weight or size ranges
OBSERVER’s trigger functionality can be used to:
- Control collection of desirable product
- Reject out-of-spec material
- Indicate process endpoints or critical alarms
Process integration options:
- In-line operation at 1 - 150 mL/min, with response time delay (RTD) below 1 second
- On-line configuration for higher flow rates or for drawing continuously from a homogenizer or reactor vessel, with typical RTD of 0.5 – 3.0 minutes
- Optional integration with production control software via OPC-UA
Applications:
- Chromatographic purification of biologics like proteins or viruses
- Microfluidic production of nanoparticles
- Ultrafiltration/diafiltration, final formulation and homogenization of biologics or nanoparticles
- Polymerization, depolymerization or conjugation reactions of biopolymers
Real-time product attributes, from lab to plant
Process analytical technologies are a key enabler of quality by design (QbD) and Bioprocess 4.0. Implementation of light-scattering based process analytical technology (PAT), inline and online, shifts the focus of PAT from process parameters to product parameters and hints at a pathway to real-time release. Learn more about multi-angle light scattering as an analytical technology for biopharmaceuticals and nanomedicines, and explore the methods available for utilizing real-time MALS as a PAT tool from process development to scale-up and production.
In-line monitoring of liposome size by RT-MALS
In liposome and lipid nanoparticle (LNP) drug-delivery applications, particle size is a critical quality attribute (CQA) that impacts bioaccessibility, retention time and biodistribution. Current on-line and off-line control methods do not sample the entire batch, and only provide average sizes, potentially hiding pockets of out-of-spec material. In this application note, Wyatt Technology demonstrates how real-time multi-angle light scattering (RT-MALS) can ensure batch consistency through in-line monitoring.
RT-MALS end-point determination of a polysaccharide depolymerization process
Current methods to monitor molar mass during polysaccharide production employ off-line SEC-MALS analysis, during which a single run can take up to 30 minutes. With a typical depolymerization time of about 90 minutes, off-line analytics cannot provide timely feedback on reaching the endpoint. In this application note, Wyatt Technology demonstrates how real-time multi-angle light scattering (RT-MALS) fills this need and ensures that the process ends as soon as the endpoint criterion is achieved.
Leveraging light scattering techniques for enhanced biotherapeutic characterization and development
Prof. Dr. Hristo L. Svilenov, Professor of Biopharmaceutical Technology at Ghent University, and Dr. Lorenzo Gentiluomo, Senior Sales Account Manager at Waters/Wyatt Technology, will present several case studies demonstrating the use of various light scattering techniques for the advanced characterization of biopharmaceutical samples. They will focus on dynamic light scattering (DLS), size-exclusion chromatography coupled to multi-angle light scattering (SEC-MALS) and real-time multi-angle light scattering (RT-MALS).
Key learning objectives
- Learn the basics of light scattering techniques
- Discover how to effectively distinguish between different light scattering methods, including DLS, SLS, SEC-MALS and RT-MALS
- Understand how different light scattering can be used to characterize therapeutic proteins
Who should attend?
Scientists interested in the analytical characterization of biopharmaceuticals using light scattering techniques.
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
No more waiting: Real-time quality measurements are reducing development timelines in gene therapy and vaccine production
Learn how the new real-time multi-angle light scattering (RT-MALS) technology continuously monitors and measures critical quality attributes during viral vector production