Steroid Panel LC-MS kit
One single kit to quickly and reliably determine 18 different and clinically relevant steroids with only one extraction process.
Benefits of the Steroid Panel LC-MS kit:
LC-MS/MS is considered the gold standard in the determination of steroid hormones due to its high sensitivity
- Steroid kit, CE IVDD
- 18 steroids
- 2 chromatographic runs (guarantees the best possible performance of all analytes)
- Only one sample preparation with 96 well SPE plate
- All standards in one mix
- Individual isotopic standard for each analyte
- Total runtime (LC MS): 10 min for 15 steroids, second injection for another 3 analytes in 6 min runtime
Multi-steroid profiles measured by LC-MS/MS
Modern laboratories face growing pressure to do more with less. The Tecan Steroid Panel LC-MS solution, validated by Labor Berlin – Charité Vivantes GmbH, enables comprehensive steroid profiling from as little as 100 μL of serum – without compromising accuracy or depth of analysis.
This modified protocol not only expands testing capabilities to samples with minimal volume but also reduces costs and turnaround time – without sacrificing quality.
Key takeaways:
- Ultra-low sample volume
- High analytical performance
- Analysis in 10 minutes
- Ready-to-use, reliable kit
*Disclaimer: The case study was prepared using the Research Use Only Steroid Panel LC-MS Kit (30220266). Consult Product IFU for the proper instructions.
Making sense of IVDR for LC-MS
In this whitepaper, Tecan provides information on how to interpret and prepare for the In Vitro Diagnostic Regulation (IVDR) on laboratory developed tests (LDTs) in mass spectrometry (MS). The IVDR is Europe’s set of rules designed to improve the quality, safety, and reliability of in vitro diagnostics (IVDs). Although the IVDR is largely targeted at manufacturers, this regulation also puts a higher burden on clinical labs, especially those doing liquid chromatography–mass spectrometry (LC-MS), because it has implications for the use of LDTs, in-house devices or ‘home-brew’ methods. Even if the tests are being run in non-EU labs, the IVDR may still apply if the samples being tested are from European citizens.
Evaluating Tecan's Steroid Panel LC-MS kit for endocrine diagnostics
Steroid hormones are strongly associated with endocrine disorders like adrenal insufficiency, Cushing’s syndrome, and congenital adrenal hyperplasia. Steroid testing has become an integral part of diagnostic procedures and is recommended by many clinical practice guidelines.
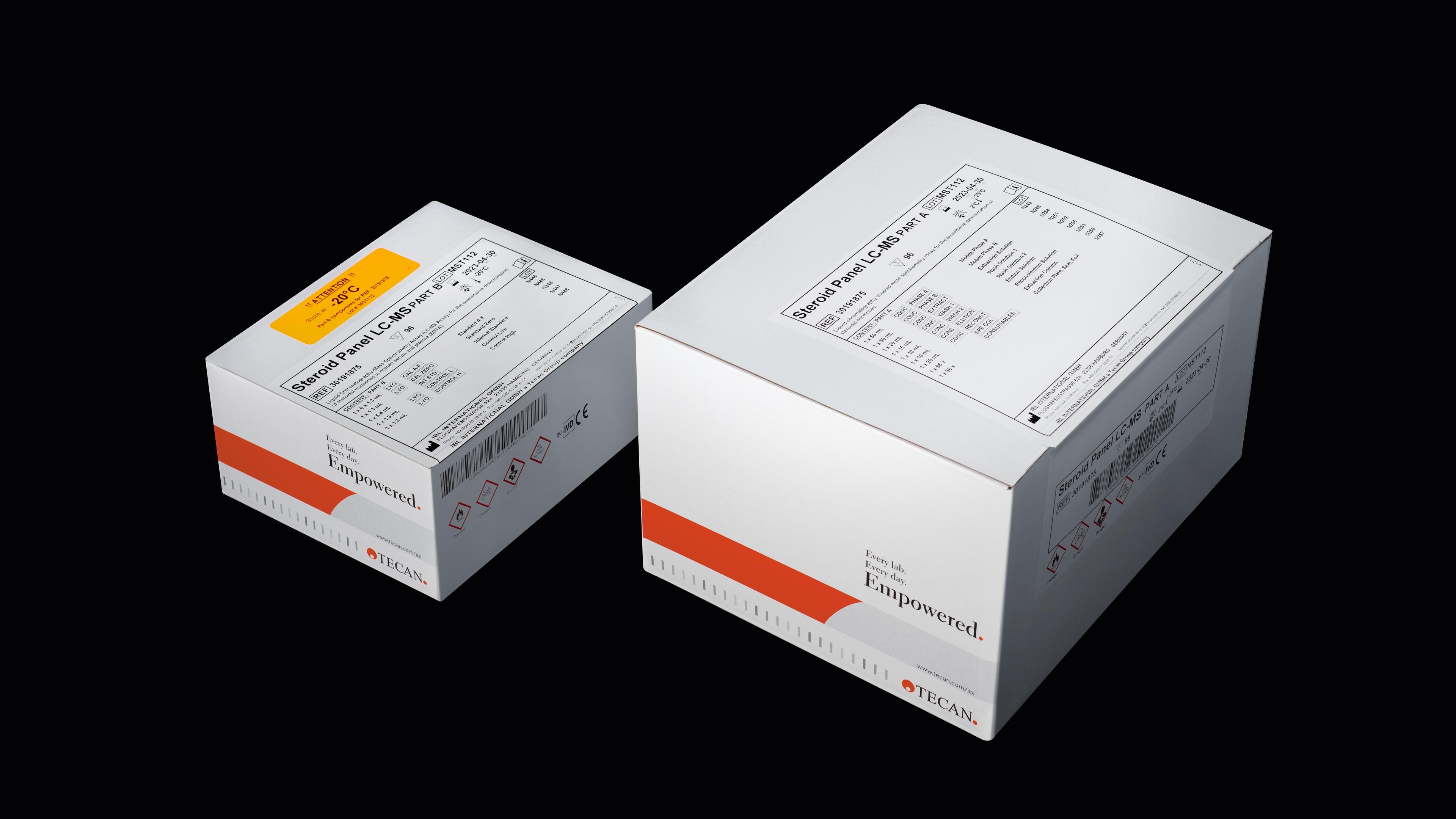
A comparative study by the Children’s Hospital in Erlangen evaluated their in-house method against the commercially available Tecan CE-IVDD Steroid Panel LC-MS kit (Ref# 30191875*), confirming the precision and reliability of the latter.
The kit’s capacity to measure 17 steroid hormones and dexamethasone, including DHT and 17-OH-Pregnenolone, expands its diagnostic utility, providing deeper insights into patient hormone levels and improving clinical decision-making.
Disclaimers:
*Product availability and regulatory status may vary across regions outside the EU depending on local country specific registration. Consult with your Tecan associate for further information.
In the US: For research use only, not for use in diagnostic procedures.
Evaluation of a Steroid Panel LC-MS Kit
LC-MS is recommended for measuring circulating steroids, however, there is nearly no commercial solution available nor an external quality assessment scheme (EQAS) available for all clinically relevant steroids. Tecan highlights a study which aims to compare the results of the laboratory-specific procedure to Tecan's commercially available kit for the measurement of 17 circulating steroids and dexamethasone. Additionally, an evaluation of the clinical bias of the mentioned kit by using external quality control samples as a reference was performed. Since no reference measurements and/or ring trial samples are available for all steroids included in the commercial kit, the results of the serum samples are set in a clinical context afterwards.
Eliminate bottlenecks in manual sample preparation with Tecan's Steroid Panel LC-MS kit and Resolvex A200
In this video, explore the capabilities of Tecan's Steroid Panel LC-MS kit and Resolvex®200 and discover how they can be used in combination to cover the entire sample preparation and steroid measurement workflow.
Disclaimer: *Product and solution may not be available in all territories. Please consult your local Tecan office for more information.
The novelty in Tecan's reagents portfolio
One single kit to quickly and reliably determine 18 different and clinically relevant steroids with only one extraction process.