Impurity Profiling Suite
Predict Genotoxic & Carcinogenic Endpoints to Meet ICH M7 Guidelines
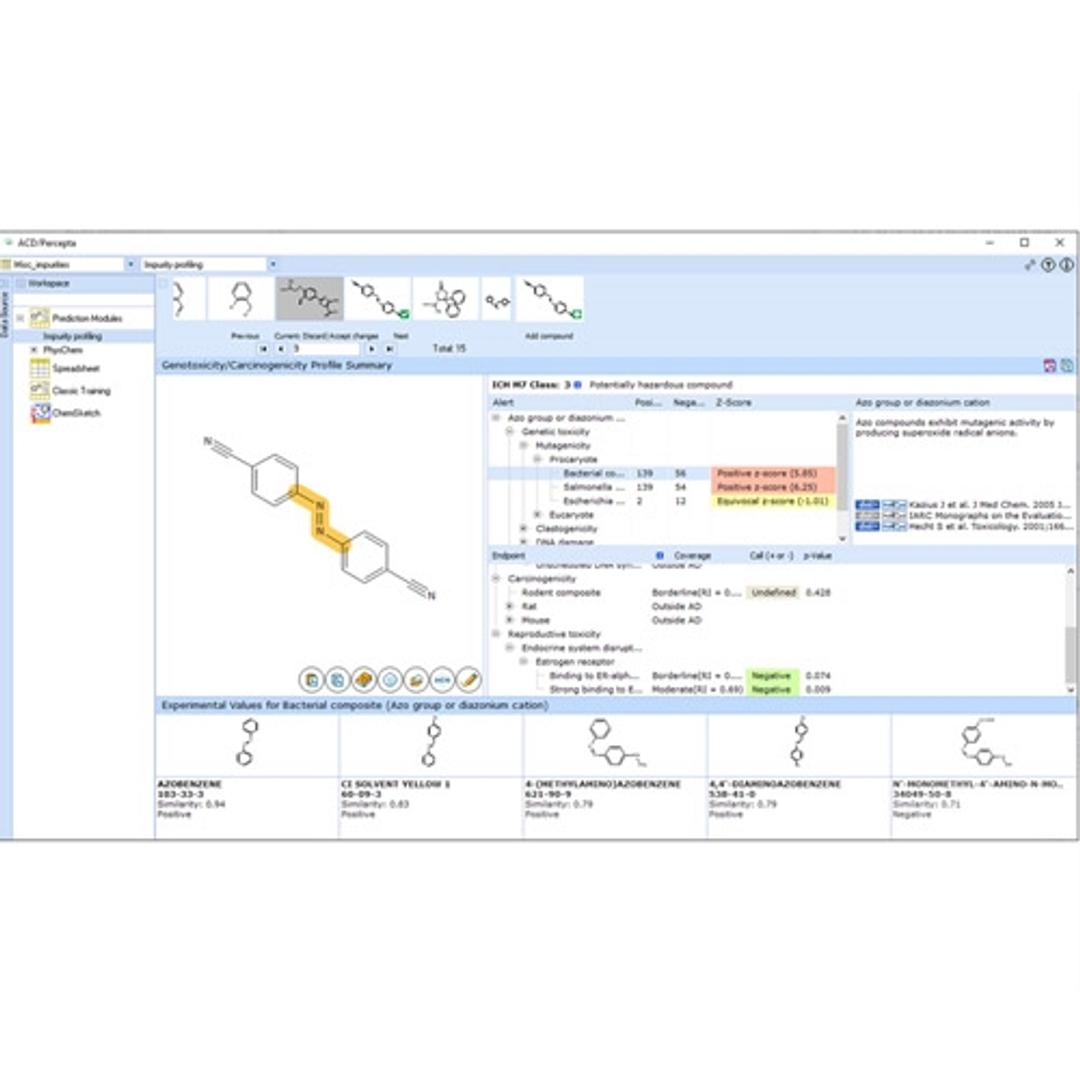
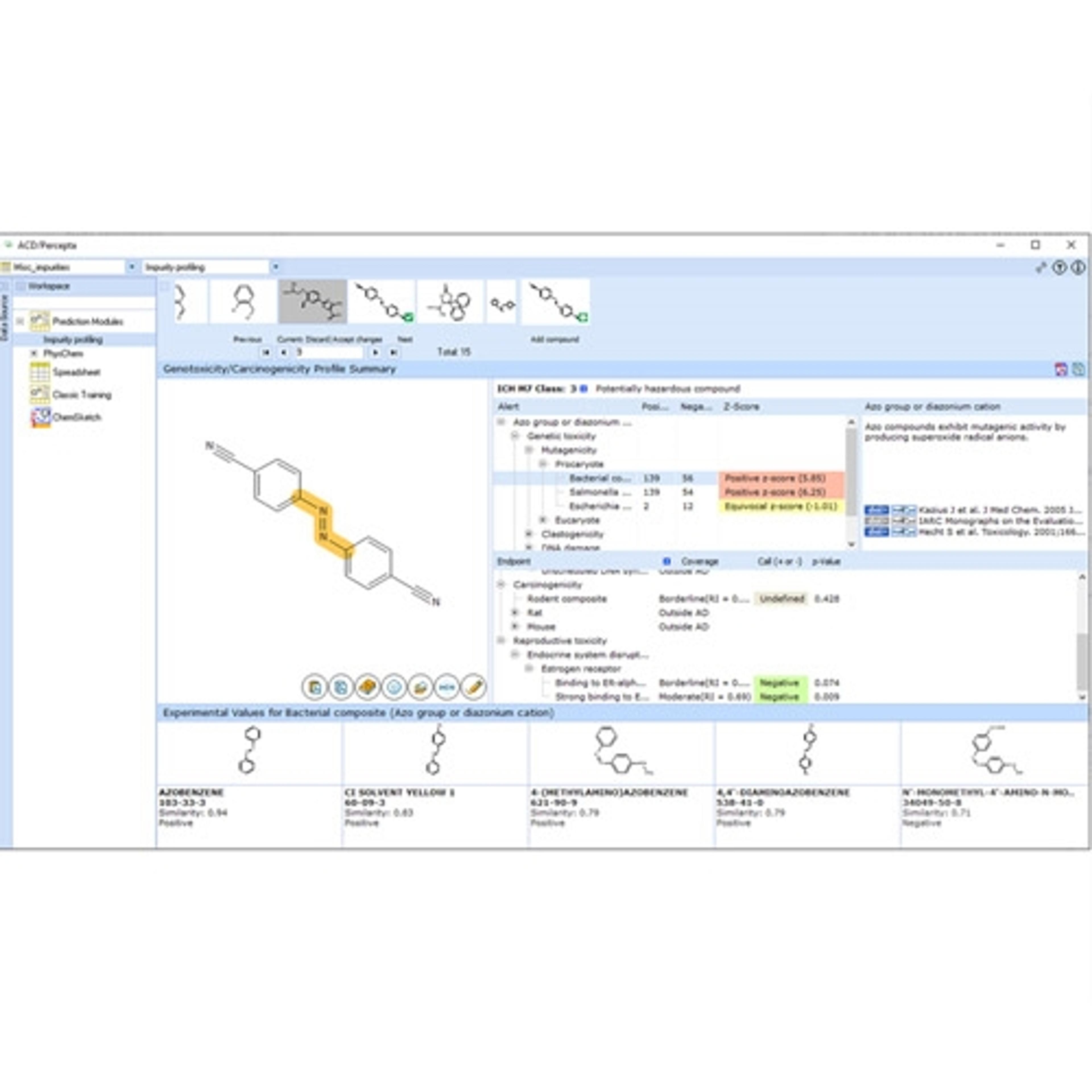
ACD/Impurity Profiling Suite predicts a variety of toxicological endpoints to help you assess the genotoxic and carcinogenic potential of impurities and degradants.
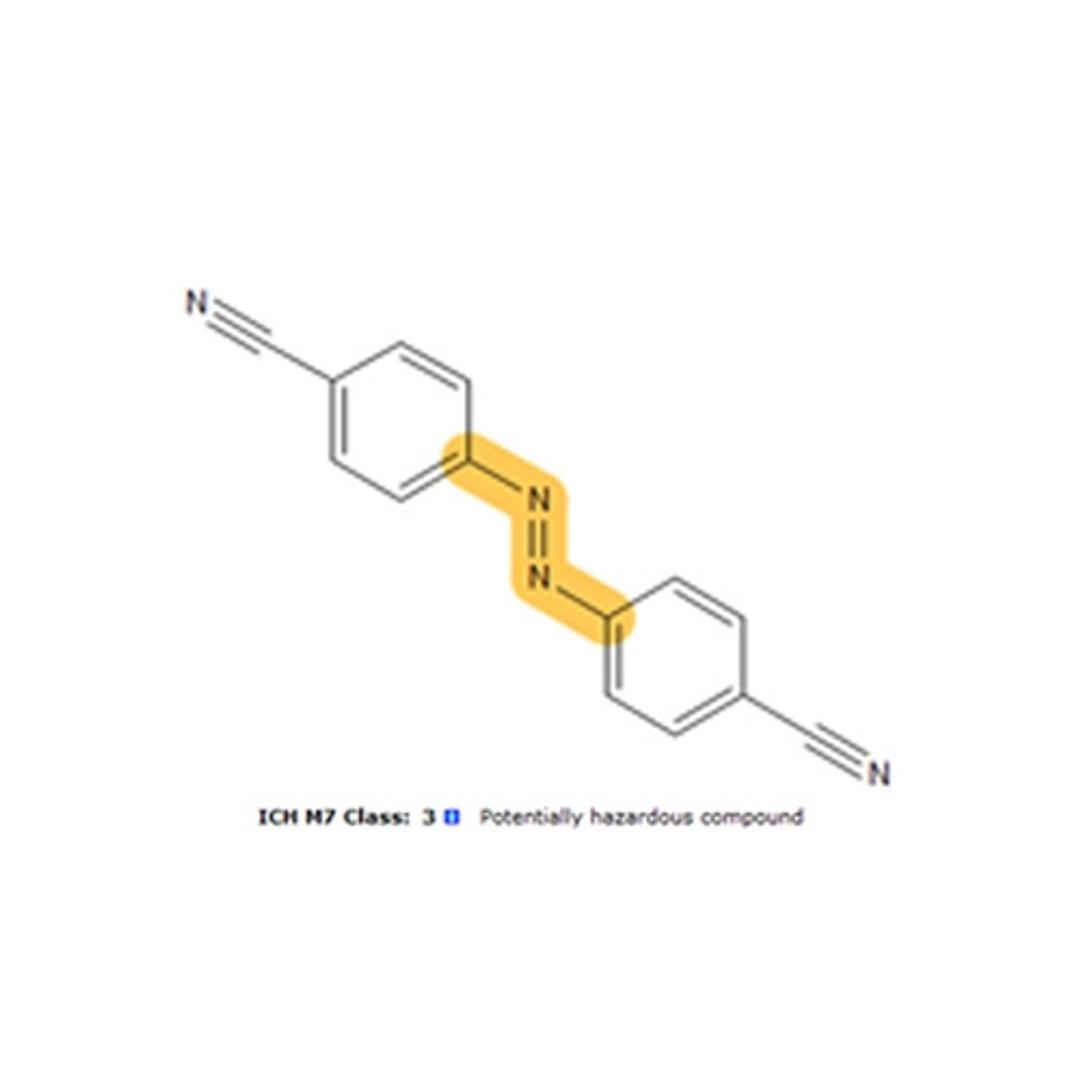
- Determine the ICH M7 classification for impurities and degradants
- Predict 21 toxicological endpoints from structure; for mechanisms of hazardous activity including:
- Mutagenicity (AMES test and other procaryote and eucaryote test systems)
- Clastogenicity
- Other DNA damage
- Carcinogenicity
- Endocrine disruption mechanisms
- Identify potentially hazardous structural fragments responsible for carcinogenic and genotoxic activity
- Gain insight into the possible mechanisms of toxic effects
Brochures
Percepta flyer
In this flyer, ACD/Labs demonstrates the Percepta™ Platform, which offers a portfolio of software suites to predict and manage physicochemical, ADME, toxicity, and other molecular property data.
Impurity Profiling Suite: Decision-support for ICH M7 workflows & regulatory submissions
In this flyer, ACD/Labs demonstrates how the Impurity Profiling Suite™, developed through a collaborative agreement with the US FDA, will help you determine the safety risk of impurities and degradants, and gain insights into the acceptable levels of exposure.