Luminata
Enterprise decision support software for pharmaceutical and chemical development. The only software that consolidates your process and analytical data in one place.
The program is very useful.
Pharmaceuticals, stress testing
We use luminata software. the program is very useful.
Review Date: 12 Mar 2020 | Advanced Chemistry Development, Inc., (ACD/Labs)
Easy to use, great support.
Pharmaceuticals, stress testing
Easy to use, great support.
Review Date: 12 Mar 2020 | Advanced Chemistry Development, Inc., (ACD/Labs)
Let Scientists Focus On Science
The future of science is collaborative. Cutting-edge pharmaceutical and chemical development involves sharing information within a multidisciplinary team who track many types of analytical and process data.
Your data should be useful. More sources of data means more spreadsheets. More incompatible files. More time spent tracking down results. Before long, data management gets in the way of research and decision-making.
Make your data work for you with Luminata. Luminata® is the only Chemistry, Manufacturing, and Controls (CMC) decision support software. It lets your team store, search, map, process, and reuse all your chemical and process data in one application.
The Data Management Solution for Your Entire Team
Analytical Chemistry
• Consolidate analytical data in one application that supports >150 data formats
• Connect analytical and process data
• Share analytical results with colleagues in real-time
Process Development and Control
• Improve reaction planning with process maps
• Simplify impurity management
• Build effective process control strategies
Team Leads and Management
• Track project progress across locations
• Assess batch data throughout the supply chain
• Accelerate regulatory submissions with report building tools
Route Scouting & Process Development
Consolidate all your process development information in one location. Connect analytical data with route schema to ensure a complete understanding of multi-stage process routes.
• Build comprehensive maps for your evolving process
• Link analytical data to each chemical in your scheme
• Integrate Quality by Design (QbD) principles in route selection
• Design a process map by importing LC/UV/MS datasets to drive creation of new entries, or select existing compounds from the onboard reference dictionary
• Categorize entries in the process scheme as Starting Materials, Intermediates, API(s), Impurities, Degradants, Solvents, or Reagents
Impurity Control
Establish effective process and analytical impurity control strategies based on live spectral data and route knowledge, in an environment built for Quality by Design (QbD) in pharmaceutical drug development.
• Use an impurity map to automatically track fate and purge data
• Auto-calculate impurity carryover from analytical results
• Develop robust controls based on all the relevant data in one place
• Build control strategies through the impurity master mode functionality
• Perform fate & purge, and spiked impurity calculations
Drug Substance & Drug Product Stability
Assemble degradation maps that cover complete drug substance and drug product forced degradation studies.
• Import and process forced degradation HPLC and MS datasets for a complete study map
• Make formulation decisions based on kinetic plots automatically created from analytical data
• Search for target impurities across forced degradation experiments
• Automatically process time-point datasets to create a forced degradant map and kinetic plot(s) that capture all forced degradant information in one location
Formulation Development
Luminata’s drug formulation tools accelerate the transition from drug substance to drug product by integrating analytical and process data in one place.
• Develop design protocols based on the chemistry of the API / Drug Product formulation
• Build comprehensive formulation maps
• Perform drug-excipient comparability studies and test formulations
• Perform excipient case studies for preformulation studies
Supply Chain Analytical Data Management
Use Luminata’s supply chain data management tools to attach analytical data to each batch for real-time decision-making and batch genealogy tracking.
• Create a comprehensive family tree of every batch for a complete supply chain map
• Identify irregularities by comparing analytical data to a reference
• Use structure search to quickly determine the presence of an impurity/entity in all batches
• Do batch-to-batch comparison with analytical data
• Register and integrate all batches created internally and externally
Data Import and Analysis
Luminata is built on the Spectrus platform, allowing you to import, process, and store your analytical data.
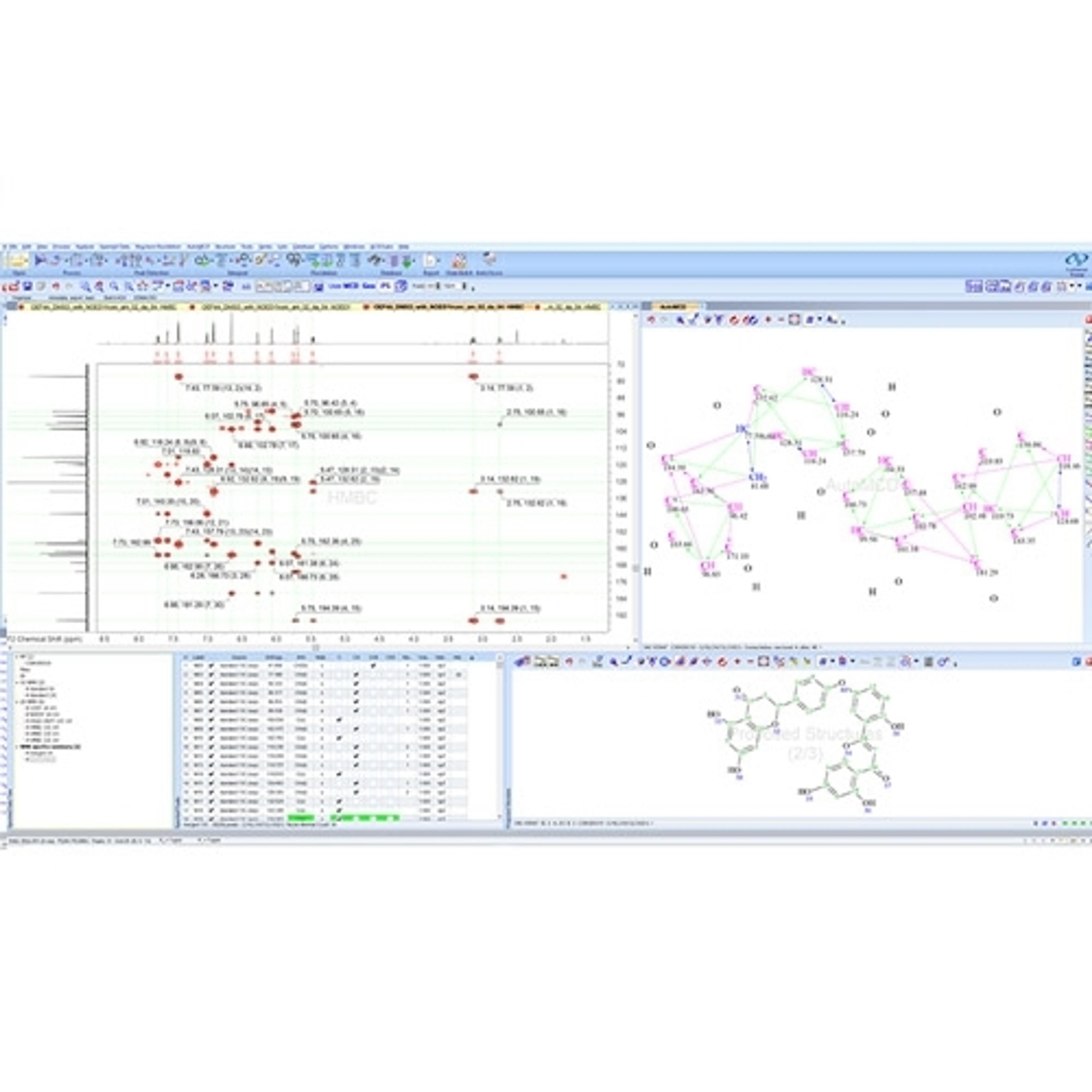
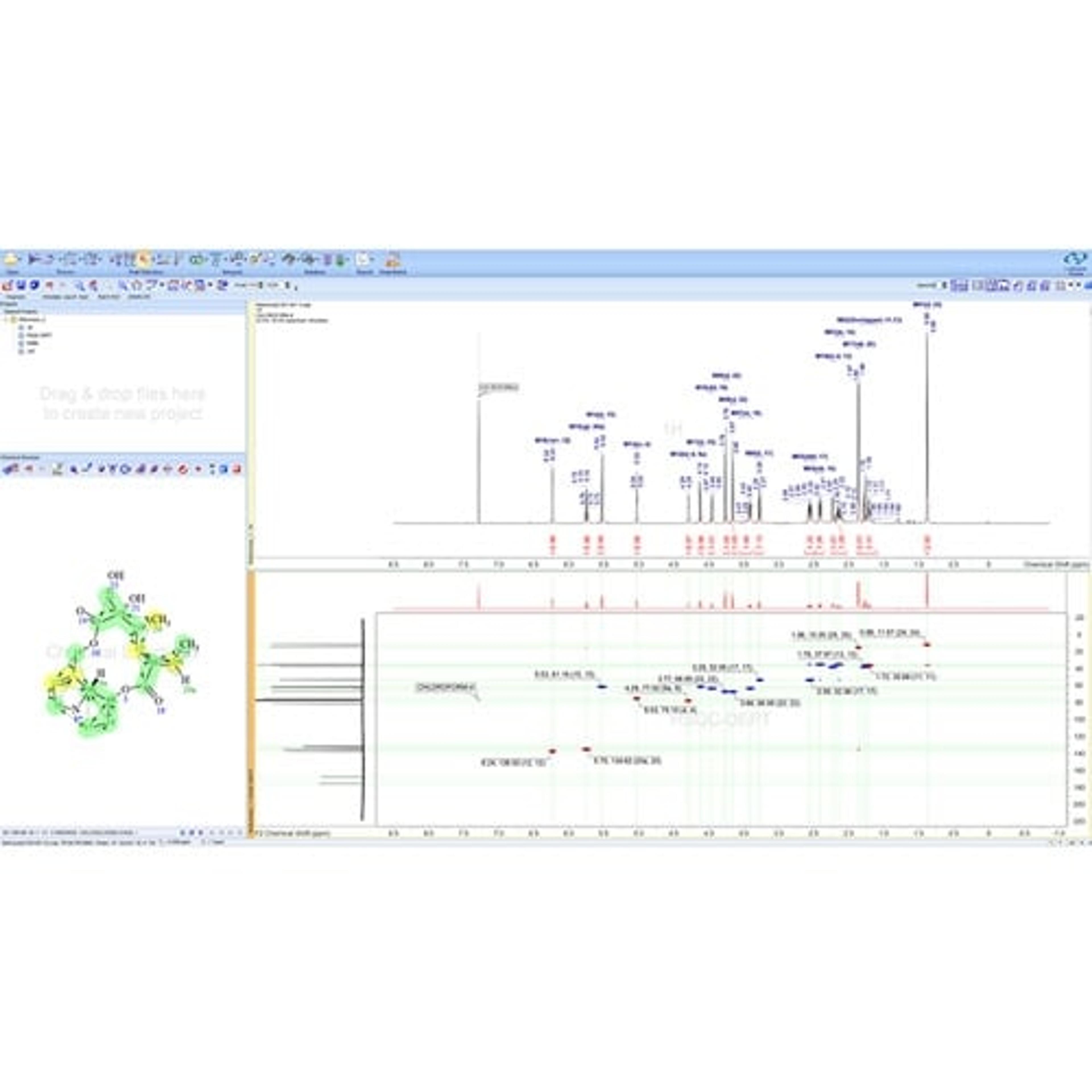
• A single data format for the analytical techniques used to characterize compounds and formulations in your labs. LC/MS, GC/MS, NMR, Chromatography, Raman, IR, UV, Visible, and more
• Native support for >150 data formats including major instrument vendors, open-source formats, and emerging standards (e.g., AnIML, JCAMP, and ADF from Allotrope).
• Luminata will detect the type of data you’re importing and offer tools to process and analyze it.
• Connect and assign full chemical and biochemical structures, structure fragments, Markush structures, or atoms to spectra and chromatograms.
Brochures
Luminata flyer
In this flyer, ACD/Labs showcases Luminata™, designed to bring together all your development data in one interface.
Accelerating process chemistry: How data management is enhancing pharmaceutical development
In this eBook, ACD/Labs explores how process chemists can increase their efficiency with the support of Luminata, which allows research teams to access process, analytical, and chemical data from across their organization.
Accelerated stability assessment with Luminata
In this application note, ACD/Labs outlines challenges in accelerated stability assessment studies, and demonstrates how the Luminata® can help you to address these challenges.
Ensuring insights from data: A view beyond ELNs, LIMS, and SDMS
In this eBook, ACD/Labs highlights major topics associated with analytical data management, including the risk and benefits associated with automating your analytical data management.
Real results from bridging gaps in data management
In this application note, ACD/Labs describes how a major biopharmaceutical company improved process development and regulatory submission efficiencies.
Enhancing impurity control with data management
Standards for pharmaceutical quality are constantly rising. Regulators expect pharmaceutical development teams to demonstrate in-depth knowledge of their chemical processes. Unfortunately, scientists must devote significant time and resources to handling their data, which interferes with effective decision-making. In this application eBook learn how Luminata software consolidates all your pharmaceutical development data in one interface. It is built on a chemically intelligent platform, allowing researchers to access and visualize analytical, chemical, and in silico data from across their team.
Luminata and forced degradation
Forced degradation is one of the essential functions of pharmaceutical development, providing insights into the stability of the drug substance and drug product, as well as identifying degradants that may pose a health risk. These studies often require teams spread across multiple sites using large amounts of data. Managing and sharing this data is challenging due to the number of sources and the variety of incompatible data types. Scientists spend more time handling their data and less time performing experiments. In this application eBook, explore Luminata, a software that consolidates all your pharmaceutical development data in one interface. Built on a chemically intelligent platform, this software allows researchers to access and visualize analytical, chemical, and in silico data from across their team.
Learn about software for impurity data management
In this video, ACD Labs introduces the Luminata, designed to cut your investment in impurity data from months to minutes.
7 essential resources in pharmaceutical quality assurance
Explore the latest technologies, free downloads, and expert insights that can help your laboratory achieve compliance successfully
ACD/Labs Releases Updates to its Foundational Spectrus and Percepta Platforms
The release of ACD/Labs’ v2018.1 software includes major enhancements to its analytical knowledge management solutions for pharmaceutical development, MS and NMR dereplication workflows, and chromatography offerings
9 Top Technology News Stories from ASMS 2018
Updates to Spectrus Informatics Platform Announced
V2017.1 release of ACD/Spectrus software introduces an innovative solution for impurity data management and exciting developments for deconvolution and analysis of mixtures