Ensuring reliable patient results: Why automated liquid handlers need volume verification
Learn how the Technology Development department at Guardant Health upholds the reliability of cancer assays that directly impact patients’ lives
25 Sept 2022

Patient samples are considered highly valuable, even if blood samples are drawn as part of routine medical check-ups. Clinical assays used for disease screening or diagnosis require precision to generate reliable, accurate results with these limited sample volumes. Additionally, when patient data is used for future diagnostic or therapeutic applications, a high level of assay-to-assay reproducibility will need to be maintained to identify trends.
Internally, these assays are subjected to rigorous quality control measures. Instruments used to dispense reagents and detectors used to quantify results are regularly calibrated to monitor accuracy. Equally, the reagents, buffers, and final readouts of an assay, too, undergo regular quality checks. In this SelectScience® article, we speak with Jessica Rakijas, Ph.D., a scientist in the Technology Development department of Guardant Health, who shares how her team upholds the reliability of cancer assays that directly impact patients’ lives – and why even large, sophisticated liquid handlers are not exempt from quality control.
When it comes to clinical assays, every microliter matters
“Our goal is to be as sure as possible that everything that is going to touch a patient’s sample is working reliably,” says Rakijas. “My team members design quality control assays, create SOPs, set acceptance criteria, and even write software that assigns a ‘pass’ or ‘fail’ output for the operators. When issues arise, we perform a root-cause analysis. In general, we’re continually improving the way we analyze patient samples through longitudinal monitoring of our performance over time.”
Clinical assay outputs carry immense significance for a patient. A single data point can influence the medical decisions being made downstream. For instance, at Guardant Health, blood tests are offered for all stages of cancer: for cancer screening to catch early-stage cancers, to explore treatment options at early and advanced stages, and to detect potential cancer recurrence in the post-treatment phase.
“Clinical test results are a life-or-death situation for the patient. These numbers can provide hope or hearthache. So, we need to be sure that we’re capturing data with utmost accuracy,” explains Rakijas. “For example, when cancer is shedding DNA into the system, there are very few circulating free DNA molecules floating in blood. To add to that, there is inevitable molecule loss in the reagent pipeline. This means we need to have robust checkpoints and algorithms in place to catch mutations or biomarkers in our patient samples despite the low amount of inputs.”
Viewing automation as an indispensable asset (and not a job-stealing robot)
Unlike academia, clinical labs process a significantly higher daily throughput of samples. Additionally, when the stakes are high and time is of the essence, there is no room for fatigue or inadvertent manual errors as they can have a direct impact on the patient’s medical journey.
As such, automation systems and liquid handlers are considered valued clinical lab partners. Rakijas adds: “I come from an academic background, and this is my first industry position. The sheer volume of liquids we handle at the company is just untenable for an individual human. I could use a multi-channel pipette all day but at some point, I’d be fatigued because I’m only human. Liquid handlers are really a necessity for any large company with clinical workflows.”
Having heard the sentiment, ‘robots are replacing our jobs’ in the past, Rakijas now sees automation as a huge asset. “The robots free up my time to do other things like thinking or designing new experiments – tasks that robots can't do!”
Additionally, automation helps keep experimental protocols consistent no matter the user’s experience, the time of day, or the number of samples. This makes it possible to compare data across different patient cohorts and mine the numbers for interesting patterns. But in the same way as every reagent and assay goes through a quality control process, so do the liquid handlers. A 2-microliter pipetting error, for instance, may not largely affect a high-volume biochemistry experiment, but when dealing with 20 microliters of a patient’s sample, that would amount to a 10% error. To avoid these issues, even the automation systems need to be checked and calibrated.

Double-checking your liquid handlers: Can you trust the volume being pipetted?
Codes written by process engineers make automation systems repeat the same action 10–15 times to confirm repeatability and reproducibility. “If there’s residual volume from an alcohol wash, it can contaminate the downstream products and invalidate the assay,” notes Rakijas. “That’s why we perform volume verification on all our liquid handlers.”
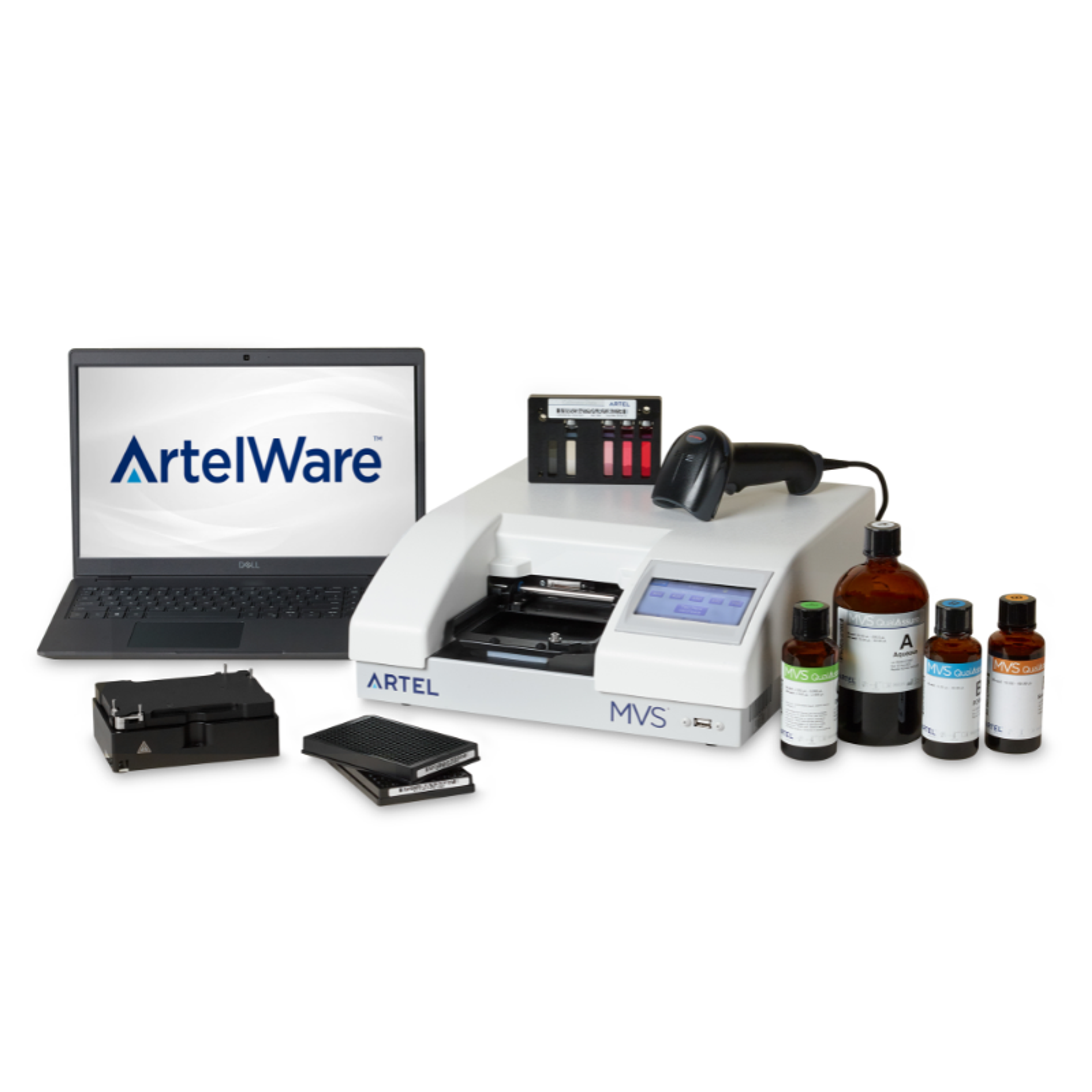
For this, the team uses Artel’s MVS® Multichannel Verification System that enables rapid calibration, verification, and optimization of dispense volumes with high precision and accuracy in liquid handlers.
“The Artel MVS is a series of liquid dyes that are manufactured at a specific concentration,” Rakijas explains. “We simply dispense these dyes into a plate using our liquid handlers and put it in a spectrophotometer. It reads the concentration of the dye and automatically calculates that into the volume that was actually pipetted. It's really amazing.”

Both low-volume and high-volume tests are performed using the Artel volume verification system and an aqueous liquid class, though other liquid class testing can also be done . “The Artel MVS is going to become an essential part of our weekly qualification for instruments. If the pipetting is off, we need to take that liquid handler out of line, and revisit its calibration,” notes Rakijas. “Thankfully, volume verification is really easy. We basically just pour the liquid dyes into troughs and the code triggers the pipetting step. The entire system is self-contained – it provides a beautiful output and creates a heat map.”
Why every clinical lab needs a volume verification system
“We need to care about the status of our robots. They're not people, but they're still an important part of our workflow,” says Rakijas. As the Guardant Health team is scaling up manufacturing lines as well as adding clinical lines for new cancer tests, current efforts are towards streamlining the volume verification over multiple liquid handlers.
“We now have over 100 liquid handlers in our company. So, I’m going to be creating a monitoring dashboard where any instrument’s performance and verification details would pop up with a few simple clicks,” shares Rakijas. “This way, if we notice that an instrument is trending towards failure, we can examine it before problems arise. I believe in the philosophy that an ounce of prevention is worth a pound of cure.”
Learn more about the Artel MVS Multichannel Verification System >>