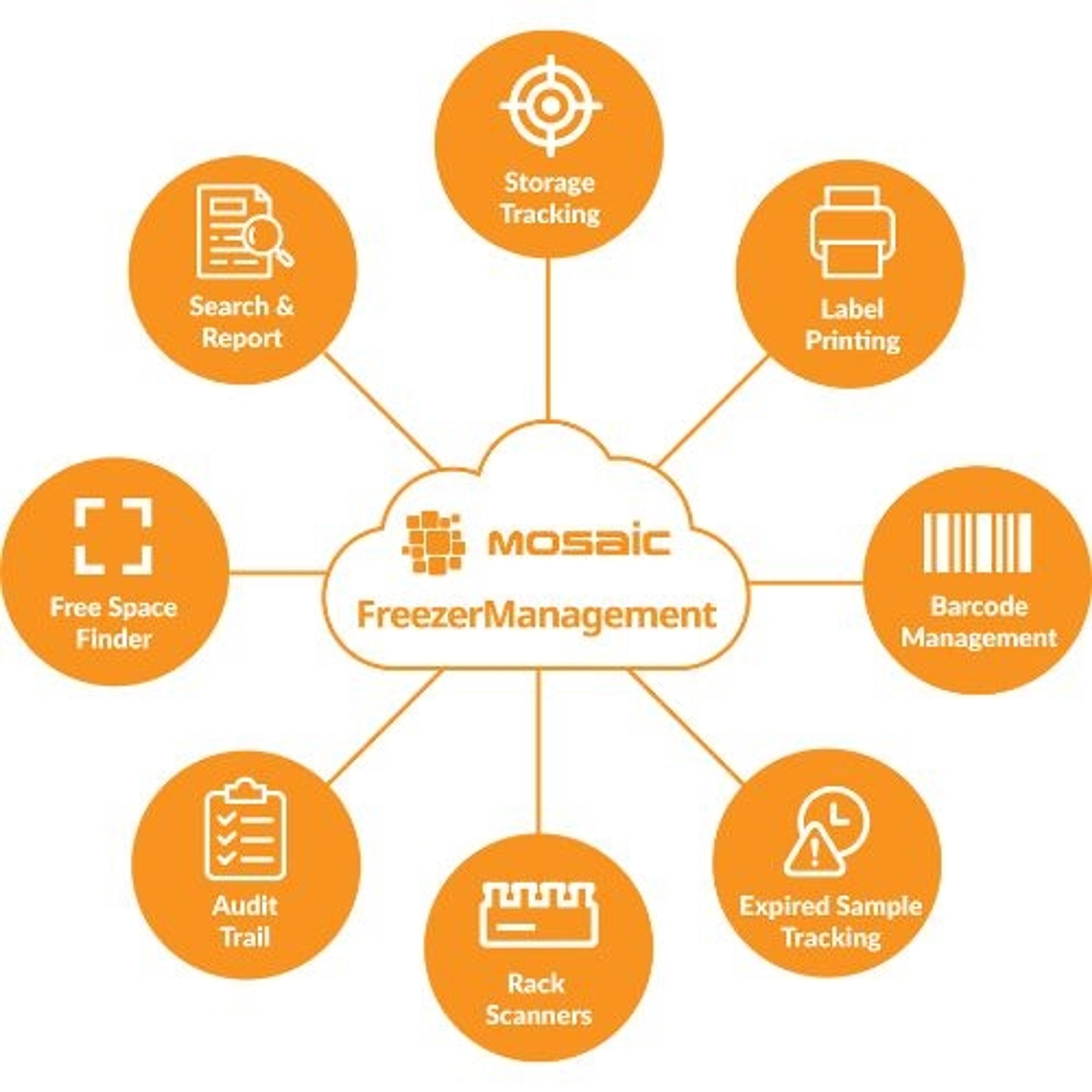
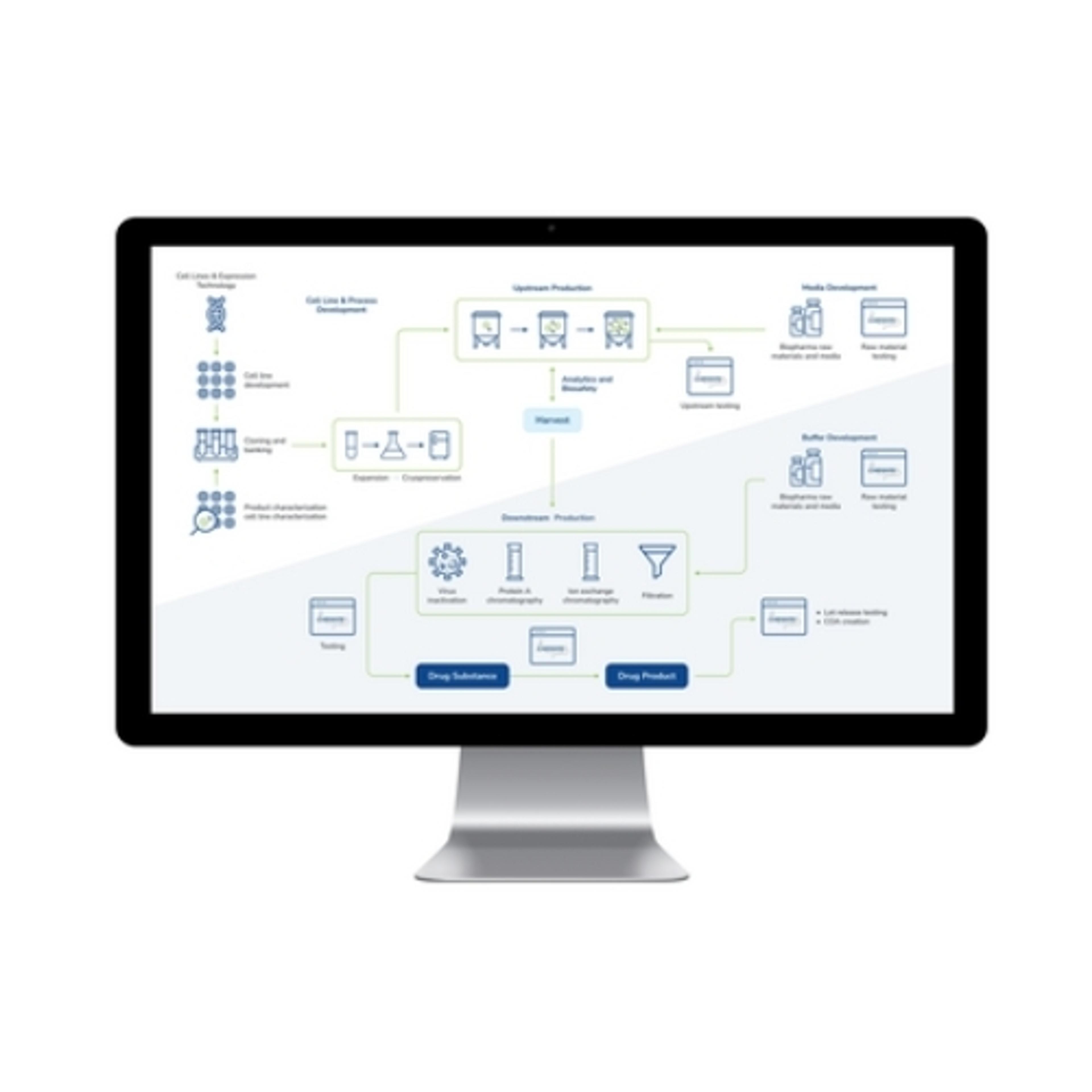
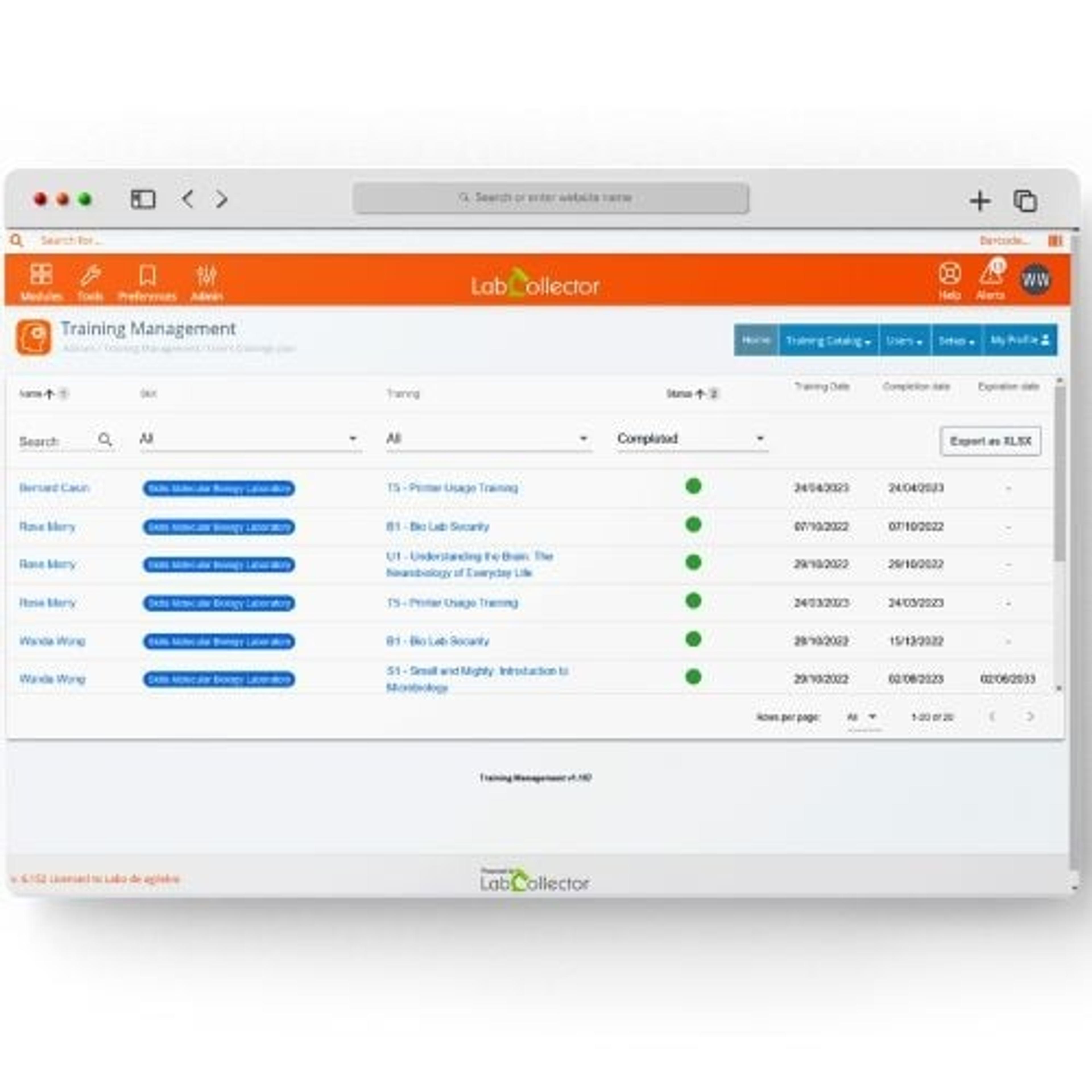
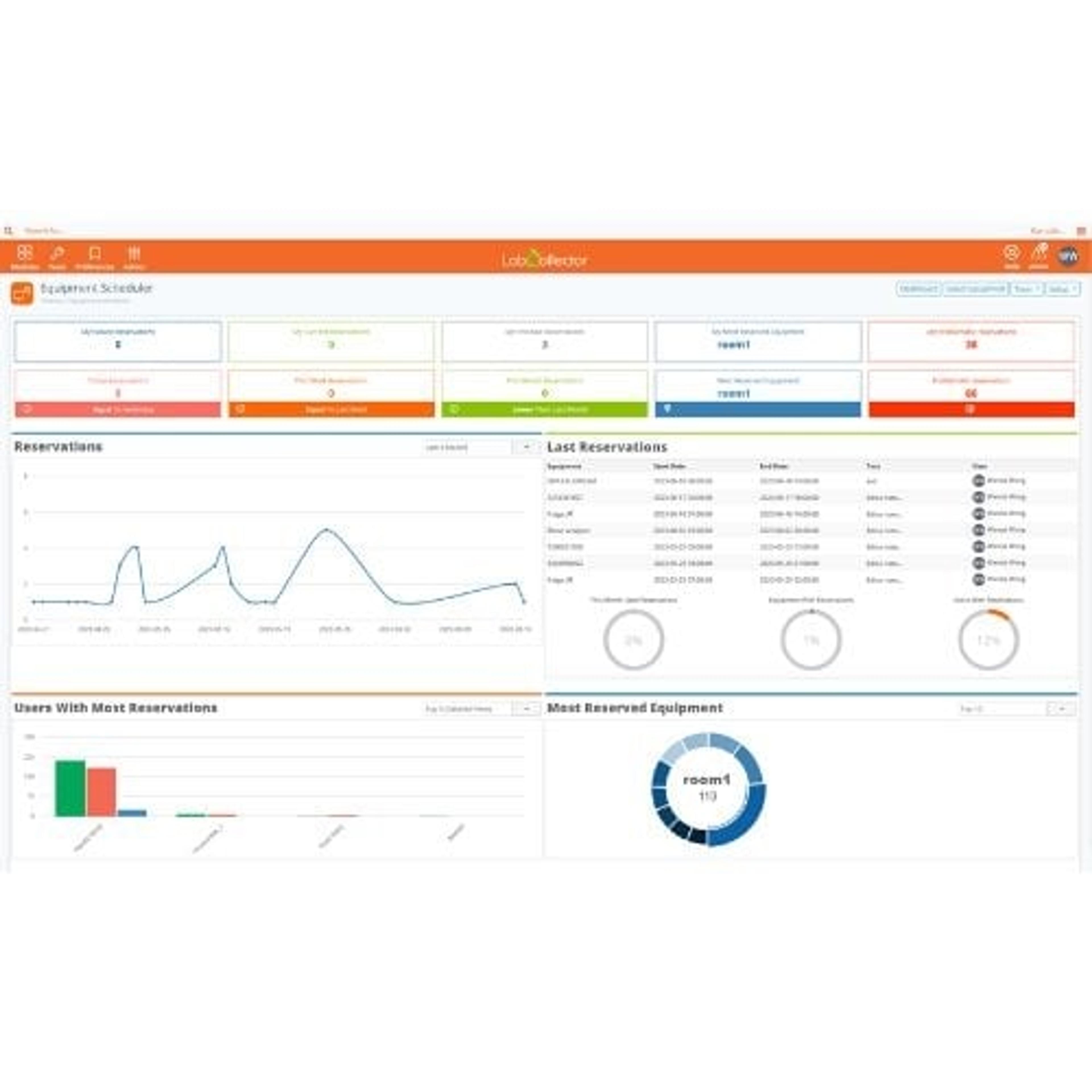
2.2mL, 96 square well 'V' Bottom (2.2S96-008V)
Irish Life SciencesIrish Life Sciences 2.2mL square well 'V' bottom plate has a total volume of 2.27mL. The top surface is particularly flat providing good heat and adhesive seal closure for long term storage. The plate is manufactured in Irish Life Sciences’ class 8 cleanrooms using a medical-grade polypropylene and conforms to the footprint dimensions defined by SLAS/ANSI standards.