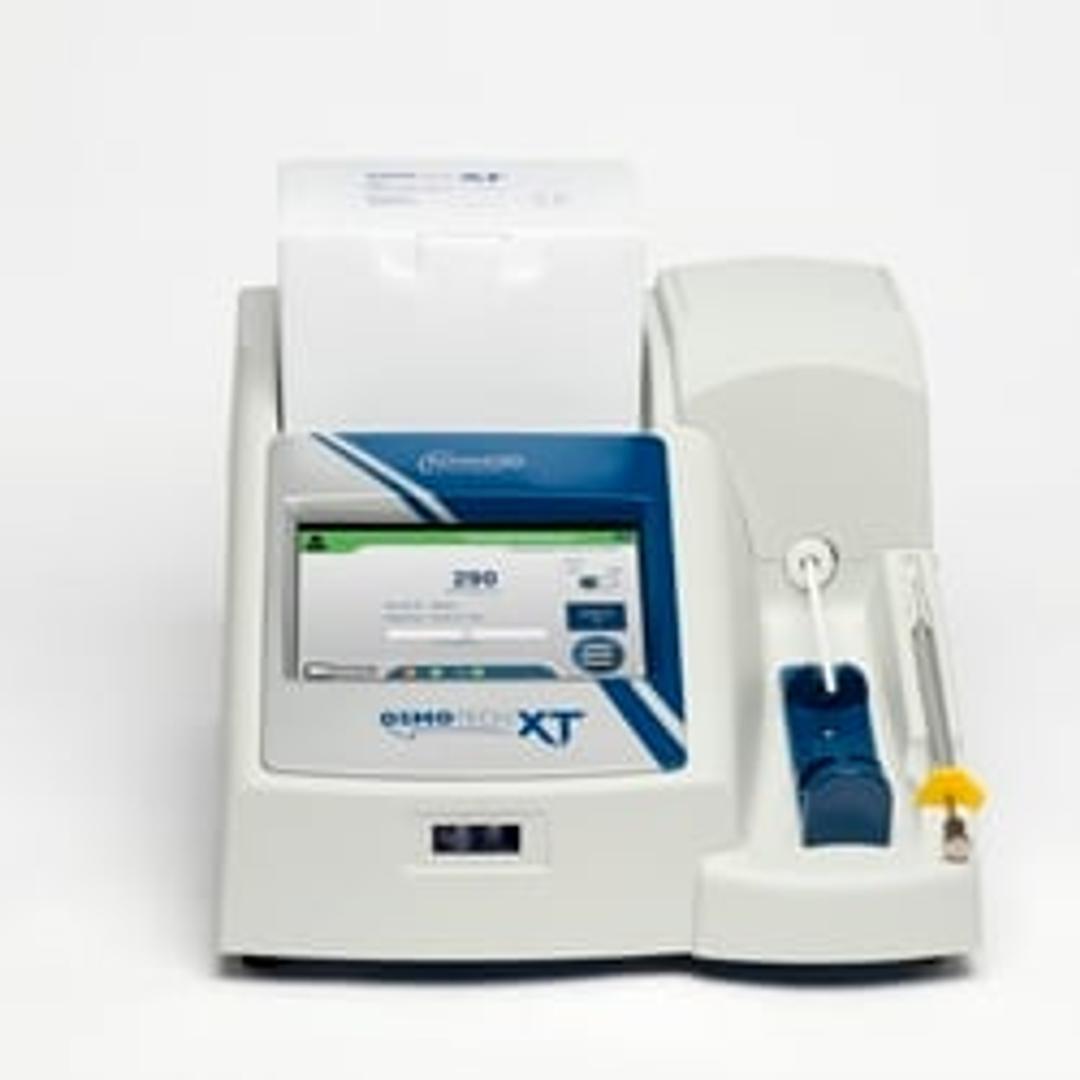
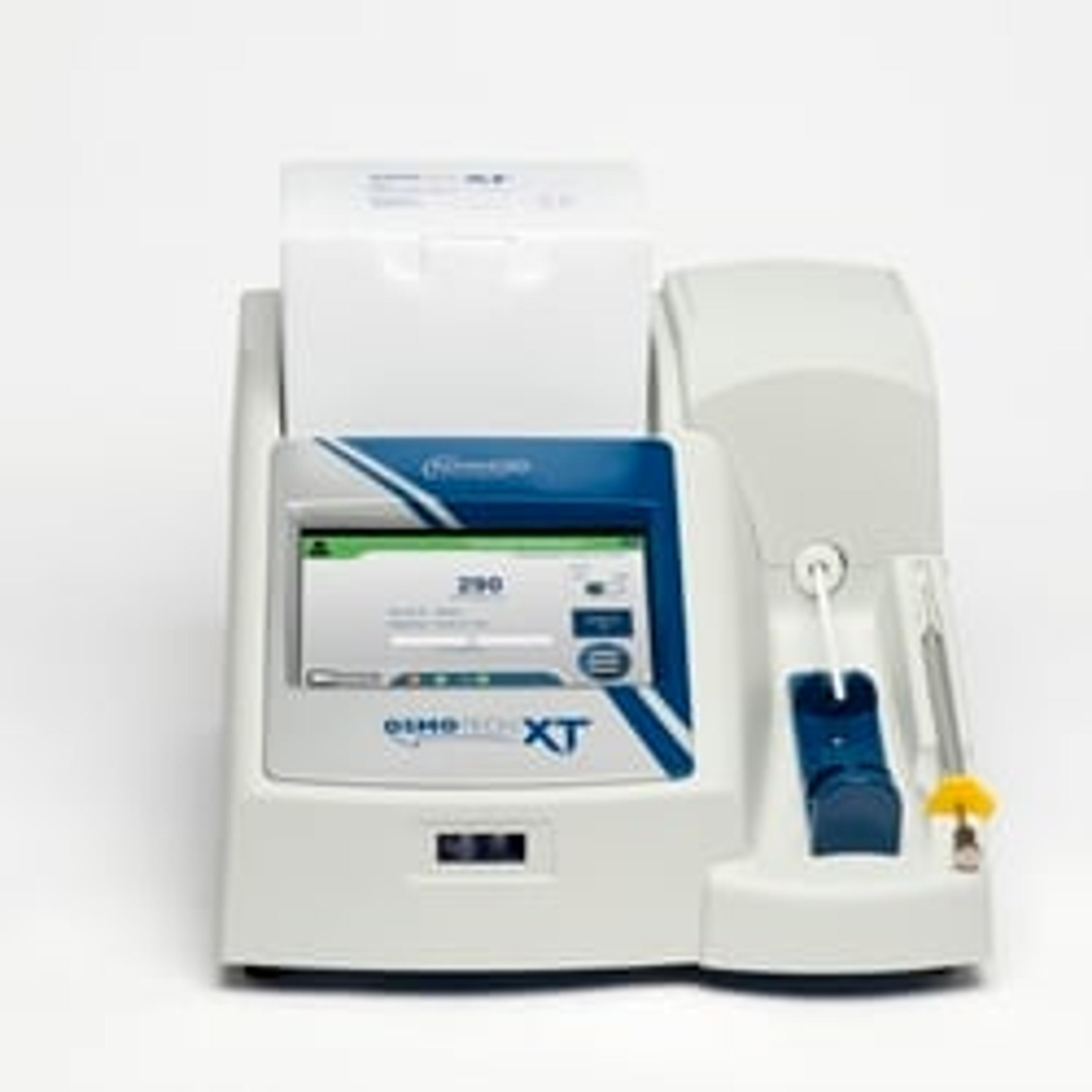
OsmoTECH® XT Single-Sample Micro-Osmometer
Designed specifically to meet the demanding needs of the biotech industry
Great instrument, easy and reliable.
Analytical testing
Easy to use, reliable and self navigating options menu.
Review Date: 22 Mar 2021 | Nova Biomedical (with Advanced Instruments)
Robust and versatile instrument that meets all of our osmolality needs!
Measure the osmolality of media and other reagents related to a cell therapy product
The instrument is very easy and has the capability to be used as an R&D instrument or for a QC lab with its GXP compliant interface. The system has excellent accuracy and precision, even between different analysts. The great thing about the XT model is that it can measure a broad range of sample osmolality to cover all of our sample needs. We have used other Advanced Instruments Osmometers in the past and the freeze point depression osmolality measurements have always been excellent and the instruments are very robust. We are grateful to have this new product that has the versatility that we require from this trusted brand.
Review Date: 2 Dec 2020 | Nova Biomedical (with Advanced Instruments)
OsmoTECH XT is specifically designed to meet the demanding needs of the biotech industry. Its data-management features support 21 CFR Part 11 compliance, and its gold-standard testing method uses freezing point depression to provide the accuracy and precision you need to optimize your process and quality control program. The easy-to-use system is designed to be intuitive, reliable and flexible for those who prefer to directly draw and test small sample volumes. The instrument is able to test various different sample types seen across the bioprocessing and cell & gene therapy workflows - high concentration proteins, sugars, cryopreservatives and even viscous samples.
Features:
- Widest operating range - The OsmoTECH XT offers the widest measurement range of any osmometer, delivering accurate and precise measurements from 0-4000 mOsm/kg
- 21 CFR Part 11 features - Security features of OsmoTECH XT enable labs to satisfy 21 CFR Part 11 compliance requirements.
- Electronic Signature - Ability to review and approve results electronically, thereby supporting truly paperless environment.
- Embedded web server - Easily and securely view, print and download results remotely from a network web browser.
- Connectivity - All data can be automatically exported to a network folder in (.pdf or .csv format) via Network Share or LIMS from anywhere.
- Active Directory - Synchronize all user ID's and passwords with LDAP to have alignment of processes and profiles.
- Intuitive touchscreen - Easy-to-use with an intuitive, color-coded menu-driven operating system, with multi-language capability.
- Industry leading accuracy and precision - OsmoTECH XT utilizes the industry-preferred freezing point depression method to accurately and precisely determine osmolality.
- Meets osmolality testing guidelines as described by Pharmacopeias ( USP, EP, JP and CP).
Brochures
Osmolality and clonality: Critical quality attributes in production of modern therapeutics
In this product brochure, Advanced Instruments presents use cases for single-cell cloning and osmometer technology in monoclonal antibody (mAbs) and cell and gene workflows.
Single-sample micro-osmometer demonstration
In this video, Advanced Instruments demonstrates the OsmoTECH® XT Single-Sample Micro-Osmometer, which has been designed for the demanding requirements of the biotech industry.
Osmolality testing for improved bioprocess development
Discover the benefits of harnessing the latest OsmoTECH osmometer technology for biologics manufacturing