Norovirus-Like Particles
To support norovirus research and development, The Native Antigen Company offers an extended range of 8 norovirus-like particles, produced from their proprietary VirtuE™ (HEK293) and insect-baculovirus expression systems.
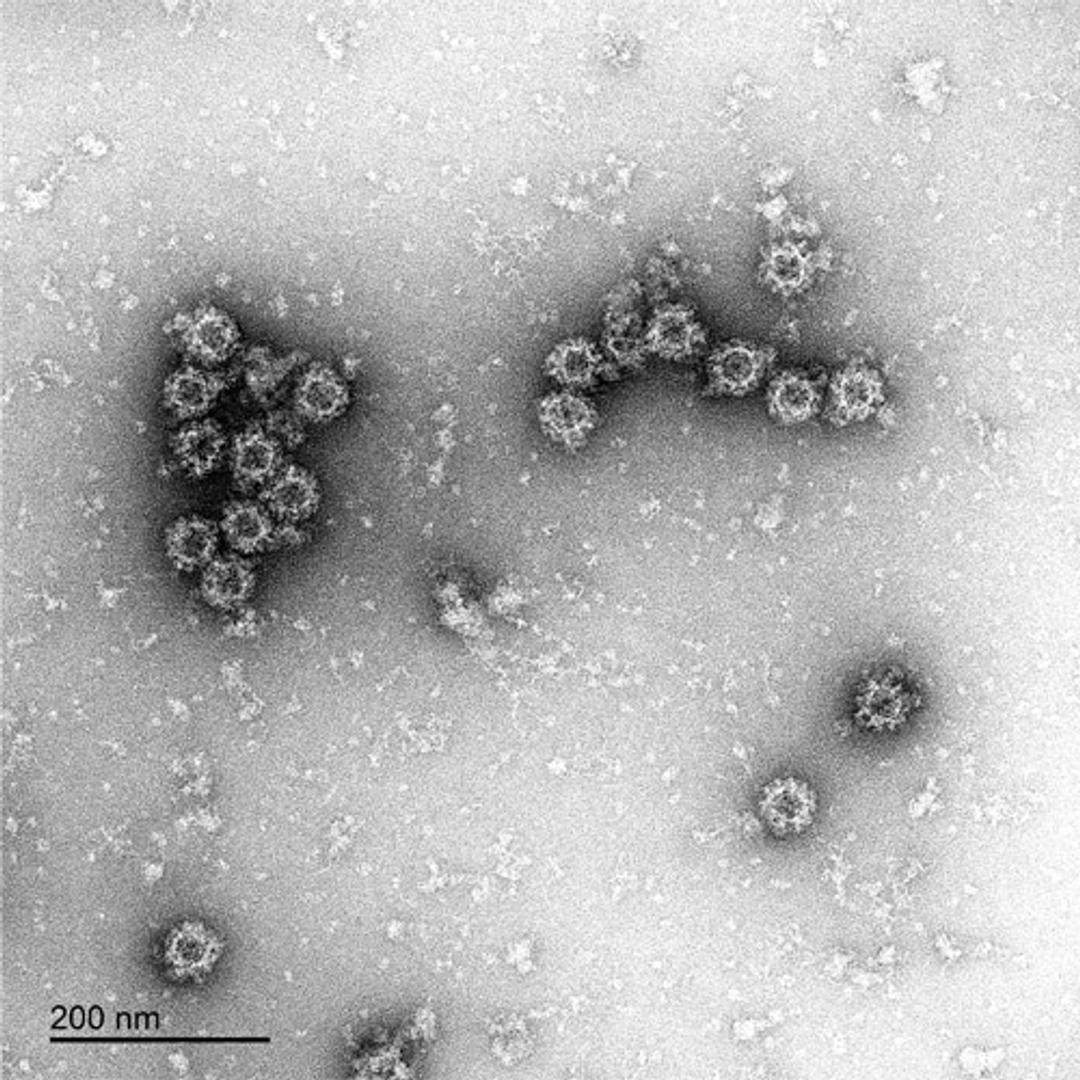
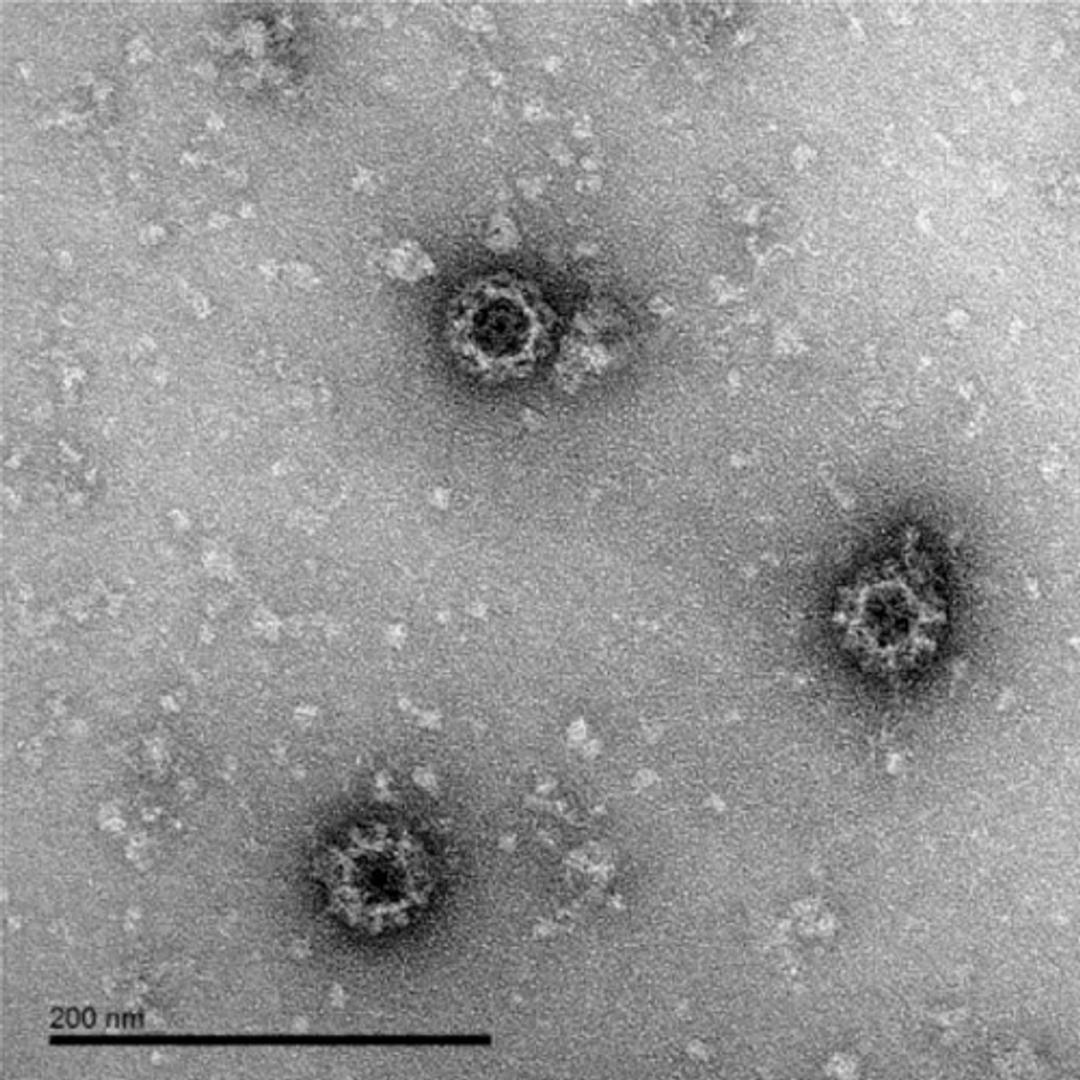
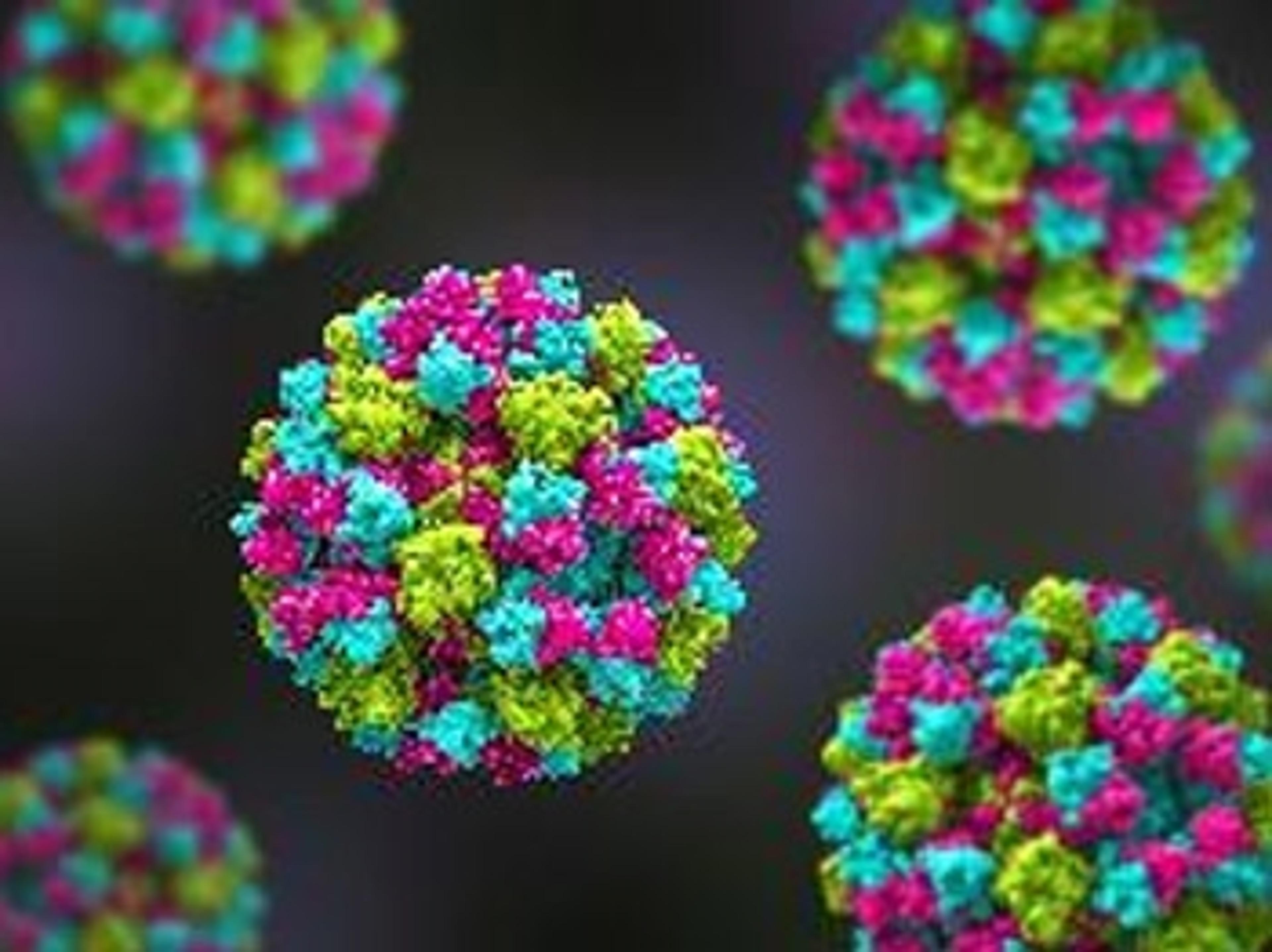
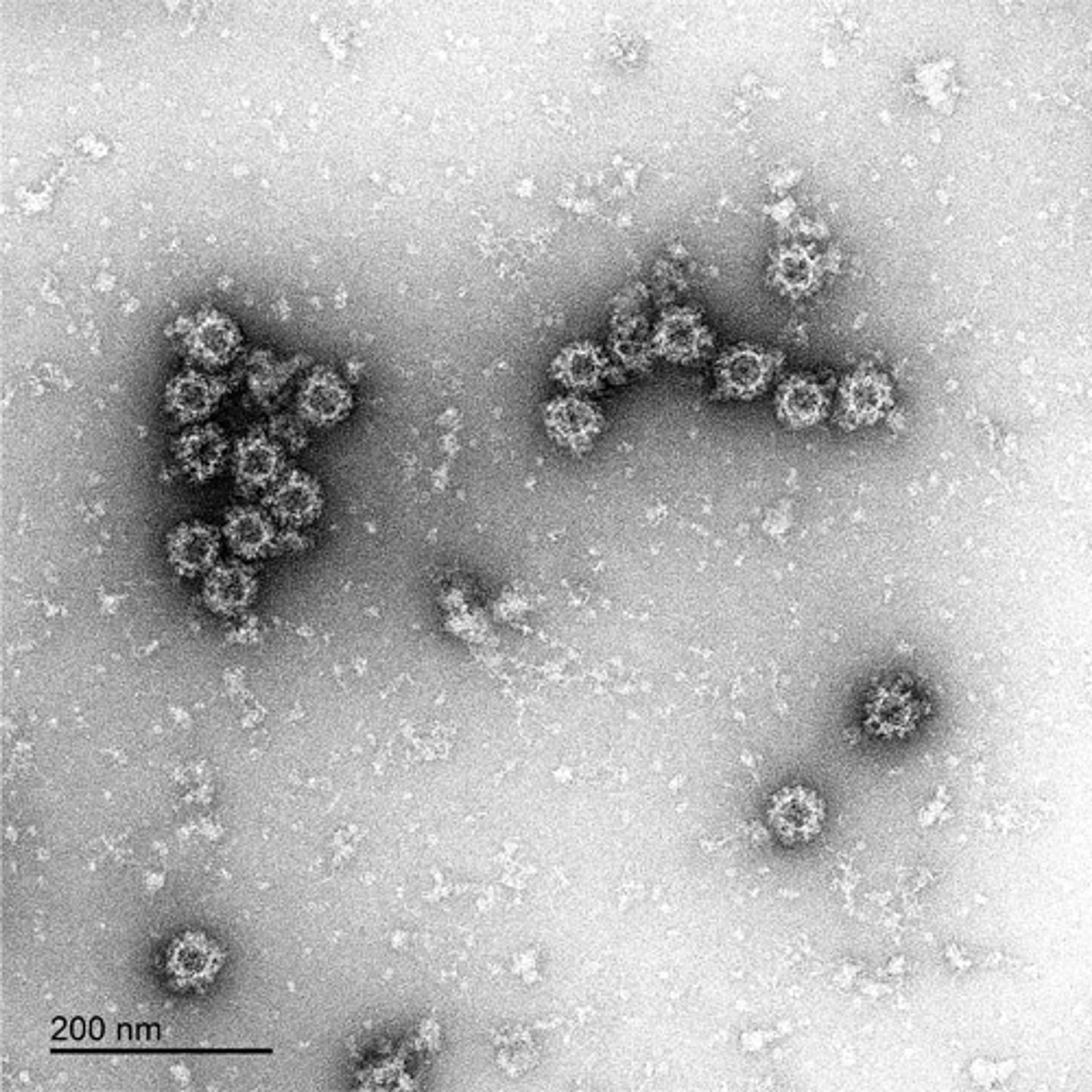
The Native Antigen Companies norovirus VLPs are comprised of VP1 proteins, that self-assemble as an icosahedron of 180 subunits. VP1 is a 59kDa glycoprotein with three key domains: The shell domain (S domain) contains elements essential for the formation of the icosahedron. The Protruding domain (P domain) is divided into sub-domains P1 and P2. The P domain interacts in dimeric contacts that increase the stability of the capsid and form the protrusions on the virion. A hypervariable region in P2 is thought to play an important role in receptor binding and immune reactivity.
The VLPs are non-infectious and safe to use, while displaying native-like epitopes and glycosylation patterns. A study by Metz and colleagues, for example, showed that the VirtuE™-expressed dengue VLPs exhibit comparable epitopes to native viral particles. VLPs are suitable for a wide range of applications, including the development of in vitro diagnostic assays and vaccines. The highly repetitive structural patterns presented by VLPs also makes them ideal antigens for raising high-avidity antibodies.
The Native Antigen Company expands range of norovirus-like particles
The new range is suitable for studies of norovirus protein structure, the research and development of vaccines, and in vitro diagnostic assays