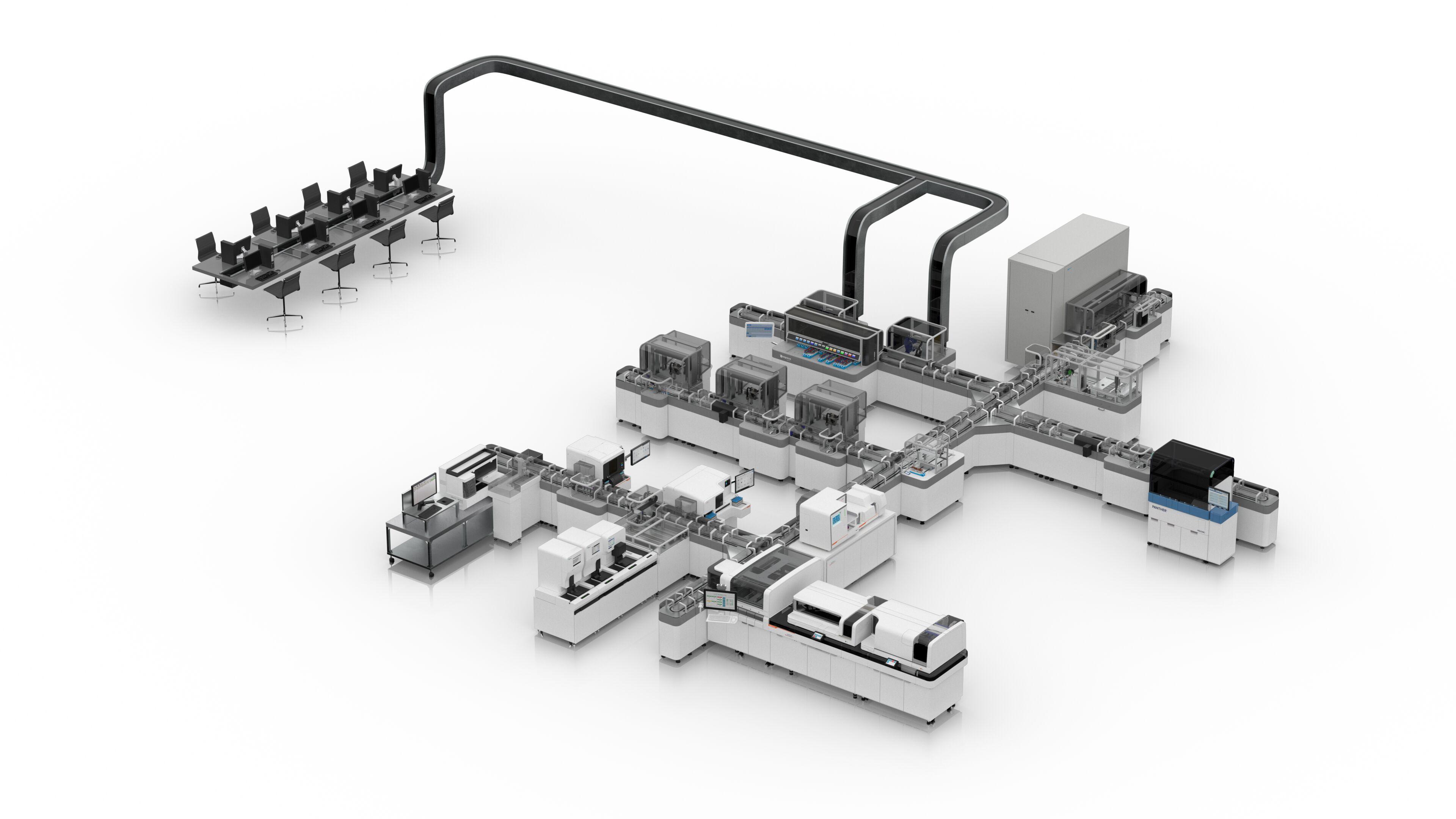
Atellica NEPH 630 System
Smart, Simple and Secure Protein Testing.
The Atellica® NEPH 630 System* is a mid-volume dedicated nephelometric analyzer that simplifies lab operations in specialty protein testing. The system offers the broadest menu of protein tests for assessment on urine, CSF, plasma, and serum, enabling enhanced quantification of certain health conditions. With innovative assays including FLC, CDT, and BTP, the system supports the assessment and monitoring of cardiovascular risk, kidney diseases, neurological disorders, nutritional status, and other diseases.
Features:
- Fast average throughput of approximately 65 tests/hour depending on the assay mix. Nominal:100 tests/hour
- The industry’s largest nephelometric menu of plasma protein assays with more than 60 assay protocols available
- Onboard reagent and control storage provides 24/7 operation, long onboard stability, and minimized operator intervention
- High sample-loading capacity allows load-and-go processing
- Positive bar-code ID of primary sample tubes minimizes manual steps and avoids sample mismatch
- Automatic dilutions and repeat measurement of out-of-range high or low samples are performed without user intervention
- System detects specimen and reagent levels prior to processing to ensure accuracy of results
- Integration with Atellica® PM 1.0 Software** simplifies lab management via aggregated system data, alerts, and the ability to remotely control Atellica NEPH 630 System* and all other connected instruments―all from one screen
* Not available for sale in the US. Product availability may vary from country to country and is subject to varying regulatory requirements. Please contact your local representative for availability.
**Connectivity to Atellica NEPH 630 System is under development. Not available for sale. Future availability cannot be guaranteed
Serum Calprotectin (S100A8/A9) in Diagnosis, Monitoring, and Prognosis of Rheumatic Diseases
Serum calprotectin plays a critical role in the pathogenesis of rheumatoid arthritis (RA), as it triggers chemotaxis, phagocyte migration, and modulation of neutrophils and macrophages. Calprotectin has been found to correlate with disease activity, and may add additional information to that provided by conventional biomarkers such as C-reactive protein (CRP).
This webinar provides an overview of the many options for use of serum calprotectin in adult and pediatric patients suffering from autoinflammatory diseases such as rheumatoid arthritis, Still’s disease, vasculitis, and juvenile idiopathic arthritis (JIA).
Key learning objectives
- Discuss the role of calprotectin as a biomarker in pediatric rheumatic diseases
- Explain the clinical applications of calprotectin in diagnosis, monitoring, and prognosis
- Evaluate how serum calprotectin aids in the diagnosis, monitoring of disease activity, and differentiation between rheumatic diseases and other conditions, such as infections and fever of unknown origin
Who should attend?
Rheumatologists, pediatricians, immunologists, internal medicine, lab directors, and lab managers.
Accreditation statement
SelectScience is approved as a provider of continuing education programs in the clinical laboratory sciences by the ASCLS P.A.C.E.® Program. Attendees of this one-hour webinar are also entitled to 1.0 ACCENT Continuing Education Credits from the ADLM. This activity was planned in accordance with ACCENT® Standards and Policies. Verification of Participation certificates are provided to registered participants based on completion of the activity, in its entirety, and the activity evaluation. For questions regarding continuing education, please email education@myadlm.org.
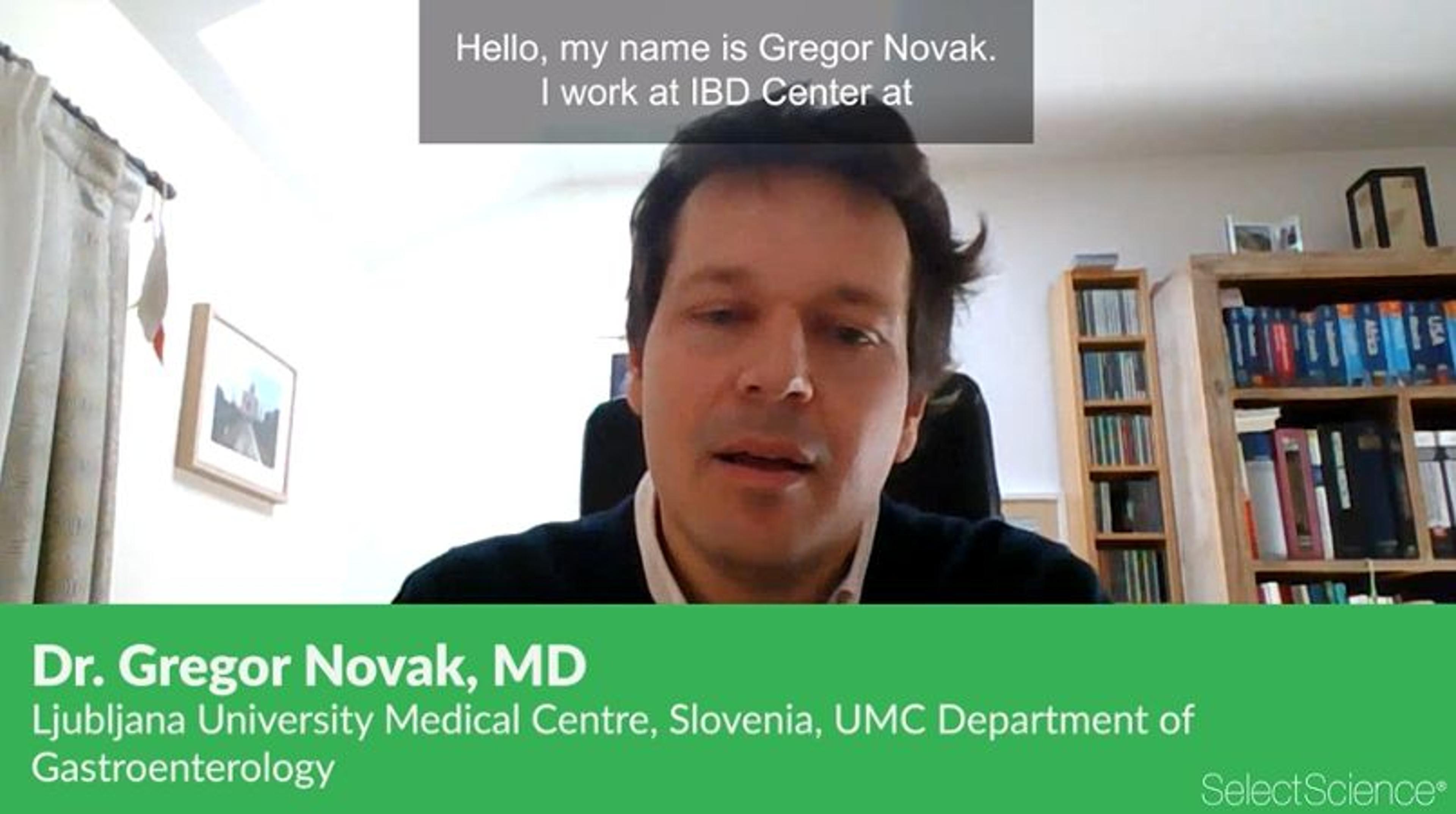
Therapeutic Drug Monitoring in Anti-TNFα Therapy of Inflammatory Bowel Disease
In this video, Dr. Gregor Novak, MD, University Medical Centre Ljubljana Department of Gastroenterology in Slovenia, discusses the current work to improve the outcome for patients with inflammatory bowel disease (IBD).
IBD is a chronic immune-mediated disease, mostly affecting the bowel, and results in symptoms such as abdominal pain, blood in the stool, and urgencies to pass stool. As a result, patients with IBD often develop serious overall reductions in quality of life. Anti-TNFα are biological drugs used to treat IBD patients. However, many patients do not respond well to these drugs or lose response. Dr. Novak’s presentation focuses on therapeutic drug monitoring (TDM) of anti-TNFα with the aim to improve clinical management of patients. Clinical scenarios are presented in which TDM can improve clinical decision-making.
Can kappa free light chains simplify multiple sclerosis diagnostics for laboratories?
New laboratory tools are prompting a rethink of how intrathecal inflammation is assessed beyond oligoclonal bands
Understanding the link between MGUS and multiple myeloma: an expert view
Clinical consultant at Siemens Healthineers explores the link between MGUS and multiple myeloma and reveals how immunoglobulins can be used to effectively aid disease detection
Mologic Achieves Breakthrough in Point-of-Care Diagnostics and Develops New Platform Technology
The novel diagnostics device delivers up to 1,000-fold improvement in sensitivity to support surveillance and elimination efforts of global epidemics