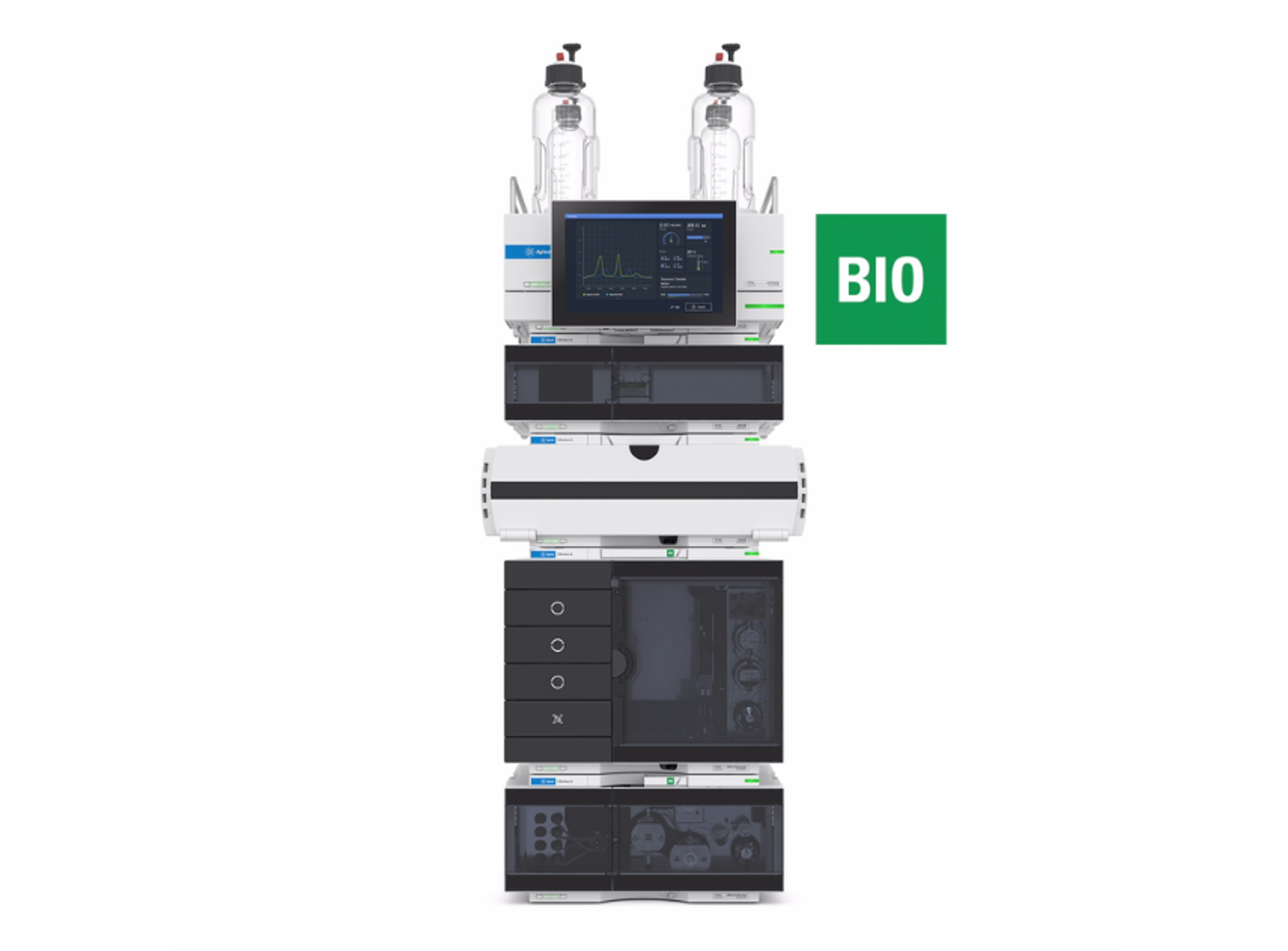
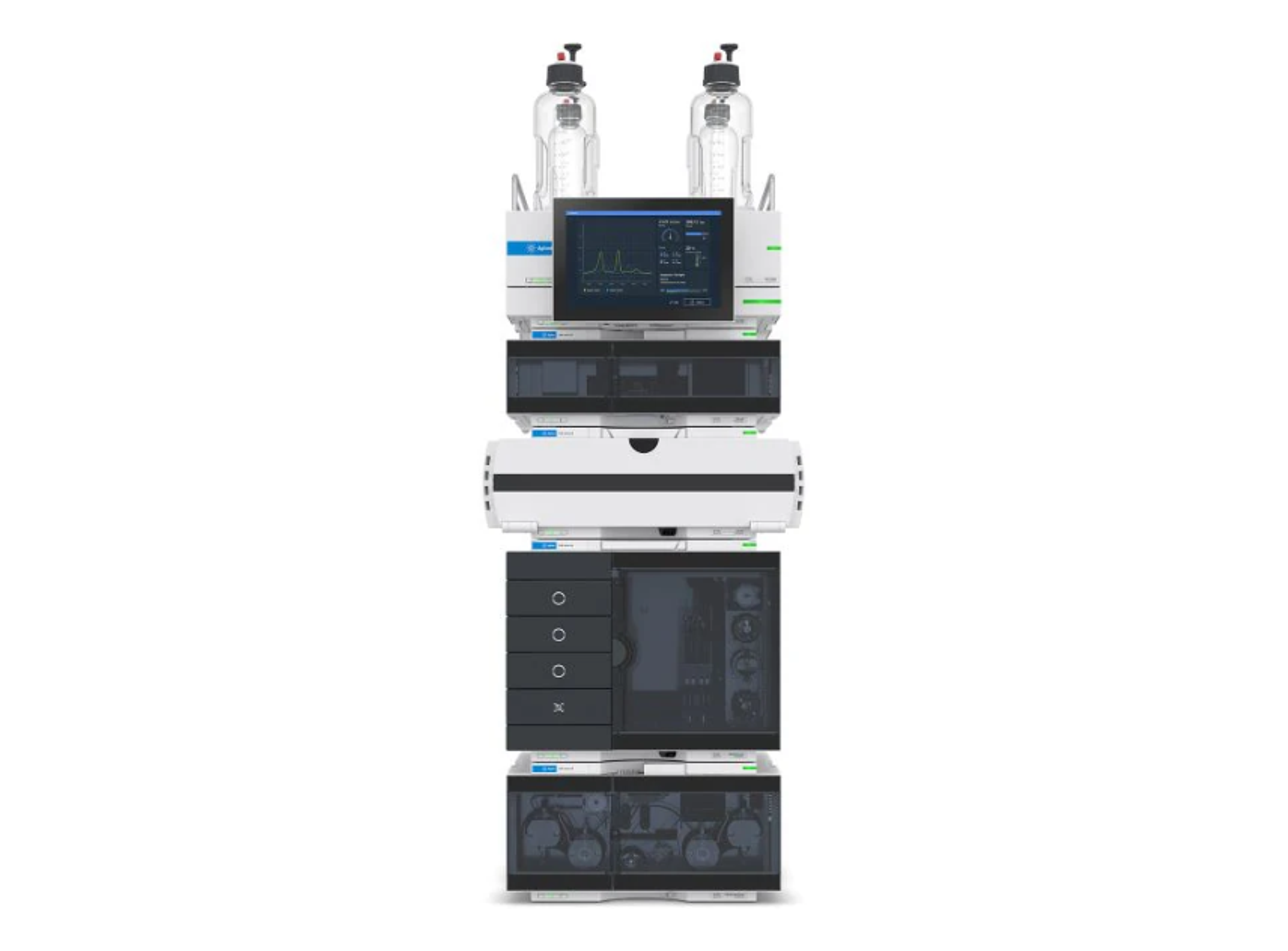
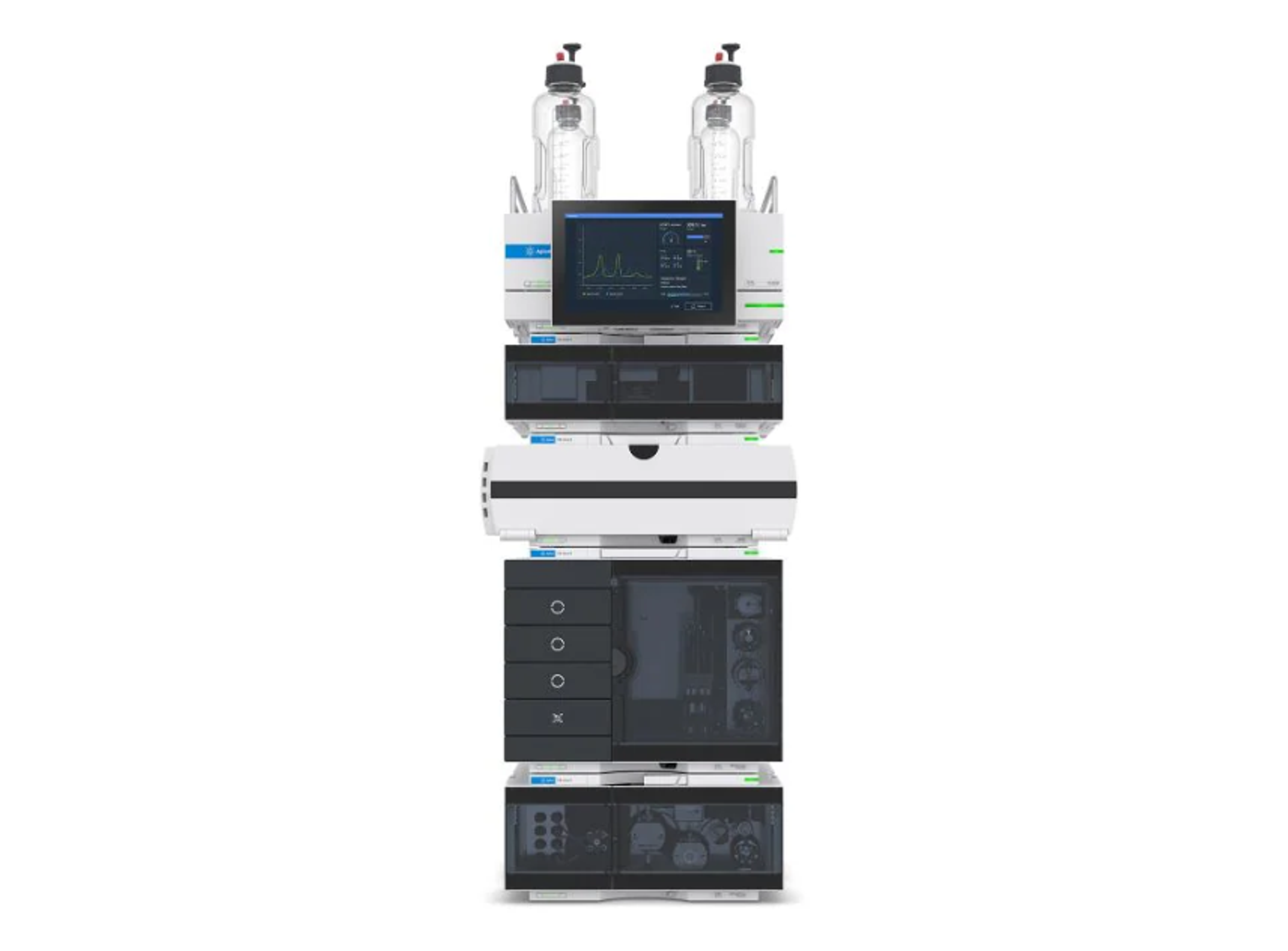
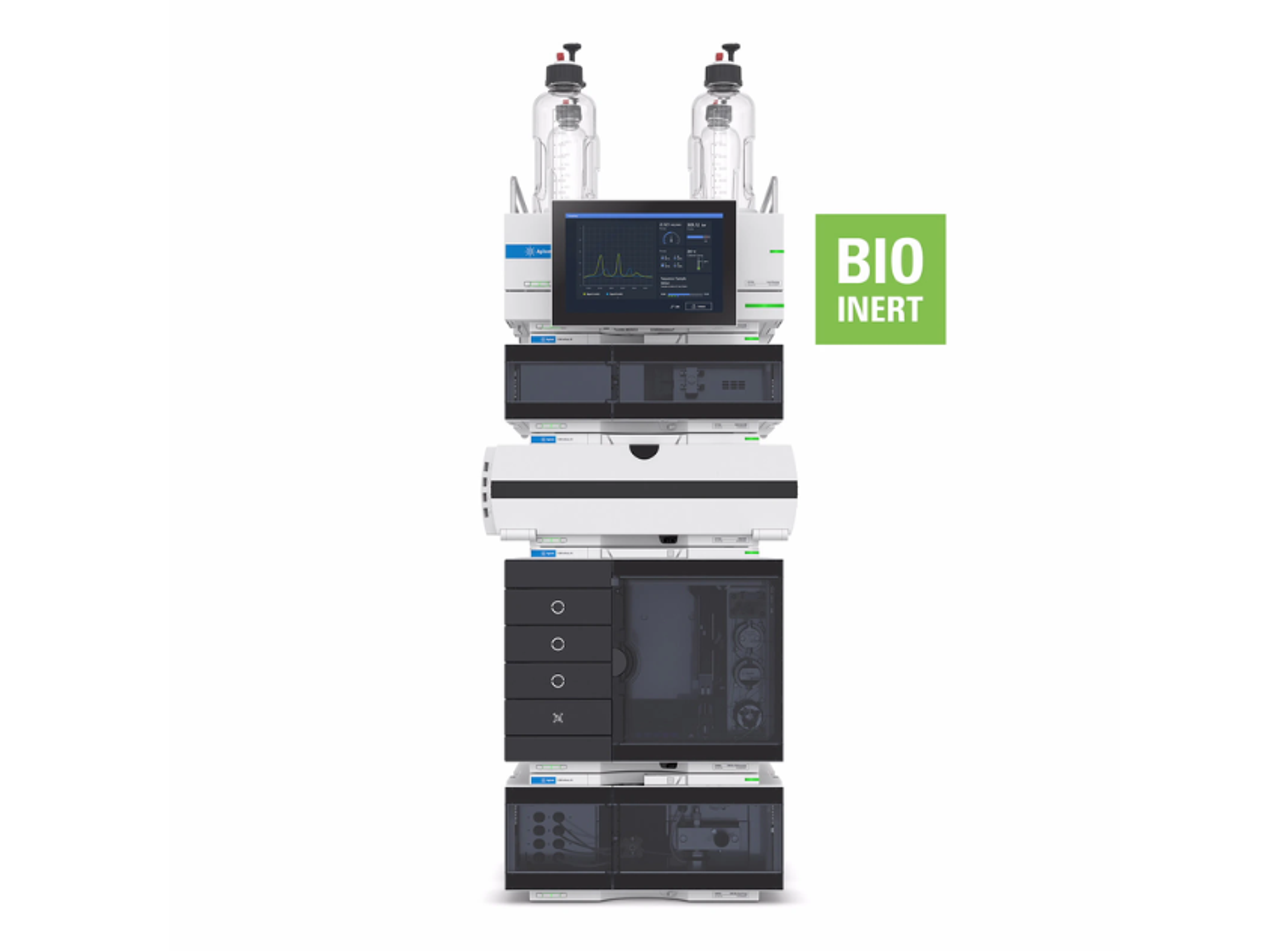
Agilent 1260 Infinity II GPC/SEC System
The 1260 Infinity II GPC/SEC System is designed to meet the challenges faced by today’s polymer analysis. This versatile size exclusion chromatography (SEC) and gel permeation chromatography (GPC) system provides accuracy and high performance.
Once you become familiar with the software, you’ll find it one of the best!
Determination of polymer molecular weight
Customer service is amazing (from quite to installation and further maintenance/support)! Price is reasonable, there are options with additional detectors to be installed. My favorite GPC on the market.
Review Date: 24 Sept 2021 | Agilent Technologies
User friendly. Multiple modes, less cost.
Pharmaceuticals and intermediates
It is easy to operate and can be used for multiple purposes like molecular weight determination and as reverse phase HPLC by using light scattering detector for GPC & UV & RI for regular HPLC analysis. It is less costly than others.
Review Date: 28 Jan 2016 | Agilent Technologies
The system includes a 1260 Infinity II Vialsampler for higher unattended sample throughput and a 1260 Infinity II GPC/SEC Column Thermostat for accurate temperature control and high column capacity for in-row column connection. The 1260 Infinity II Isocratic Pump achieves extra flow precision, and the 1260 Infinity II Refractive Index Detector gives precise results from your GPC/SEC instrument. Additional GPC/SEC special detection capabilities like multi-angle light scattering (MALS) detectors or viscometry can be added for flexibility.
Features:
- Robustness: GPC/SEC instrument handles even the most challenging experimental conditions and solvents used in GPC/SEC applications and workflows
- Accuracy: Repeatable, consistent molecular weight distributions ensure higher-quality data
- Powerful data analysis: Comprehensive, dedicated WinGPC Software or the GPC/SEC Software for OpenLab CDS provides instrument control, data collection, and polymer analysis
- Advanced detectors: Adding optional light scattering and/or viscometry detection enables more information about polymer size and configuration
- High performance: Rapid system stability considerably increases sample throughput
- Your choice for performance and sustainability: Independently audited for environmental impact throughout the product life cycle, received My Green Lab’s ACT (Accountability, Consistency, Transparency) label
Expanding the GPC/SEC Experiment Beyond Conventional Limits using Viscometry and Light Scattering Detection
The advantages of multi-detection GPC to determine absolute molecular weight, size and shape of polymers are highlighted using the 1260 Infinity Multi-GPC/SEC System
From Start to Finish: How Agilent Can Support Better Decisions in IVT mRNA-Based Biopharmaceutical Analysis
Jan 21st 2025 - 16:00 GMT / 17:00 CET / 11:00 EST / 8:00 PST
Development and production of IVT mRNA vaccines require measurements of Critical Quality Attributes (CQAs), such as identity, purity, quality, and quantity. Orthogonal methods developed for such purposes are outlined in the USP guidelines for mRNA vaccines.
Agilent Technologies provides many analytical solutions that are used by leading pharmaceutical companies in early-stage drug development through production quality control (QC) and final product release. These systems help users save time and money with optimized workflows, streamlined processes, and fast time-to-results.
Whitney Pike, M.Sc, Application Scientist and Dr. Sonja Schneider, Manager, Application Development, Agilent Technologies, will discuss how automated electrophoresis and LC/MS systems solutions from Agilent provide reliable insights for IVT mRNA workflows at many steps, from QC of the initial plasmid DNA, throughout the IVT process, to the finished mRNA drug product.
Key learning objectives
- Explore QC throughout the entire IVT mRNA workflow, including data from various nucleic acids and LNP analyses
- Discover how Agilent instruments/technologies, such as the Fragment Analyzer and LC/MS systems, can be used for different CQAs
- Learn how Agilent is working together with its partners to provide reliable insights for all aspects of the IVT mRNA workflow
Who should attend?
- Researchers, lab managers, lab directors, QA/QC managers performing IVT mRNA analysis in biotherapeutics research and production
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
For Research Use Only. Not for use in diagnostic procedures. PR7001-2498
Agilent Technologies Receives the Scientists’ Choice Award for Best New Separations Product
Agilent Technologies won the Scientists’ Choice Award for the Best New Separations Product of 2016 for the 1260 Infinity II LC system. In this video, hear Helmut Schulenberg-Schnell, Director of Business Development at Agilent Technologies, explain how much the accolade means to the company.
Agilent Wins Reviewers’ Choice Award
Marc Boreham, from Agilent Technologies, accepts the Reviewers’ Choice Award for Customer Service of the Year 2015. The award, which was voted for by the global SelectScience community, was presented at the exclusive Scientists’ Choice Awards presentation ceremony at Pittcon 2015 in New Orleans, LA.