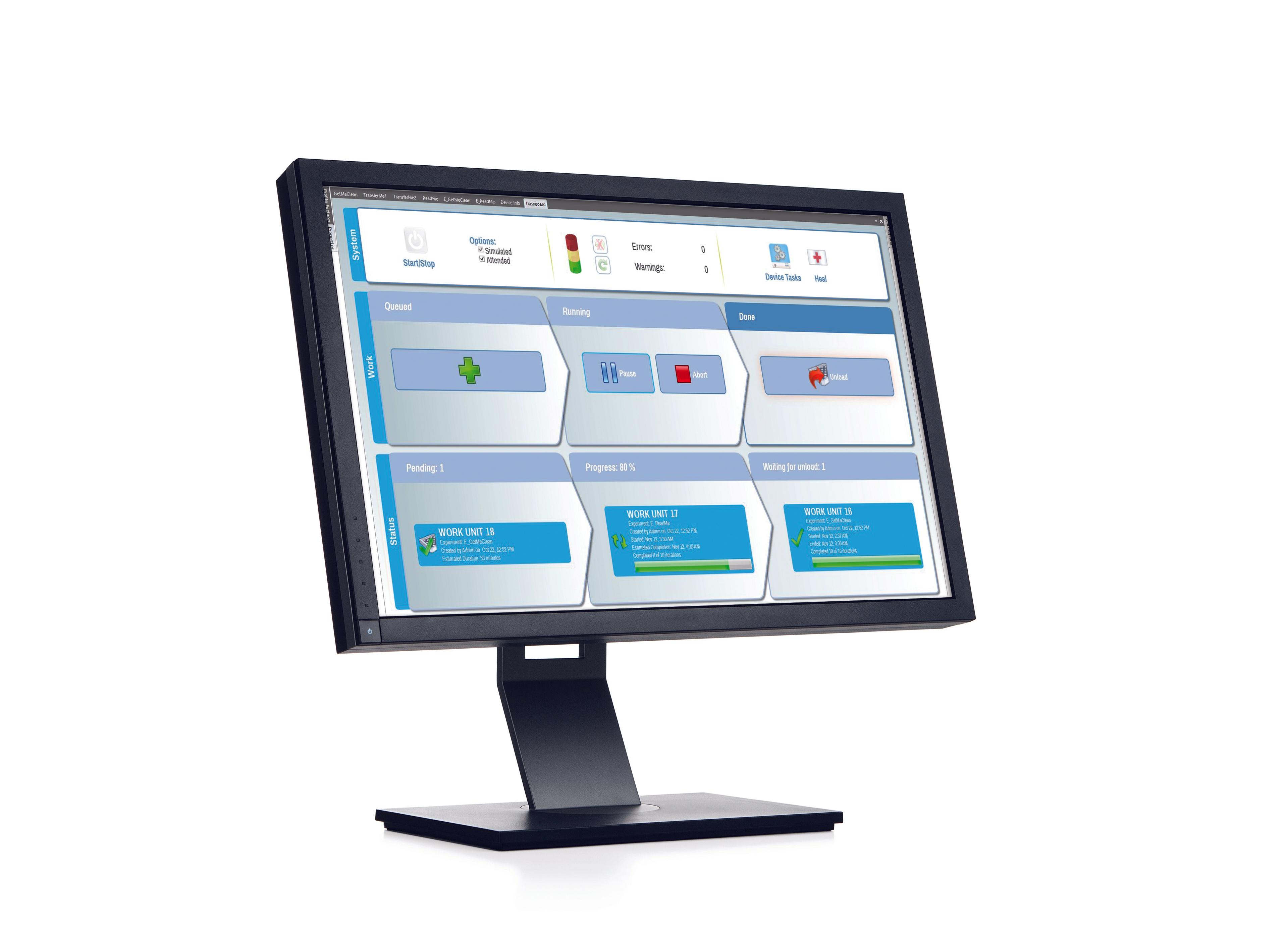
Thermo Scientific™ SureTect™ PCR Automation Platform
Enhance your laboratory productivity and performance with the Thermo Scientific™ SureTect™ Automation Platform, utilizing the CyBio™ FeliX Instrument: Take your food safety testing to the next level with automated lysis and PCR preparation. The simple workflow and rapid setup allows you to reduce hands-on time while increasing the accuracy of your testing, enabling your technicians to focus on other tasks.
Scalable automation for reliable food safety testing results
Fully validated by AFNOR and AOAC for use with all SureTect PCR Assays, the Thermo Scientific™ SureTect™ Automation Platform utilizes the CyBio™ FeliX Instrument's flexible programs, user-friendly interface, and rapid yet precise setup to ensure maximum efficiency, whilst the compact design saves valuable bench space.
Time saving
- Fully automated lysis, PCR setup or complete workflow
- Rapid 5-minute setup for loading reagents
Intuitive software
- Pre-programmed runs for quick setup
- User-friendly interface with no specialist training required
Control
- Control your consumable costs and wastage with the Flex Package option, allowing you to use the Head R96 Pipetting head as either a 96-channel pipetting or 8-channel pipetting, at any time
- Increase efficiency of operation with choice of runs, reducing service cost and time
- Be confident in your processes with the decontamination unit
Accurate and traceable
- Barcoded sample entry for full traceability
- Precise pipetting
- Robust and factory calibrated instrument
Brochures
Thermo Scientific SureTect automation workflow
In this product brochure, Thermo Fisher Scientific demonstrates the Thermo Scientific™ SureTect™ automation workflow, which can help you take your testing to the next level with automated lysis and PCR setup.
Visibly simple food pathogen testing
In this product brochure, Thermo Fisher Scientific demonstrates how you can transform your complete food safety testing workflow with the innovative Thermo Scientific™ SureTect™ Food Pathogen Detection PCR System, enabling you to work efficiently and release products quickly with a simple, rapid pathogen detection system validated for a broad range of matrices.
Rapid detection of spoilage organisms in beverages
Beverage producers face increasing pressure to ensure product integrity, quality, and brand protection in the face of complex spoilage risks. From yeast and mold contamination to spoilage bacteria, identifying and addressing these issues early is critical to maintaining quality and operational efficiency.
Discover how the Thermo Scientific™ SureTect™ Beverage Spoilage Multiplex PCR Assay provides a fast, broad-range molecular solution for detecting key spoilage microorganisms that threaten beverage quality.
The PCR automation advantage for food safety professionals
In an industry where precision, speed, and compliance are non-negotiable, this guide to PCR automation for food safety offers a comprehensive roadmap for laboratories ready to modernize their microbiological testing workflows. Developed in partnership with Thermo Fisher Scientific, this essential resource explores how automated PCR technologies are transforming food safety testing, reducing human error, increasing throughput, and ensuring regulatory compliance across global markets.
Inside, you'll discover how the Thermo Scientific™ SureTect™ PCR System delivers rapid, reproducible results for high-volume labs, with proven equivalency to manual workflows. Real-world case studies from Coca-Cola Europacific Partners and OMIC USA Inc. illustrate how automation has helped leading organizations overcome labor shortages, scale operations, and maintain accuracy under pressure. Whether you're testing for Salmonella, Listeria, E. coli, or other pathogens, this guide provides actionable insights into selecting the right workflow, manual or automated, based on your lab’s size, goals, and regulatory environment.
Download this SelectScience guide to explore:
- Learn how PCR automation can future-proof your lab against rising sample volumes and staffing challenges.
- Compare manual vs. automated workflows with detailed pros and cons.
- Explore validated methods and certifications (ISO 16140-2:2016, AOAC) for global compliance.
- Access implementation checklists, workflow audits, and expert recommendations to streamline your transition.
If your lab is ready to move beyond traditional culture-based testing and embrace a faster, more scalable molecular approach, this guide is your starting point.
Resource details:
- Document type: SelectScience guide
- Page count: 28
- Read time: 42 mins
- Edition: 1st
The Thermo Scientific SureTect PCR System – OMIC USA’s choice for reliable, robust foodborne pathogen testing
In this case study, Thermo Fisher Scientific explores why OMIC USA Inc., an independent analytical testing lab located in Portland, Oregon, selected the Thermo Scientific™ SureTect™ PCR System for foodborne pathogen testing.
Bringing the benefits of PCR and automation to high-throughput beverage testing, with the SureTect Automation Platform
In this case study, Thermo Fisher Scientific explores how a team at Coca Cola Europacific Partners integrated the Thermo Scientific™ SureTect™ Automation Workflow using the CyBio™ FeliX, to reduce the hands-on demand of the upfront sample preparation.
How to navigate the food regulatory landscape for Listeria spp. and Listeria mono. testing
Friday, November 21, at 13:00 GMT | 14:00 CET | 08:00 EST | 05:00 PST
Listeriosis remains a serious public health concern. In 2023, thirty EU/EEA countries reported 2,993 confirmed cases, the highest annual total since surveillance began. In the U.S., around 1,250 cases occur each year, leading to approximately 1,070 hospitalizations. Between 2016 and 2023, a multi-country cluster linked to ready-to-eat foods resulted in 64 cases across five EU/EEA countries
These persistent outbreaks continue to challenge food producers worldwide and in response to this, regulatory requirements globally have been rapidly evolving in recognition of these threats. Remaining compliant in this fast-changing landscape adds further complexity for testing laboratories.
Regulatory bodies have responded to these threats with revisions and tightening of the current regulatory frameworks for Listeria spp. and L. monocytogenes:
- FSIS broadened its testing to include non‑Listeria monocytogenes species from January 17, 2025.
- EU Regulation 2024/2895 sets a '0 cfu/25 g' requirement for Listeria in ready‑to‑eat foods.
Register for this exclusive webinar to hear from microbiology experts, Katharine Evans, François Le Nestour and Dr. Salman Zeitouni as as they discuss how labs can stay ahead by adopting internal standards that exceed minimum regulatory requirements and by implementing robust, adaptable pathogen‑testing programs.
Key learning objectives:
- Uncover the fundamentals of Listeria and its pathogenicity
- Review major Listeria outbreaks and their human impact
- Decode recent regulatory changes from the US and EU on Listeria
- Learn how to design and validate testing protocols that go beyond minimum regulatory requirements, ensuring continuity when regulations change
- Explore Thermo Fisher Scientific's microbiology solutions for rapid detection and enumeration of Listeria and Listeria spp.
Who should attend?
- Food testing professionals
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.
Reaching new heights in food safety testing using automated PCR workflows
Tuesday, September 30, at 15:00 BST | 16:00 CEST | 10:00 EDT | 07:00 PDT
Food safety testing laboratories are under growing pressure to do more with less. As testing volumes surge, labs must deliver accurate and regulatory compliant results, while navigating staffing shortages and limited resources. These challenges are further compounded by increasing sample sizes and throughput demands.
In this webinar, meet with Dr. Salman Zeitouni and Jani Holopainen to explore real-world examples of how automation is driving transformation in food safety testing laboratories. See how labs are leveraging automated solutions to enhance accuracy and efficiency, reduce human error, and build resilient testing capabilities.
Key learning objectives:
- Explore the drivers behind automation of molecular workflows in food safety testing laboratories
- Understand key considerations and common challenges in adopting lab automation
- Discover real-world examples of successful automation implementation in microbiology labs
- Gain insight into how automated molecular workflows can support lab scalability, traceability, and long-term resilience to build a future lab today.
Who should attend?
- Laboratory professionals, including food microbiologists, lab managers, and technical service directors.
- Quality, safety, and regulatory leaders, such as quality managers, operations directors, and compliance professionals.
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.