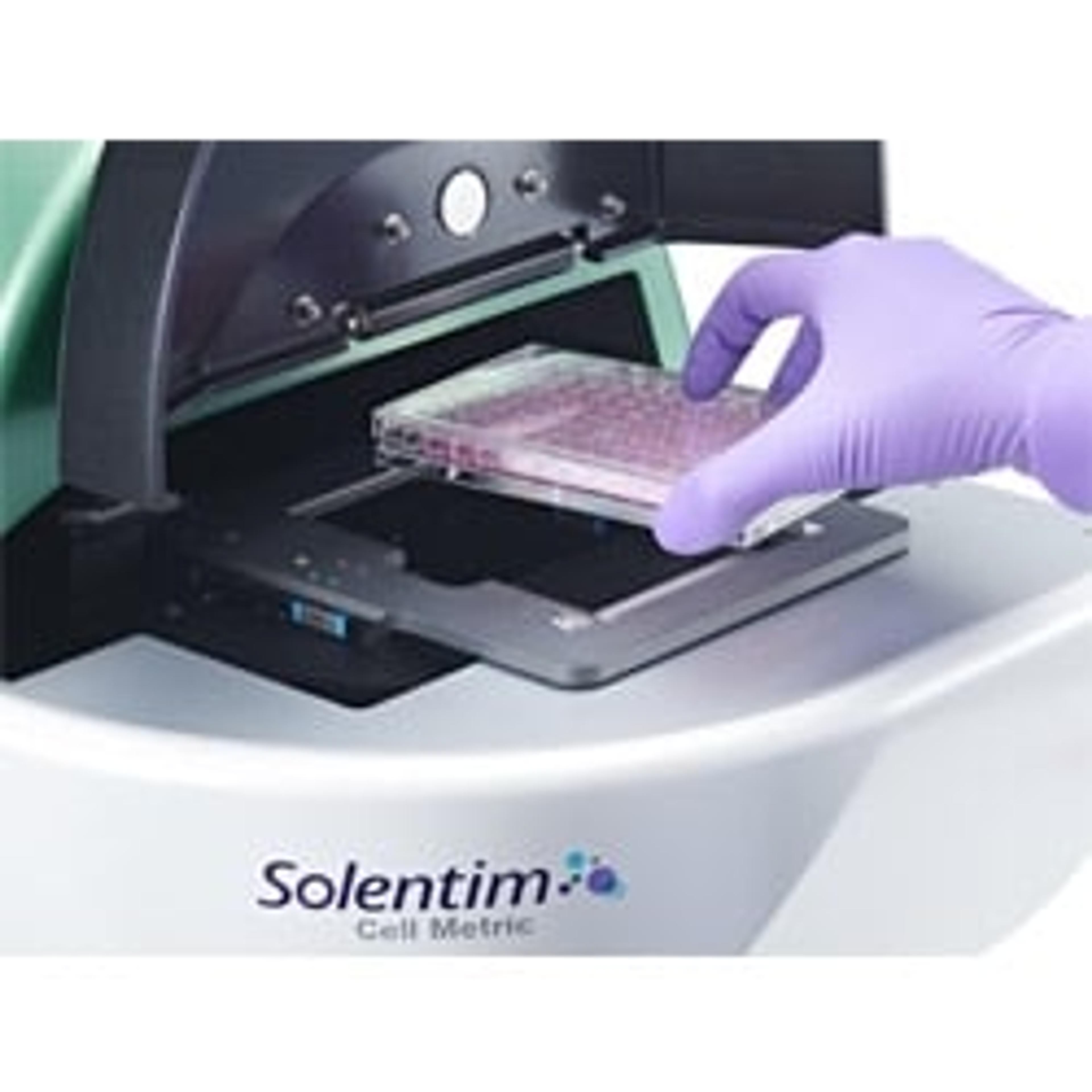
Solentim VIPS® PRO
VIPS PRO’s powerful seeding technology gently and efficiently dispenses cells, reducing hands-on time and compressing timelines from months to days while providing certainty of clonality and superior imaging with advanced data management and GMP-friendly features.
Its single-use Seeding Kits eliminate the possibility of sample carryover and contamination between projects and the integrated STUDIUS™ software platform provides security features to support 21 CFR Part 11, and more.
VIPS PRO is an all-in-one gentle seeder and imaging platform designed to perform single cell isolation and whole-well imaging to support cell line development. Gentle seeding combined with artificial intelligence (AI)- -driven cell detection ensures fast and extremely precise clonality determination. Industry-leading imaging system provides undisputable proof of clonality and AI-based confluence determination to support IND and BLA submissions.
Our STUDIUS™ software with its 21CFR Part 11 compliant features enables clone tracking across the CLD process and audit trail to ensure compatibility with GMP-workflows.
Our GMP-certified consumables are designed to facilitate gentle seeding with minimal hands-on time, while maintaining cell viability, seeding efficiency, and throughput.
Ideal solution for:
- Cell line development
- Biologics production
- Stem-cell based cell therapy
- Viral vector manufacturing
Proof or probability? Latest technology for assurance of clonality in cell therapy manufacturing
For cell therapy manufacturing, an important aspect of an investigational new drug (IND) application is demonstrating that the generated cell line is clonally derived while demonstrating GMP compliance. In this webinar, we will explore key considerations for the early stage of clinical GMP manufacturing including single cell deposition methods, the importance of monoclonality assurance, consistency in the workflow, and the reduction of project timelines.
Choosing the right technology, tailored to comply with GMP regulations, will have a huge impact not only on the automation of the single cell cloning, but also on reducing hands-on time. This provides the audit trail and documentation needed for IND submission and compresses the manufacturing timelines from months to days.
Key learning objectives
- Understand the key considerations for clinical manufacturing under GMP regulations
- Understand the importance of monoclonality assurance for IND submission
- Learn about the new GMP-compliant technology for improved single cell deposition
- Discover the best approach to track clones during their journey through the manufacturing process
Who should attend?
- Biotech, large pharma, start-ups or CDMOs and CROs carrying out cell therapy, regenerative medicine and artificial meat production using:
- Induced pluripotent stem cells (iPSCs)
- Mesenchymal stem cells (MSC)
- Embryonic stem cells (ESM)
- CAR-T cells
- Academics working in cell line development and iPSCs and stem cells
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
Solentim VIPS PRO: Elevate your single cell cloning workflows
In this video, learn more about the Solentim VIPS™ PRO Single Cell Seeder from Advanced Instruments. Explore how the instrument Is designed to optimize your workflow productivity and give you complete confidence in single cell seeding and clonality by delivering imaged-based proof of monoclonality combined with proprietary artificial intelligence-driven cell recognition.
Stem cell research – Expert insights into the new frontier of modern medicine
Keep up to date with the latest techniques and technologies and techniques driving stem cell research into 2023
Cell therapy: A GMP-compatible solution for ensuring clonality during iPSC manufacturing
The journey to the VIPS™ PRO single-cell seeding platform