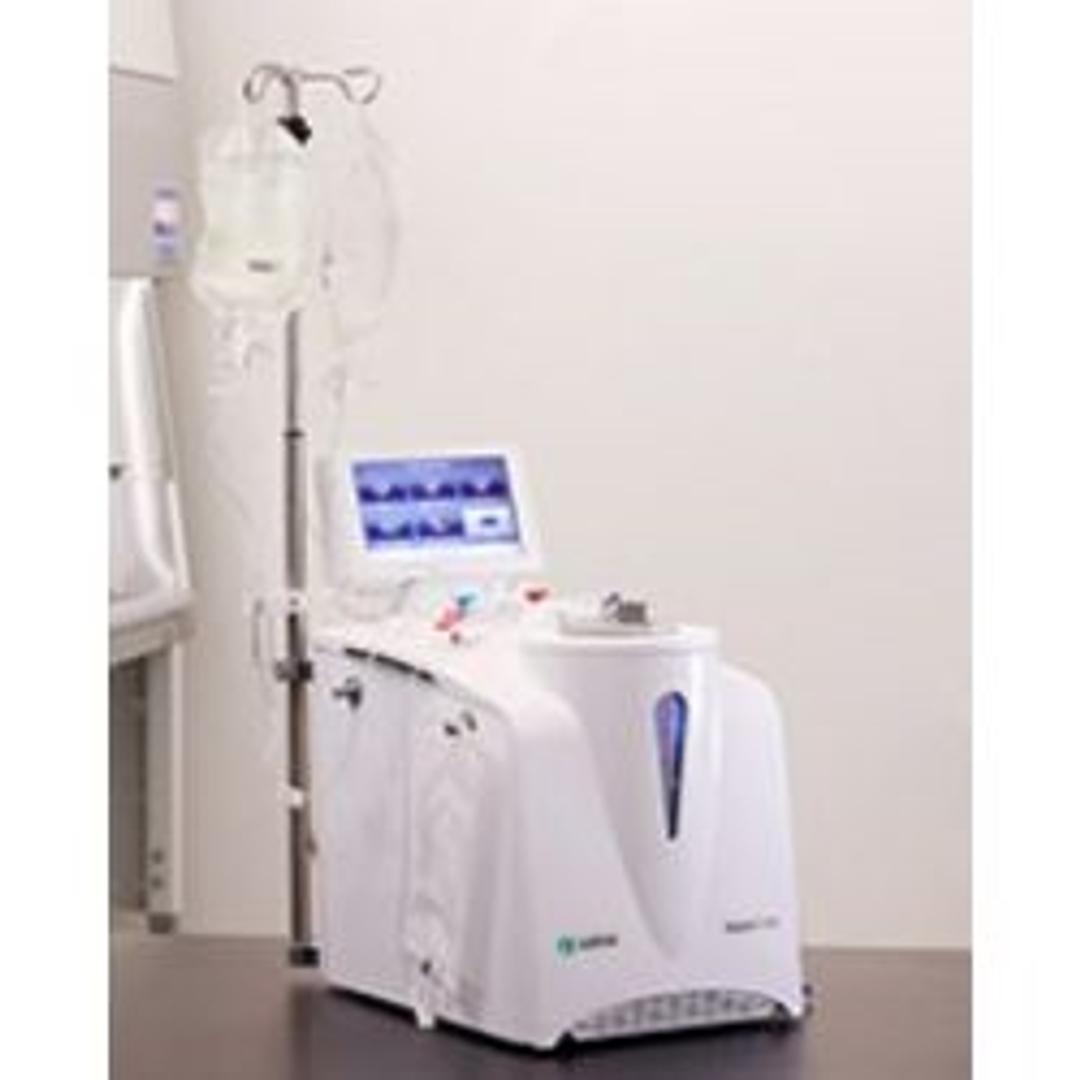
Sepax™ C-Pro Cell Processing System
The Sepax C-Pro Cell Processing System, which is comprised of the instrument, Sepax C-Pro protocol software, and kits, allows you to combine multiple processing steps in a versatile manner. These steps include but are not limited to enrichment, magnetic bead incubation, spinoculation, concentration, washing, dilution, and splitting.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Use this flexible instrument in combination with protocol software and kits to automate and functionally close specific operations in cell therapy.
- Support your requirements and system implementation. Automated and functionally closed system to support GMP compliance.
- Simplify your operations. Compact design and user-friendly interface minimize your space and resource requirements.
- Maintain flexibility. Sepax C-Pro is a multipurpose solution allowing you to perform diverse and dedicated applications.
- Simplify record-keeping. Sepax C-Pro can transmit instrument data and alarms for remote monitoring or for incorporation into a Chronicle eSOP.
- Keep operations running smoothly. Integrate with Chronicle manufacturing automation software to monitor all connected instruments (Sepax C Pro and others) and manage service records.
Streamlining the CAR T cell therapy workflow: A look into closed and semi-automated processes
In this application note, Cytiva outlines a CAR T cell manufacturing workflow, addressing challenges associated with the complex and costly manufacturing process of T cell immunotherapies. The described semi-automated, closed CAR T process is designed for commercial production, achieving a substantial expansion of T cells (1 × 1010) with high transduction efficiency (greater than 80% eGFP) over an 8-day manufacturing period. This approach aims to enhance the feasibility and efficiency of CAR T cell production for widespread clinical applications.
TIL therapy manufacturing
In this infographic, Cytiva addresses challenges at each stage of the TIL (Tumor-infiltrating lymphocyte) therapy manufacturing workflow. It considers the challenges of each stage and provides solutions. The workflow presented covers tumor tissue resection and transfer, tissue disaggregation and feeder cells, selection and expansion (pre-REP), TIL activation and expansion (REP), harvest and formulation, cryopreservation and shipment, and therapy delivery.
Optimize your cell therapy manufacturing workflow with the Sepax C-Pro system
Join us for an informative webinar introducing the SepaxTM C-Pro cell processing system. This session delves into strategies for optimizing cell recovery during harvesting and how to obtain a high-efficiency automated spinoff relation gene transfer.