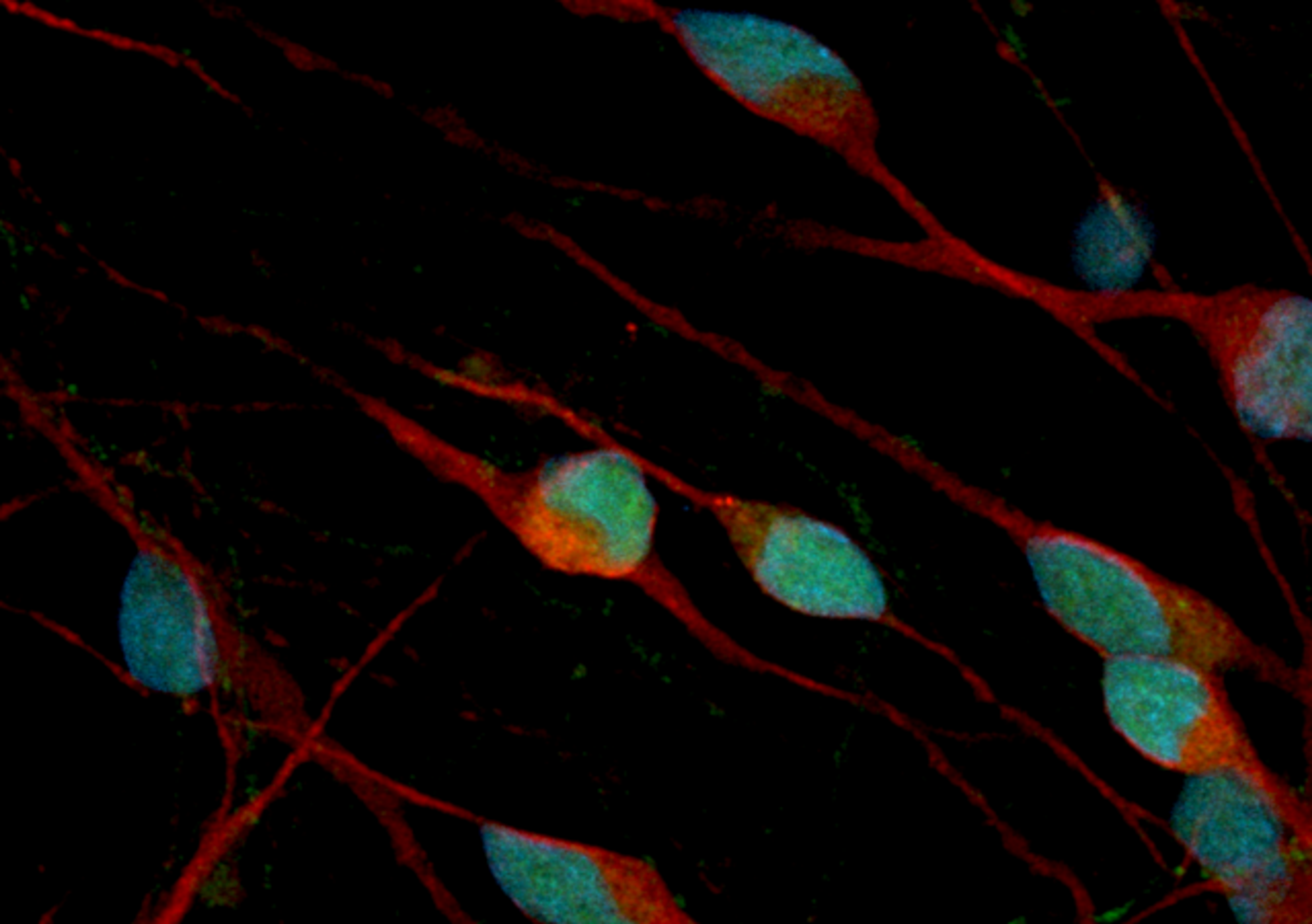
Rubella Virus VLP
Recombinant Rubella (strain F-Therien) virus-like particles consisting of spike glycoprotein E1, spike glycoprotein E2 and Capsid protein, produced in mammalian HEK293 cells.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Recombinant Rubella (strain F-Therien) virus-like particles consisting of spike glycoprotein E1, spike glycoprotein E2 and Capsid protein, produced in mammalian HEK293 cells.
Rubella virus-like particles are produced by recombinant expression of Rubella structural polyprotein (amino acids 1-1063, Uniprot accession number NP_062884.1) in HEK293 cells.
Rubella virus is an enveloped, positive single-stranded RNA virus and a member of the genus Rubivirus, which belongs to the Togaviridae family. It consists of three structural proteins: a capsid protein and two membrane-spanning glycoproteins, E1 and E2, localized in the virus envelope (Oker-Blom, et al., 1983).
E1 and E2 exist as a heterodimer and form the viral spike complexes on the virion surface. Formation of an E1-E2 heterodimer is required for transport of E1 out of the endoplasmic reticulum lumen to the Golgi apparatus and plasma membrane (Yang, et al., 1998). E1 is the dominant surface molecule of the virus particle representing the main target for the detection and subsequent elimination of virus by the host’s immune system (Green & Dorsett, 1986; Wolinsky, et al., 1983). Although both E1 and E2 provide lifelong immunity, hemagglutination (HA) and viral neutralization (VN) is believed to be mainly targeted to E1 protein epitopes (Chaye, et al., 1992).
Immunogenicity studies have shown Rubella VLPs to be significantly more active than soluble E1 protein in inducing antibody responses in mice, especially for producing VN and HA-inhibiting activity. VLPs have also been shown to stimulate cell-mediated immune responses to Rubella virus and Rubella virus structural proteins, which may be important in providing protective immunity against infection. Therefore, VLPs show good potential for safe vaccine development (Qiu, et al., 1994).
Achieving consistent, high-sensitivity Rubella IgM and IgG detection
Rubella antibody testing is critical for diagnosing recent infections and determining immunity. However, developing accurate IgM and IgG assays is challenging due to the need for high sensitivity, reproducibility, and properly designed antigens that mimic the native virus.
The Native Antigen Company offers a portfolio of Rubella virus antigens and recombinant antibodies specifically developed for IgM and IgG detection in diagnostic assays. These reagents provide the sensitivity, reproducibility, and long-term supply security required for serology testing, assay validation, and diagnostic assay development.
New candidate of choice for diagnosing and preventing rubella
Discover how The Native Antigen Company is improving infectious disease management and looking ahead to the future at virus-like particle (VLP) vaccines, which offer a scalable and cost-effective alternative to traditional vaccines. Learn how rubella is set to benefit from the advancements of VLP vaccines, as rubella VLPs, developed through recombinant expression in HEK293 cells, offer a reliable solution by mimicking the virus’ structure and self-assembling into high-quality particles. Explore how these VLPs provide researchers with a consistent and efficient tool for vaccine production, research, and diagnostics.
The Native Antigen Company introduces antigen and antibody conjugation kits
Highly scalable kits enable researchers to prepare liquid-stable antigen and antibody conjugates with a wide range of labels including enzymes, fluorochromes, and biotin
The Native Antigen Company introduces Rubella virus-like particles
New Rubella VLPs promise a reliable source of reagents for consistency in immunoassay development and manufacturing