Monocyte Distribution Width Sepsis Risk Marker
Host response biomarker of sepsis risk

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
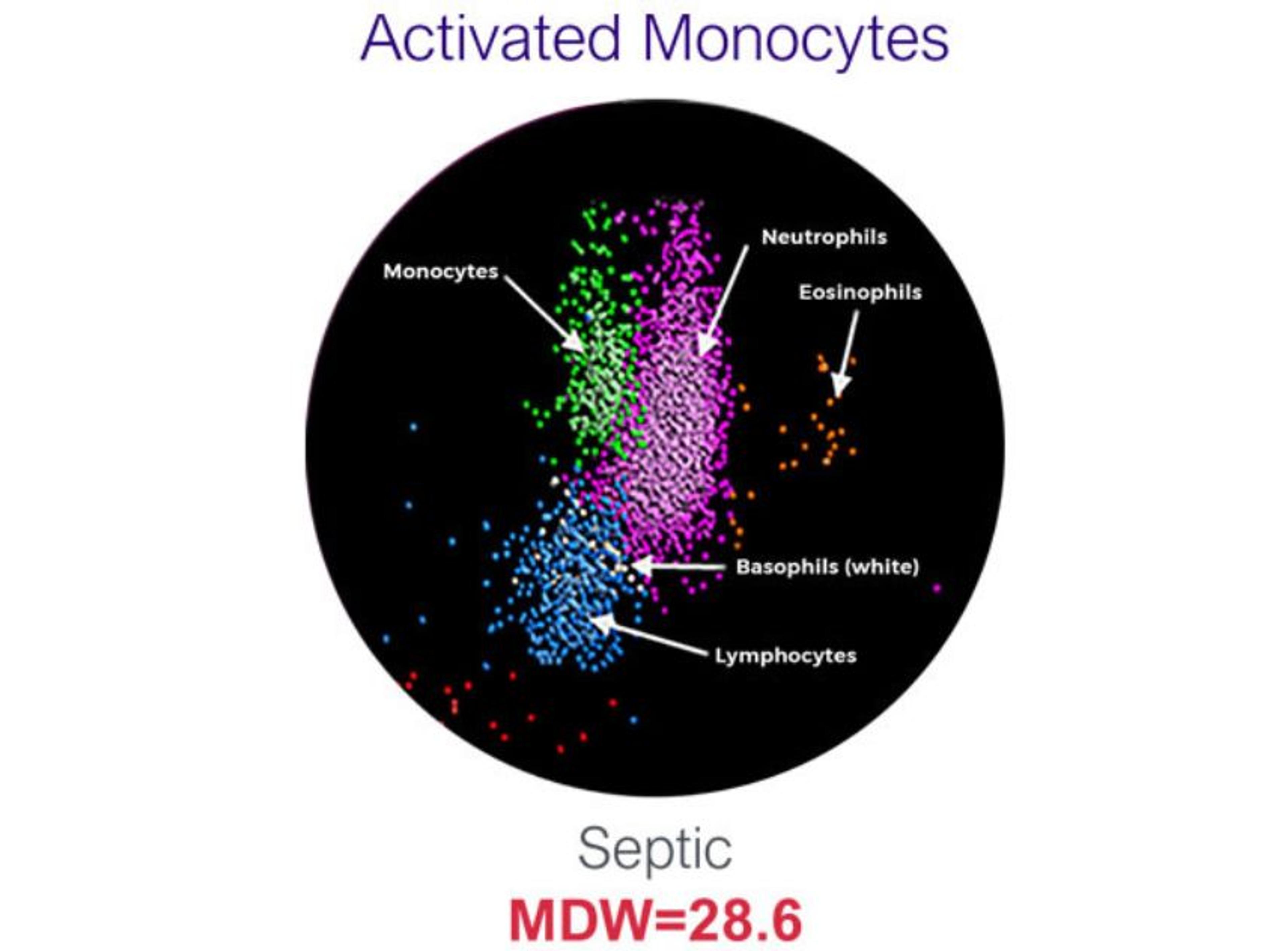
Monocyte Distribution Width (MDW) is the only regulatory-cleared hematological biomarker that helps to establish risk of severe infection and sepsis in adult patients in the emergency department. MDW is a measure of increased morphological variability of monocytes in response to bacterial, viral or fungal infections.
MDW is reported automatically as part of a routine CBC with Differential test using the Early Sepsis Indicator (ESId) application*, which enables automatic reporting with no workflow changes or need to order an additional test.
What Makes MDW Unique?
- Monocyte Activation is a common physiological signal in risk of severe infection and sepsis
- MDW is backed by rigorously designed patient trials and several rounds of in-depth review to meet the standards required for regulatory-clearance and CE mark
- MDW has been studied for the past decade with more than 60 peer-review publications and 30,000+ patients, confirming the robustness and reproducibility of MDW performance in different patient populations
- MDW helps reduce diagnostic uncertainty. It is available early to enhance the initial risk assessment and potentially shift your clinical decision point helping clinicians escalate or de-escalate care
Early sepsis detection: The critical role of biomarkers
In this video, Dr. Elena Sukhacheva from Beckman Coulter discusses the recently FDA-approved sepsis biomarker that can aid in early sepsis detection. The biomarker is part of a scientific effort to help clinicians worldwide detect sepsis early, which is critical for effective treatment and successful patient recovery. Whilst there is no single best biomarker for sepsis, combining biomarkers with clinical information from the hospital along with the use of diagnostic algorithms can help identify patients who need further differential diagnosis. Beckman Coulter is working towards biomarkers with high levels of sensitivity and specificity, that are cost-effective and can be made available for the majority of patients.
This video was filmed at ECCMID 2023, Copenhagen.
Beckman Coulter achieves CE Mark for its MDW-SARS CoV-2 application as an aid in identifying patients with risk of COVID-19
The MDW-SARS CoV-2 application has shown to aid A&E patients