bioZen WidePore C4
Intact core-shell reversed phase column

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
bioZen™ 2.6 µm WidePore C4 is an intact core-shell reversed phase column that provides good peak shape and selectivity for both intact monoclonal antibodies (mAbs) and subunit analyses. Its robust surface grafting and optimal particle and pore size morphology ensures high reproducibility for analytical methods.
bioZen WidePore C4 is designed for better resolution of large biologics using:
- Core-Shell Advantage for High Efficiency
- Optimal Pore Size for Better Separation
- Bioinert Hardware for Improved Peak Shape
- Robust Surface Chemistry for Improved Column Stability
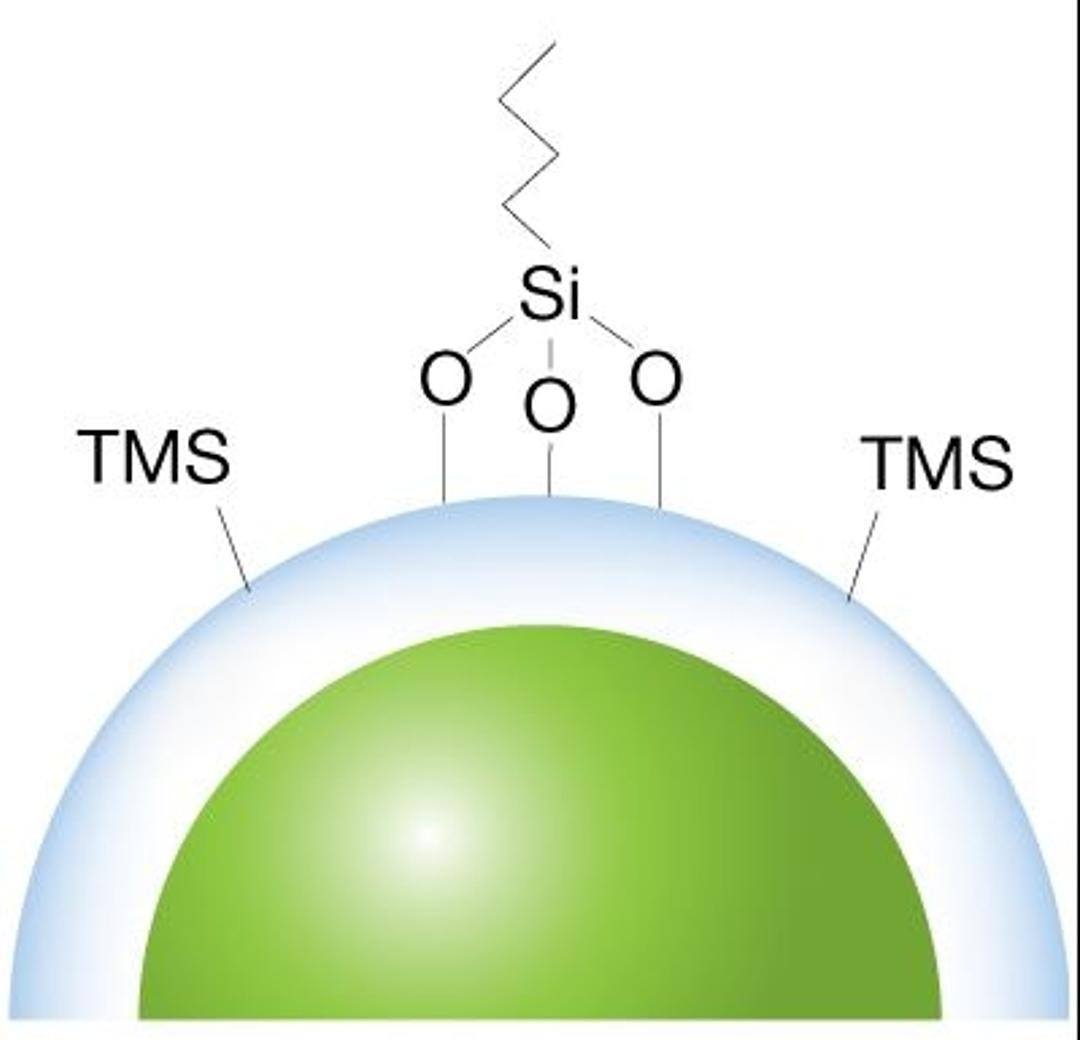
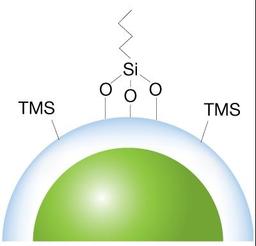
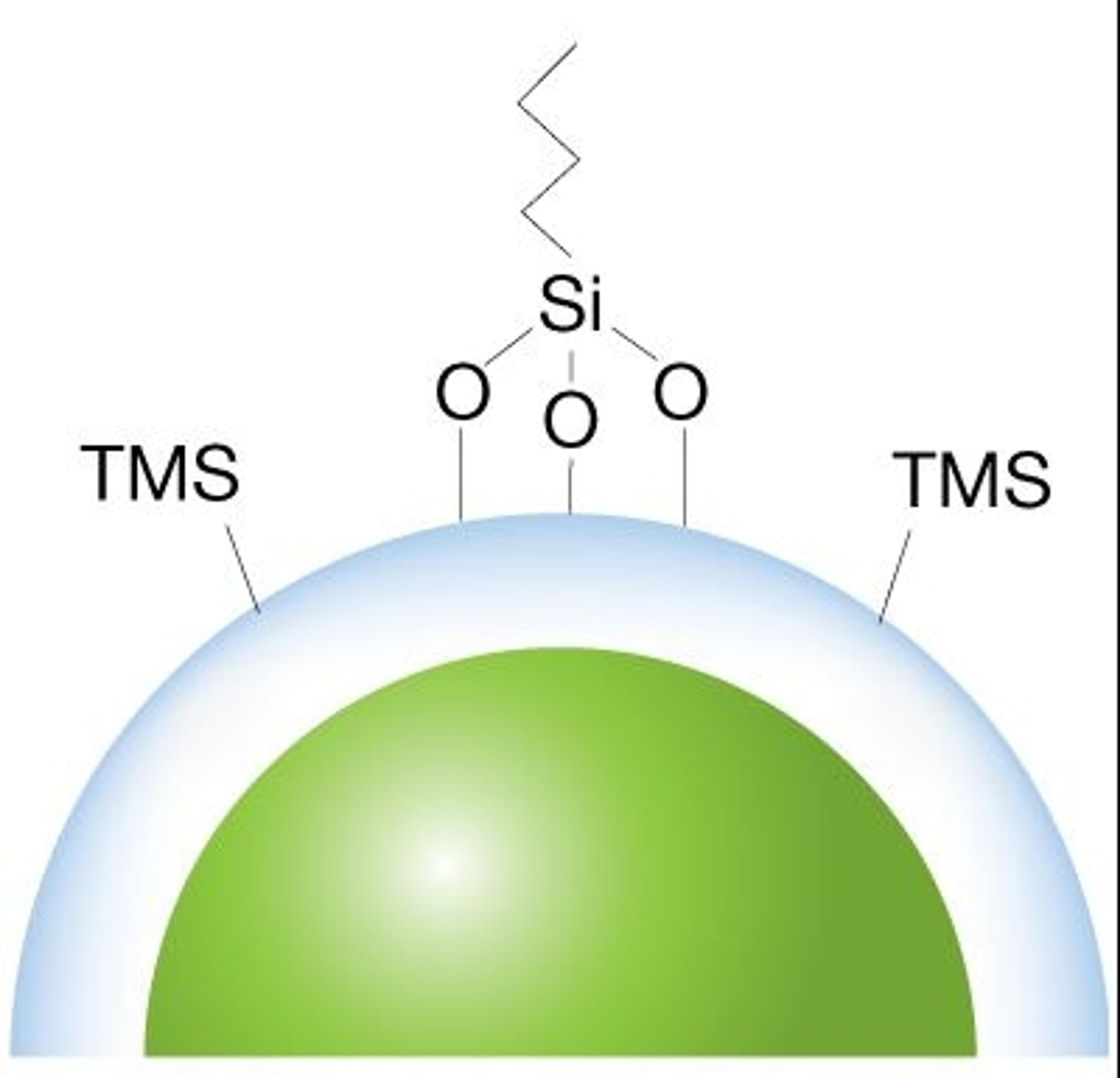
Core-shell advantage
For an increase in resolution, along with faster and more consistent results, our scientists create a durable, homogeneous spherical porous shell uniformly grown on a solid silica core.
Controlled wide pore technology
A wider pore LC column (~400Å) allows for better separation of large biologics and with a controlled manufacturing process, bioZen WidePore introduces a new solution for analyzing intact and subunit mAbs.
Brochures
Achieve better resolution for large biologics
This product brochure by Phenomenex demonstrates a new solution for your intact mAbs and subunit analysis using bioZen™.
Tandem digestion of monoclonal antibodies using novel cysteine proteases
In this application note, Phenomenex discusses the use of cysteine proteases is an increasingly useful sample preparation technique to gain insight on mAb heterogeneity in conjunction with a wide pore core-shell LC column, bioZen™ 2.6 µm WidePore C4.
Analysis of IdeS digested monoclonal antibody fragments
In this application note, Phenomenex discusses the analysis of antibody fragments can provide key insights into sample heterogeneity. This application note highlights how the bioZen™ 2.6 µm WidePore C4 LC column can be used to separate variants such as lysine and deamidated variants by digestion using a site-specific cysteine protease.
Purity analysis for intact monoclonal antibodies and fragments
Monoclonal antibodies (mAbs) are well-established therapeutics, with a variety of different analytical methods associated with purity analysis. An analytical technique common for mAb purity is reversed phase LC (RPLC). This is a primary technique utilized for mAbs because of its relatively short analysis time, with high resolution, and its ability to separate hydrophobic variants such as oxidation, glycoforms, and lysine variants. As such, intact reversed phase methods can be implemented during lead selection and optimization, and on to downstream, including stability-indicating methods and quality control lot release. One particularly appealing aspect of analysis by intact reversed phase, is the capability of implementing a “platform” method.
Analysis of IgdE digested monoclonal antibody fragments
In this application note, Phenomenex discusses how the analysis of antibody fragments can provide key insights into sample heterogeneity. By using the cysteine protease IgdE, further characterization of the Fab region can be obtained.
Method optimization of purity analysis of a Fc-Fusion protein by reversed phase HPLC
A primary analytical technique for purity for recombinant proteins is reversed phase LC (RPLC). This is a simple, robust methodology with a relatively short analysis time, with high resolution, its ability to separate hydrophobic variants such as oxidation, glycoforms, and clipping. As such, RPLC is a common analytical approach for monoclonal antibodies (mAbs), especially considering it can be implemented at both intact and subunit levels without extensive method optimization when combined with a bioZen™ 2.6 µm WidePore C4 LC column.
Optimization of a gradient profile on reversed phase separations of monoclonal antibodies
The analysis of intact monoclonal antibodies by reversed phase HPLC is a common technique for assessing protein variation, such as clipping and heterogeneity due to post-translational modifications. In this technical note, Phenomenex demonstrates how utilizing a bioZen™ 2.6 µm WidePore C4 LC column allows for better resolution and reproducibility through a wider pore core shell particle combined with bioinert hardware. In order to optimize the method, the gradient profile is analyzed.
Effect of temperature on reversed phase separations of monoclonal antibodies
In this technical note, Phenomenex demonstrates using bioZen™ 2.6 µm WidePore C4 LC Columns how increases in temperature can lead to improvements in peak shape in large molecule reversed phase LC methods.
Effect of flow rate on reversed phase separations of monoclonal antibodies
In this technical note, Phenomenex analyzes how changes in the flow rate effect the overall analysis when using a bioZen WidePore C4 column. It is shown that when choosing a flow rate, it should be selected for good peak shapes and to increase the throughput of the application.
Optimization of chromatography for intact mass analysis of monoclonal antibodies
In this application note, temperature, gradient program, and flow rates are explored and optimized for intact mass applications for mAbs when using bioZen WidePore C4 a core-shell wide pore column with bioTi hardware.