AdvanceBio HIC
Agilent AdvanceBio HIC columns deliver robust and reproducible high-resolution separations of native proteins at the intact level.
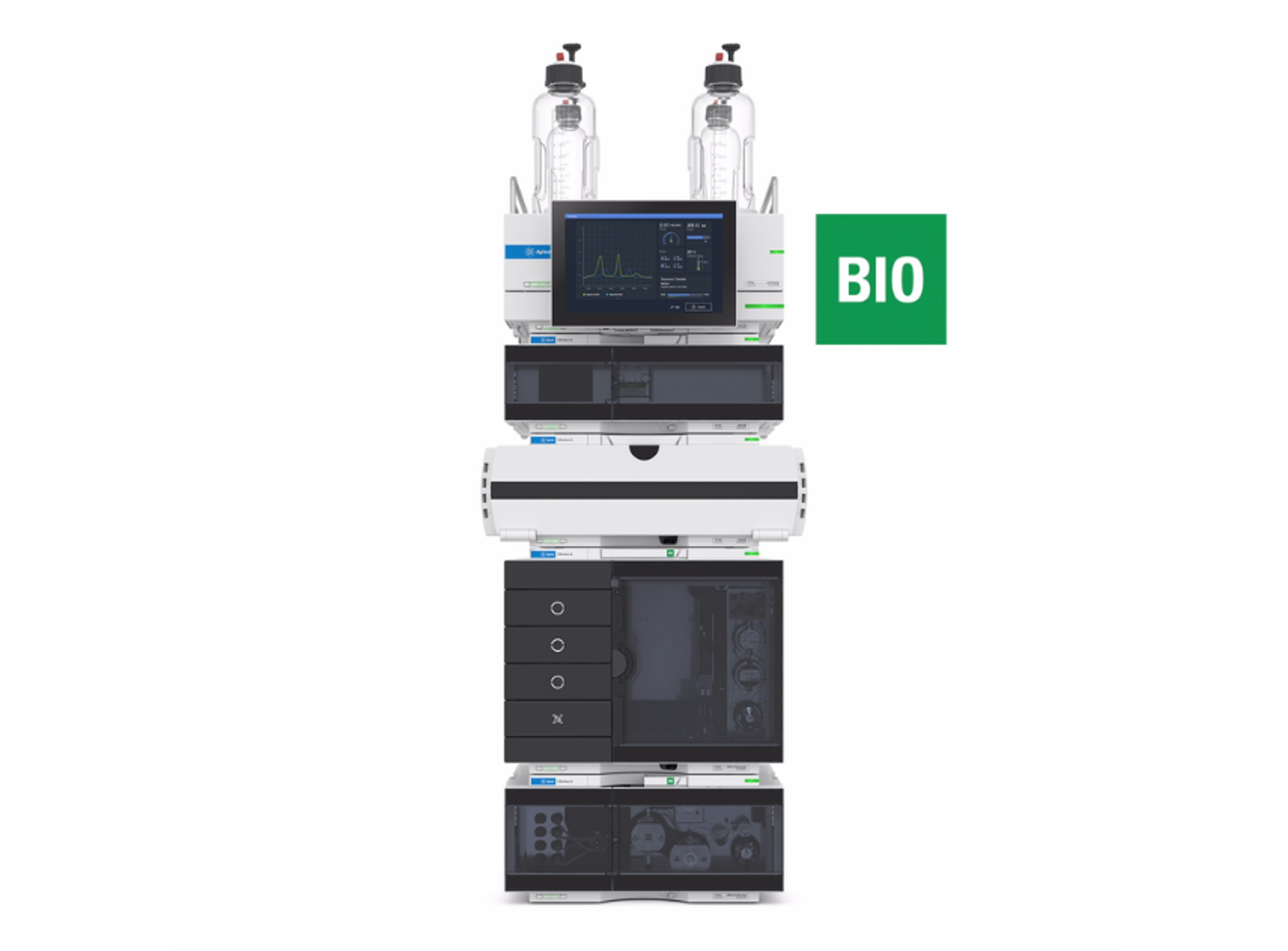
Agilent AdvanceBio HIC columns deliver robust and reproducible high-resolution separations of native proteins at the intact level. Built using the capabilities of ZORBAX fully porous particles and proprietary bonding technology, AdvanceBio HIC columns provide new levels of hydrophobicity and selectivity to address particularly challenging molecules such as monoclonal antibodies (mAbs), antibody drug conjugates (ADCs), and other recombinant proteins. Together with the Agilent 1260 Infinity II bio-inert LC system, AdvanceBio HIC columns provide uncompromised performance and data consistency during characterization and validation.
Features:
- Optimized selectivity – Ideal for mAb oxidation and ADC DAR ratios
- Single chemistry – Reduces the need for multiple-column screening for different CQAs
- Enhanced robustness – Improved column lifetime for ultimate confidence in your data
- Proven performance – Every batch of media is tested with NIST mAb
- High quality – Each column is individually tested to ensure packing efficiency
- Greater productivity – Shorter columns reduce analysis time while maintaining separation performance
Charge variant and aggregation analysis of innovator and biosimilars of rituximab
Monoclonal antibodies are an important class of biomolecules used for the treatment of various diseases. Biosimilars, the copy versions of an innovator molecule, need to be characterized in detail for their critical quality attributes such as aggregates and charge variants. This application note compares two rituximab biosimilars from different manufacturers to the innovator for their aggregation and charge variant profiles by following two analytical workflows using Agilent 1260 Infinity II Bio-Inert LC and Agilent AdvancedBio columns.
Separation of deamidated peptides with an Agilent AdvanceBio Peptide Plus column
Deamidations of glutamine and asparagine are among the most common degradations affecting proteins. This application note demonstrates that a charged surface C18 column greatly enhances selectivity for deamidated variants of peptides versus their unmodified forms when compared to a traditional C18 column.