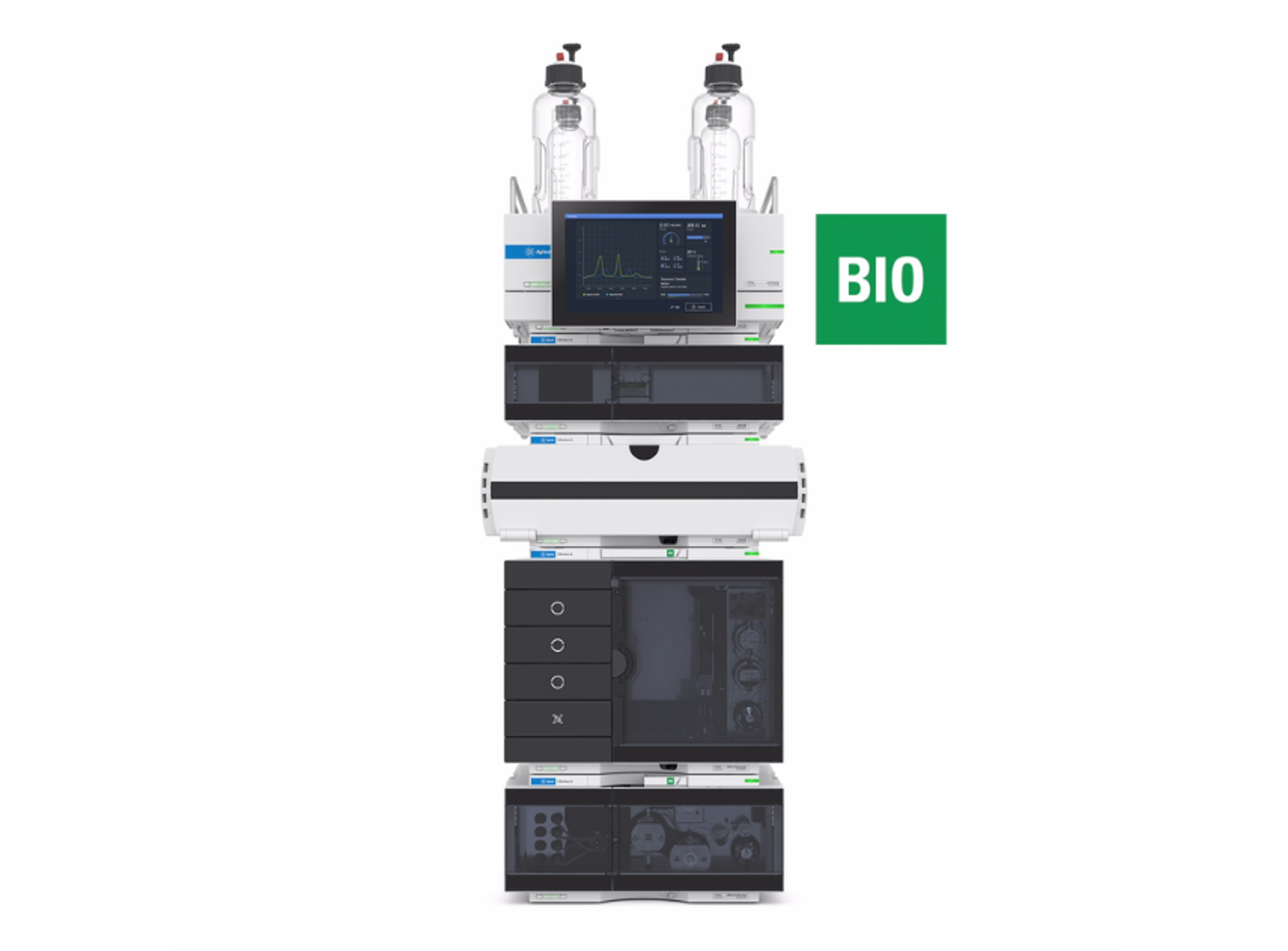
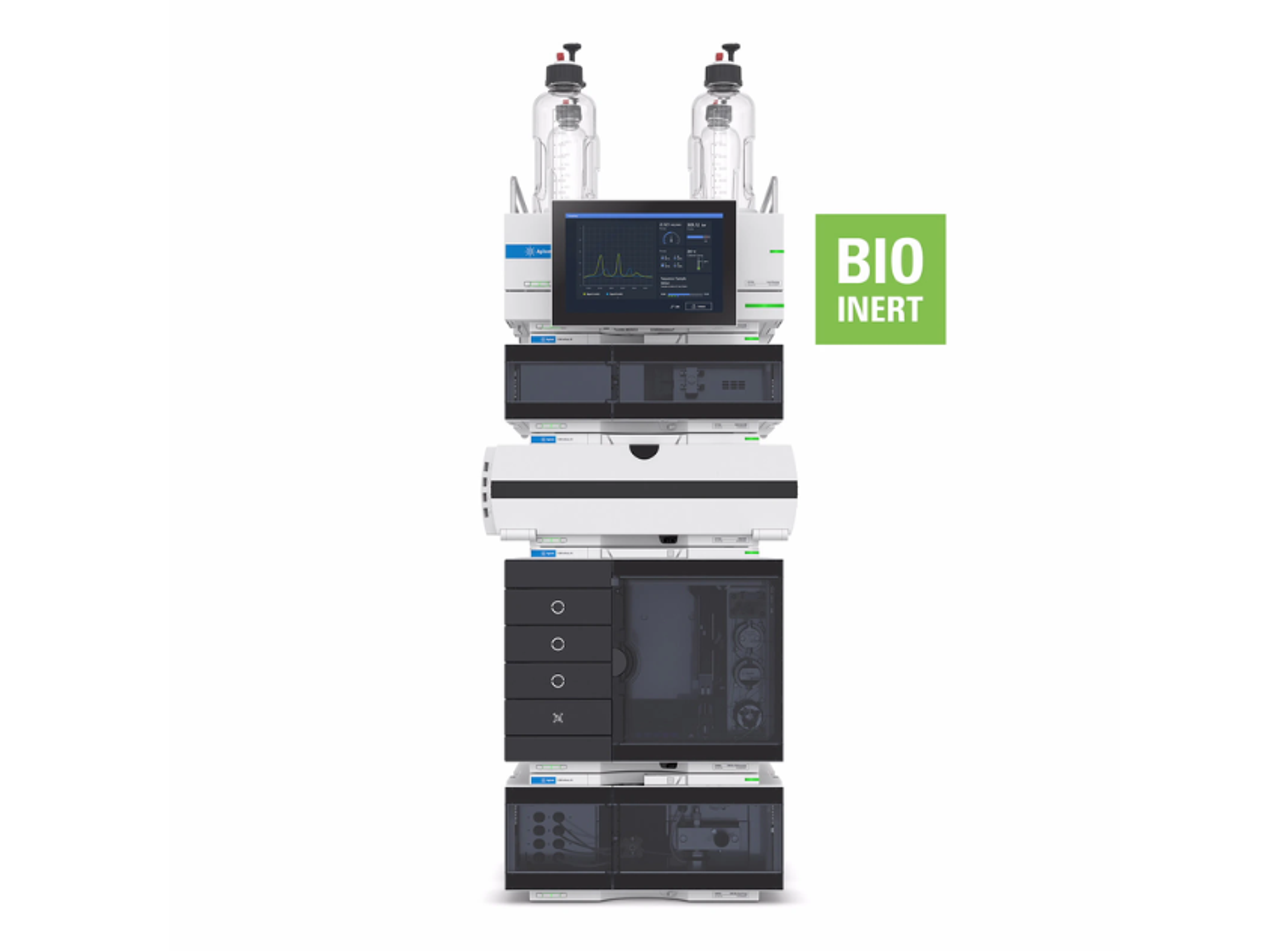
6545XT AdvanceBio LC/Q-TOF
The Agilent 6545XT AdvanceBio LC/Q-TOF system is designed to handle multiple workflows in biopharmaceutical characterization. When you need to access the most detailed information at the intact protein level, automatically confirm protein sequence at the peptide level, and confidently understand post-translational modifications, the 6545XT AdvanceBio Q-TOF delivers results you can trust.
Good and reliant instrument for Biopharma applications
Mass Spectrometry
Good software for relatively easy operation (for a mass spectrometer). Software to analyze biopharma data is unflexible, and has room to improve. After salescare by Agilent is good, and online support is fast (within 24h). The instrument delivers consistent results, calibration is quite stable but sensitive to temperature shifts. Good value for money.
Review Date: 2 Feb 2024 | Agilent Technologies
Great instrument, thank you so much
Proteomics and high resolution PTM analysis
Reproducible results and highly robust. The preventive maintenance annually helps in smooth functioning of the instrument. The customer care is amazing with software updates, application notes and quick virtual call for troubleshooting. The service calls help resolve all the minor issues too.
Review Date: 23 May 2022 | Agilent Technologies
we are happy getting good result for proteomics research
Bio
So good product and excellent service
Review Date: 5 May 2022 | Agilent Technologies
Perfect balance of power and user friendly interface!
Analytical Chemistry
This LC/Q-TOF is very simple to use for a beginner user. Although there is a very simple and user-friendly interface, the system has incredible power and resolution. We use this for intact mass of monoclonal antibodies and it is unmatched. We have 3 other mass spectrometers in the lab and this is our routine analyzer.
Review Date: 30 Nov 2021 | Agilent Technologies
It's great, Value for money.
Drug discovery
Better than instruments available from other manufacturers from the market.
Review Date: 18 Apr 2018 | Agilent Technologies
Development of Omics is wonderful
In development Omics
Agilent Technologies' 6545XT AdvanceBio LC/Q-TOF MS System is - and has software that is - very important for my work
Review Date: 17 Apr 2018 | Agilent Technologies
It's nice and I like the technology
For characterizing the compounds from the LC to separate them using TOF
Easy to use, Easy for data analysis. Results are satisfactory
Review Date: 17 Apr 2018 | Agilent Technologies
Great combination of technology with cost.
Research / discovery
Useful product of high quality (as all products from Agilent).
Review Date: 16 Apr 2018 | Agilent Technologies
Great Research....Wonderful Results......
Characterization of Proteins and Peptides
The Agilent 6545XT AdvanceBio LC/Q-TOF system is designed to handle multiple workflows in biopharmaceutical characterization. When you need to access the most detailed information at the intact protein level, automatically confirm protein sequence at the peptide level, and confidently understand post-translational modifications, the 6545XT AdvanceBio Q-TOF delivers results you can trust.
Review Date: 16 Apr 2018 | Agilent Technologies
Great instrument
Laboratory equipment
high-quality, and easy to use. I recommend it to others.
Review Date: 22 Mar 2018 | Agilent Technologies
The Agilent 6545XT AdvanceBio LC/Q-TOF system is designed to handle multiple workflows in biopharmaceutical characterization. When you need to access the most detailed information at the intact protein level, automatically confirm protein sequence at the peptide level, and confidently understand post-translational modifications, the 6545XT AdvanceBio Q-TOF delivers results you can trust.
Features:
- Get up and running fast with quick-start methods that include Agilent AdvanceBio LC columns for intact protein and peptide workflows.
- Characterize both major and minor intact protein variations with an automated data workflow featuring MassHunter BioConfirm software.
- Adapt your instrument to your needs with SWARM autotune to ensure optimum performance with no manual adjustment. New large molecule setting provides sub-nanogram detection levels for intact glycosylated monoclonal antibodies.
- Be sure of your results with mass accuracies within 10 ppm for glycosylated intact proteins.
- Dig deeper into your digested samples with the powerful new iterative MS/MS feature.
- Maximize your uptime with a system tested to perform through thousands of protein injections without degradation of performance.
- Minimize maintenance delays with vent-free cleaning of entrance optics.
An improved workflow for profiling and quantitation of sialic acids in biotherapeutics
This application note describes the use of the Agilent AdvanceBio Sialic Acid profiling and quantitation kit to profile and perform absolute quantitation of sialic acids present in biotherapeutic glycoproteins as well as the NISTmAb.
In-depth peptide mapping with iterative MS/MS acquisition on the Agilent 6545XT AdvanceBio LC/Q TOF
Therapeutic monoclonal antibodies (mAbs) represent one of the fastest growing classes of protein-based drugs in the biopharmaceutical industry. Here, Agilent Technologies presents an integrated workflow for peptide mapping of a mAb, from automated sample preparation to data analysis, featuring an Agilent AssayMAP Bravo liquid-handling robot, the Agilent 1290 Infinity II LC system, the Agilent 6545XT AdvanceBio Q-TOF, and the Agilent MassHunter BioConfirm B.09 software.
N-glycan analysis: Better together
In this brochure, Agilent Technologies introduces the Agilent and ProZyme sample preparation workflows that are designed to help push your glycoprotein analysis to new levels of performance.
Rapid high-throughput profiling and quantitation of sialic acids in biotherapeutics
This poster presents a new high-throughput workflow based on a 96-well plate format for the release, labeling, and analysis of sialic acids from therapeutic glycoproteins using rituximab, etanercept, and NISTmAb as examples. Sialic acid residues are released then labeled with 1,2-diamino-4,5methylenedioxybenzene (DMB) in a two-step procedure.
LC/MS technologies have become a choice for HCP analysis
In this application note, Agilent Technologies present a study to demonstrate an HCP analysis workflow that includes the AssayMAP Bravo platform for automated sample preparation, the 6545XT AdvanceBio LC/Q TOF for LC-MS/MS analysis, and the vendor-neutral software from proteinmetrics for data analysis.
Integrate your workflows for quantitative analysis of monoclonal antibodies in a serum matrix
In this application note, Agilent Technologies describe a method for the quantitative analysis of intact monoclonal antibodies (mAbs) from a biological matrix. The workflow is comprised of an Agilent AssayMAP Bravo platform coupled with an Agilent 6545XT AdvanceBio LC/Q-TOF.
Integrated Workflow for Intact and Subunits of Monoclonal Antibody Accurate Mass Measurements
Monoclonal antibody (mAb) based entities represent a rapidly growing class of biologics that require extensive characterization to obtain approval for clinical trials and subsequent market release. Accurate mass measurement is a challenging step in the analytical characterization of antibodies because of their large size and the presence of post-translational modifications such as glycosylation. These characteristics also make determining the location of modifications more complex.
Agilent SWARM Auto-tune for LC/Q-TOF innovation minute
In this innovation minute, learn how to get expert results specific to your application every time using the Agilent SWARM Auto-tune for LC/Q-TOF.
Designed for biopharma: The Agilent 6545XT AdvanceBio LC/Q-TOF System
In this video, discover how the Agilent 6545XT AdvanceBio LC/Q-TOF system is designed to handle multiple workflows in biopharmaceutical characterization.