Advancing Cancer Research Using High Throughput Screening in Cancer Cell Lines
Learn about cutting-edge drug discovery techniques at Southern Methodist University, Dallas
14 Dec 2016
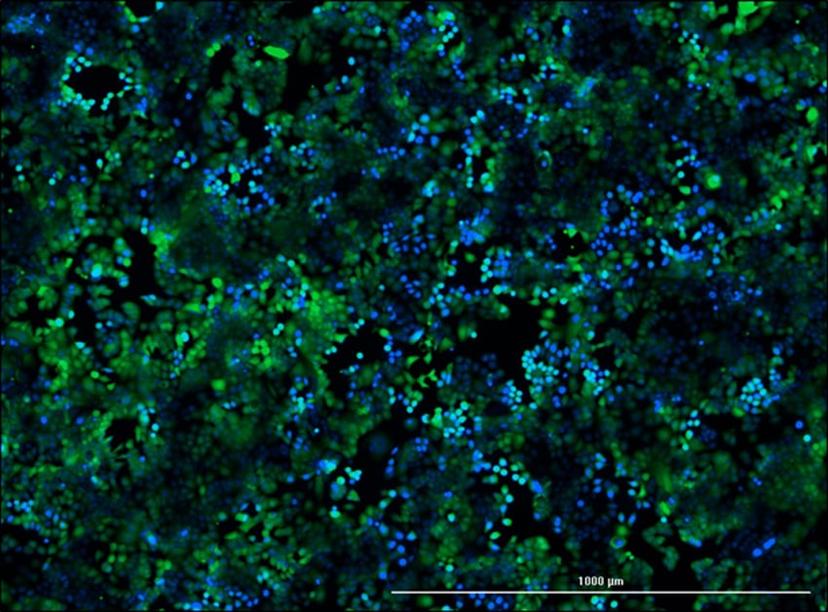
Mitoxantrone resistant breast cancer cell line MCF-7 M100 stained with calcein (Green) and Hoechst 33342 (Blue).
Amila Nanayakkara, a researcher in the laboratory of Dr Pia Vogel and Dr John Wise in the Department of Biology at the Southern Methodist University of Dallas, spoke to SelectScience® about the lab’s techniques and technologies and how they are used to advance their cancer research.
This multidisciplinary lab is focused on drug discovery and drug delivery approaches, including the development of improved cancer treatments. One current project is aimed at targeting two proteins, known to have a major role in mediating multidrug resistance to cancer medications. These two transporters, P-glycoprotein (P-gp) and breast cancer resistance protein (BCRP) can pump chemotherapeutics from cells and there are currently no clinically approved drugs available to inhibit these proteins and prevent drug resistance. The Vogel-Wise lab uses computational modeling to screen candidate molecules for inhibition of these two transporters. Mr Nanayakkara’s project involves in vitro screening of hits from the computational model, using cancer cell lines. Utilizing this approach the group have identified several compounds that are now being further investigated for potential therapeutic benefits for cancer.
High throughput screening technology
The screening method uses cell survival assays to assess the efficacy of chemotherapy in the presence of the candidate compounds. In addition to the conventional 2D cell culture methods, the lab also use 3D cancer spheroids to screen the candidate inhibitors. The group measure the accumulation of chemotherapeutics in 96 well plates using the Cytation 5 Cell Imaging Multi-Mode Reader from BioTek Instruments, Inc. to detect and image fluorescent markers. Mr Nanayakkara explains, “The ability to use conventional 96 well plates for both fluorescence detection and imaging enabled us to screen large numbers of inhibitors in a single experiment.” The group also use the Cytation 5 for kinetic studies of drug accumulation, which enables identification of the most efficient inhibitors. These studies are facilitated by Gen5 Data Microplate Reader and Imager Software, which Mr Nanayakkara states, “is very useful for designing automated experiments.” Automated imaging also enables real time visualization of drug accumulation, in both 2D and 3D cultures. Mr Nanayakkara further comments on the versatility of the plate reader, “we have also used the Cytation 5 for other types of assays. Since the Cytation 5 is compatible with different platforms, we have even used glass slides, with fixed cells to obtain higher magnification fluorescent images.” Mr Nanayakkara remarks, “I would highly recommend the Cytation 5 as a high throughput device with multimode actions, as it helped us to obtain critical data over a short time period.”
Future research
Mr Nanayakkara hopes to be involved in the development of more efficient and faster methods for identification of candidate P-gp and BCRP inhibitors and to continue to work in the field of cancer biology after completion of his PhD. Although significant advances have been made in the fight against cancer, multidrug resistance remains to be solved. Effective, high throughput methods are important for cancer drug screening and Mr Nanayakkara believes that more affordable and larger scale assays will become available in the future. The Vogel-Wise lab is continuing to develop more advanced computational, cell culture and biochemical methods for identification of therapeutic compounds.


