Microglia precision reprogrammed from human iPSCs expand the toolkit for neurodegenerative disease modeling
Discover how a leading contract research organization is utilizing hiPSC-derived microglia to provide more clinically relevant assays
23 Nov 2023

Human microglia derived from iPSCs by precision reprogramming are proving to be a reliable and translatable in vitro tool providing new avenues for neurodegeneration research and therapeutic discovery.
Dr. Christopher Cook is a Team Leader at Concept Life Sciences, a leading contract research organization (CRO) that has successfully incorporated hiPSC-derived microglia into its assay workflows. In this article, Cook explores the benefits of using hiPSC-derived microglia to model neurodegenerative diseases and discusses how partnering with bit.bio has provided the team with confidence in using this new cell type.
Microglia are immune cells within the central nervous system, where they help to maintain brain homeostasis as well as respond to infection, tissue damage, or injury along with other glial cell types. Increasing evidence suggests that neuroinflammation, largely mediated by microglia, contributes to the onset and progression of neurodegenerative diseases such as Alzheimer’s. Microglia are therefore an important therapeutic target in a range of neurodegenerative diseases such as multiple sclerosis, Parkinson’s and Alzheimer’s disease. However, the lack of accurate, consistent, and scalable human microglial cellular models continues to hinder the drug discovery and development process.
The neuroscience team at Concept Life Sciences design and run bespoke microglia assays to support drug development programs from concept to clinic. “We use microglia in our assays to understand the effects of compounds on microglia functions, ranging from inflammatory cytokine response following activation, to other functional responses like phagocytosis and migration. We're also looking to incorporate these cells into more complex model systems,” explains Cook.
Historically, primary rodent microglia and immortalized microglial cell lines have been used to model neurodegenerative diseases during drug development. However, these models present ethical concerns and often lack translational validity, limiting their ability to predict clinical responses accurately. The neuroscience team at Concept Life Sciences recognized the need for a more reliable and human-relevant model system. “ioMicroglia™ from bit.bio offered a solution by providing consistent and reproducible hiPSC-derived microglia cells,” says Cook.
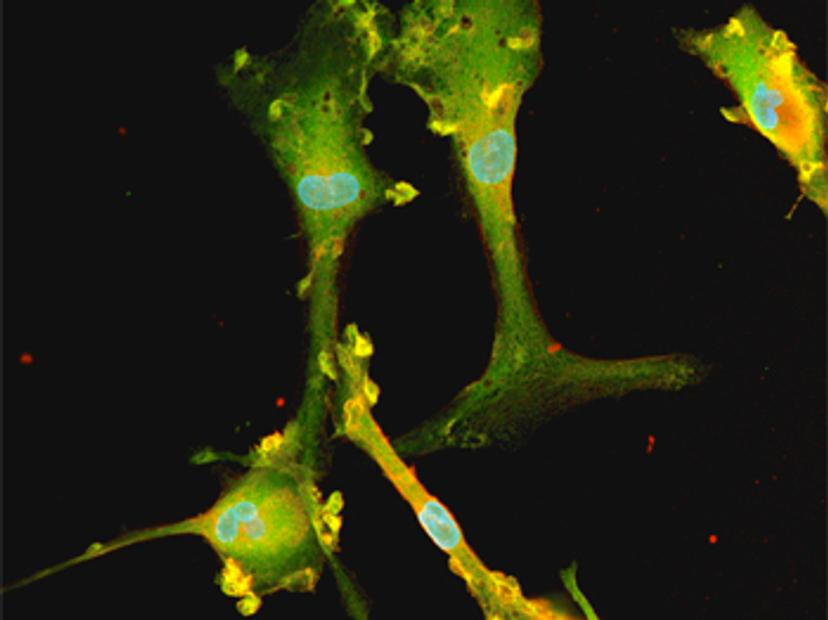
ioMicroglia are human microglial cells generated from iPSCs using bit.bio’s opti-ox™ precision cellular reprogramming technology, which enables the controlled expression of cell-fate-specific transcription factors. This allows the rapid and consistent generation of defined, functional human cells, including microglia, from iPSCs, at scale. ioMicroglia recapitulate key human microglia functions including cytokine secretion and phagocytosis with lot-to-lot consistency, thereby providing a reproducible human-relevant model for neurodegenerative disease research and drug development.
Integrating hiPSC-derived microglia into Concept Life Science’s workflows initially presented some challenges, explains Cook, “Using hiPSC-derived microglia was a relatively new area for us. Handling and treatment of the cells differed from our experience with other cell types we've used previously.” However, the collaboration with the bit.bio technical team proved invaluable, assisting the CRO’s scientists in gaining confidence in working with human microglia. “What we found positive about our experience with ioMicroglia and bit.bio was the level of aftercare that we've been provided,” says Cook. “We welcomed bit.bio’s field application scientist and research scientist on-site to help our team gain confidence with handling the cells.”
An expanded suite of translational models
The adoption of ioMicroglia has enabled Concept Life Sciences to provide more complex human-relevant models to its clients, resulting in a better understanding of how potential therapeutics may behave in vivo in a human. “Having a human in vitro model system that behaves predictably is crucial. One of the key advantages of ioMicroglia is the low inter-lot variability, which ensures consistent assay performance and facilitates long-term screening studies,” says Cook. “Consequently, we can now screen multiple compounds over extended periods and be confident in the reliability and reproducibility of the findings.”
One of the key advantages of ioMicroglia is the low inter-batch variability, which ensures consistent assay performance and facilitates long-term screening studies.
Dr. Christopher Cook
Concept Life Sciences
The CRO’s research efforts with ioMicroglia have yielded promising results. “One of our recent aims has been the optimization of phagocytosis assays, and we have successfully used ioMicroglia to test the effect of different pro- and anti-inflammatory stimuli on the cells' phagocytic activity,” says Cook. “By comparing ioMicroglia with other cell types, such as primary rodent microglia and microglial cell lines, we have identified interesting parallel between these models.” This exciting finding allows the CRO to potentially offer a comprehensive package of different cell types to its clients. “We're excited about the possibility of collaborating with bit.bio on this further to explore other assays, including cytokine secretion and activation assays,” explains Cook.
Future outlooks for drug development services
The reliability and translational validity of ioMicroglia means the cells are providing new avenues for understanding neurodegeneration and developing effective treatments. The neuroscience team at Concept Life Sciences has gained more confidence in the predictive capabilities of their assays by incorporating ioMicroglia into their workflows and by leveraging the expertise of bit.bio.
Looking ahead, the team is focused on expanding its capabilities with hiPSC-derived cells such as ioMicroglia. “We want to begin to leverage the potential of these cells not only in simple assays but in more complex models also,” states Cook. “Bringing in defined cells such as ioMicroglia to help ensure our assays are reliable, perform consistently, and as expected, is crucial in enabling us to provide a comprehensive suite of drug development services.”

