Brain organoids and MEA tech reveal new epilepsy mechanisms
Neurological disorder research is evolving from single cells to complex networks thanks to 3D brain cultures and multielectrode arrays
26 Mar 2026
Editorial article

Prof. Robert Halliwell trains student pharmacists on neurological and psychiatric disorders and what he describes as the “ever expanding array” of medicines to treat these debilitating conditions. He is also a member of the medical ethics committee at St Joseph’s Medical Center in Stockton, CA and an advisor on mental health for the Wellness Center of San Joaquin County.
Epilepsy is a complex and chronic neurological disorder that affects around 50 million people worldwide1, and about one in five people with epilepsy have intractable seizures that do not respond to treatment2. Traditionally, epilepsy research has relied heavily on animal models, with a focus on finding agents to inhibit these seizures. Because the underlying pathophysiology of seizures was not initially well understood and the mechanism of action of the early anti-epilepsy drugs was unknown, serendipity also played a significant role in finding antiseizure medicines. For example, valproic acid (Depakote) was used as a solvent for dissolving other experimental agents, until it proved to be a powerful anticonvulsant itself.
“Today, because of access to detailed neurophysiological and molecular studies, we know much more about the mutated receptors and ion channels that predispose the brain to seizures,” says Dr. Robert Halliwell, Professor of Neuroscience at the Thomas J. Long School of Pharmacy, in the Schools of Pharmacy & Dentistry at the University of the Pacific in Stockton, California. “This also means that drug development is increasingly targeting a diverse range of sites in the brain that are involved in normal and abnormal neurotransmission and in epileptogenesis.”
Brain organoids and the application of human stem cell research
Prof. Halliwell and his team are focused on investigating the properties of neurons and glia derived from human stem cells for disease modelling. “We carry out detailed functional phenotyping of neurons derived from a variety of human stem cells cultured in 2D and more recently 3D human cerebral organoids,” shares Prof. Halliwell. “If these neurons, with their associated glia, are valid and accurate in vitro models of human brain neurons in vivo, this will be a fundamental area of research.”
Prof. Halliwell’s team is exploring the power of iPSC-derived neurons for drug discovery and safety evaluation. “As an example, some of our studies have successfully identified a class of fenamate nonsteroidal anti-inflammatory drugs (NSAIDs) that are powerful anti-seizures agents in both human 2D cultures and 3D organoids,” says Prof. Halliwell3.
Cerebral organoids, which can contain hundreds of thousands to millions of neurons, enable closer study of brain development, dysfunction, and disorders. They allow more accurate visualization of human brain structure and function than animal models or cell monolayers.
“During my Ph.D. and post-doctoral fellowships, I did a lot of single-cell recordings from rodent neurons using patch-clamp techniques, and two-electrode voltage clamp from oocytes expressing recombinant human receptors. These approaches provide very powerful data, but they are technically demanding, slow, and ultimately you’re looking at one cell in isolation.” shares Prof. Halliwell. “Because the brain operates through complex, integrated neural networks and neurological diseases like epilepsy are the result of abnormal neural network activity, the complexity of 3D cerebral organoids allows more accurate in vitro modelling of conditions like epilepsy.”
As organoid systems become increasingly complex, functional electrophysiology provides an important way to validate whether these models truly replicate disease-relevant neural behavior.
Powerful MEA technology for neurological disease research
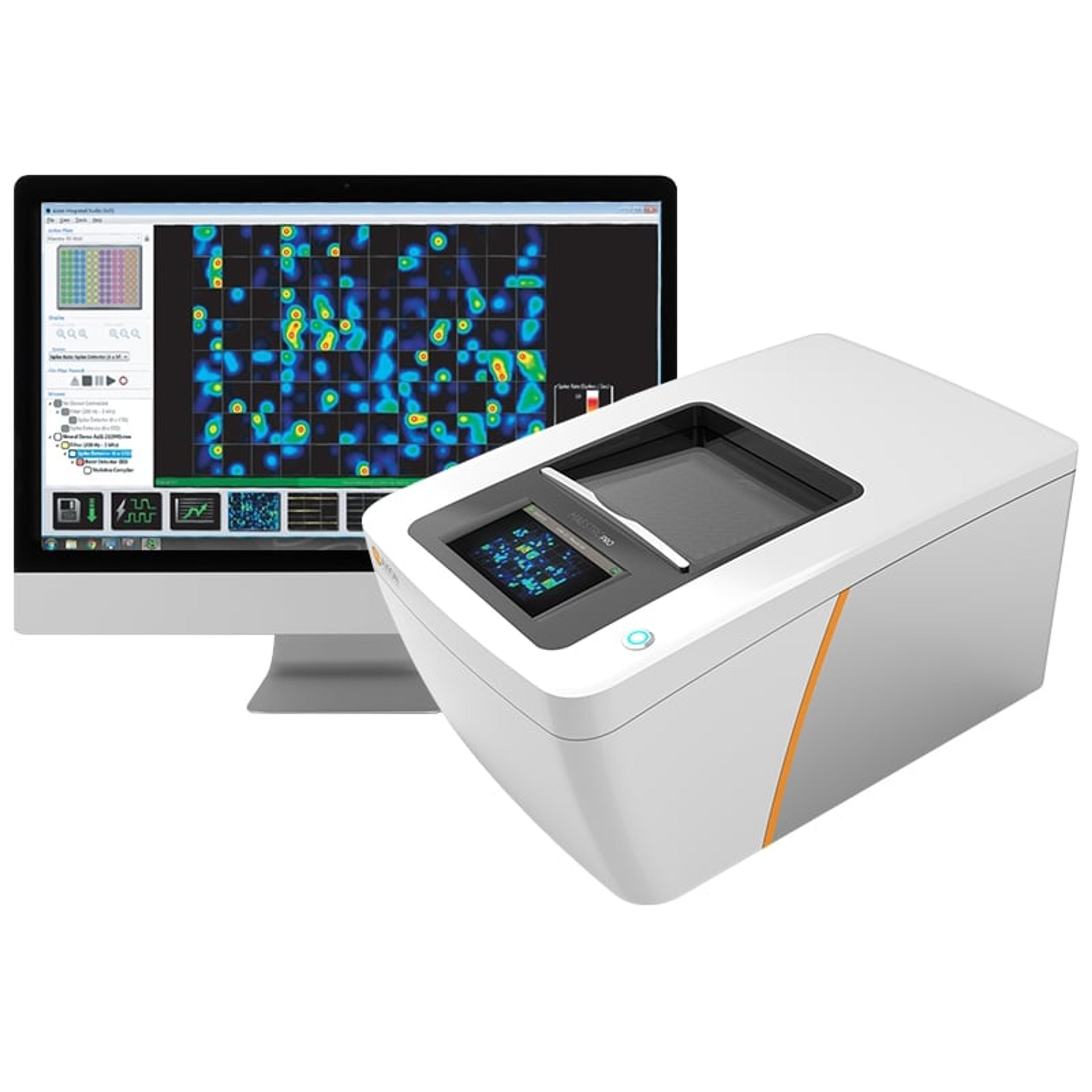
Technologies capable of measuring neural network behavior across entire cultures are therefore becoming essential tools for modern disease-modeling research. Multielectrode arrays (or microelectrode arrays; MEAs) are grids of tens to thousands of microelectrodes that record electrical activity from electrically active cells plated onto the arrays in vitro. The Maestro MEA platform from Axion BioSystems can monitor real-time, label-free, plate-wide neural activity across multiple cultures and track the activity in the same plates over time.

Maestro MEA can record neural activity in up to 96 wells simultaneously and supports a range of organoid-optimized plate formats, giving researchers the flexibility to study both 2D and complex 3D. (Left) A Maestro Pro MEA system records neural activity. An organoid (middle) and activity (right) on a 3DMap MEA plate.
“We used to use patch-clamp techniques to sample different single neurons in 2D cultures every few days to test if they were functional. That was difficult, inefficient and essentially limited to the question of ‘could a single cell fire an action potential or not’,” says Prof. Halliwell. “The Maestro MEA platform has been a significant addition to our lab to support longitudinal neural analysis.”
With the MEA system, Prof. Halliwell and his team can record activity from hundreds of neurons within networks over time. They can also evoke epileptic-like seizures in 2D and 3D neural networks4 and determine the impact of multiple experimental conditions over long time courses.
“Maestro MEA means that we can record cells as often as we want for months, or even years. We can plot the development of their electrophysiological activity and the maturation of these neural networks. Equally importantly, with MEA we can now follow the increasingly complex firing patterns of 2D and 3D synaptic networks. This is very powerful,” explains Prof. Halliwell. “In vitro, we have recorded 2D stem cell-derived neurons for almost 2 years and 3D neural organoids for up to 8 months. Without the combination of MEA technology and the availability of human stem cells, this would have been almost impossible.”
Neurological research of the future
The availability of 3D human stem cell-derived cerebral organoids allows researchers to characterize structural organization, cell composition, and their electrophysiological activity. These models have the potential to reveal immediate and long-term effects of diverse experimental conditions on early neural development, such as genetic mutations or short- or long-term exposure to antiseizure drugs.
“In the longer term, we aim to construct assembloids to represent multiple brain regions and include some form of vascularization to model the astronomically complex human brain on a mini-brain scale,” concludes Prof. Halliwell.
By combining advanced stem cell models with longitudinal electrophysiological measurements of networked cultures, researchers are gaining new insight into how neurological diseases emerge and evolve over time. These insights may ultimately help guide the discovery and evaluation of new therapeutic strategies.
References
1. World Health Organisation (WHO): https://www.who.int/news-room/fact-sheets/detail/epilepsy
2. US National Institute of Neurological Disorders and Stroke: https://www.ninds.nih.gov/health-information/disorders/epilepsy-and-seizures
3. Salmanzadeh H and Halliwell RF (2024) Antiseizure properties of fenamate NSAIDs determined in mature human stem-cell derived neuroglial circuits. Frontiers in Pharmacology, 15:1385523. https://doi.org/10.3389/fphar.2024.1385523
4. Salmanzadeh, H and Halliwell, RF (2025) Human Stem Cell-Derived Neural Organoids for the Discovery of Antiseizure Agents. Receptors 2025, 4, 12. https://doi.org/10.3390/receptors4030012