Achieve reproducible and scalable zebrafish toxicology research with automation
How automated workflows enable consistent, high-throughput zebrafish screening to advance chemical safety science
12 May 2026
Editorial article
Zebrafish, or Danio rerio, are more than just striking aquarium fish. Because of their small size, regenerative capabilities, and fast embryonic development, zebrafish are one of the most commonly used model organisms in developmental and genetic studies and are ideal for high-throughput whole-organism phenotypic screening. They also conveniently share around 70% genetic homology with humans.

Dr. Lisa Truong, Deputy Director of Sinnhuber Aquatic Research Laboratory (SARL) and Associate Professor, Department of Environmental and Molecular Toxicology, Oregon State University
“Within just five days of fertilization, you can assess dozens of developmental and behavioural endpoints simultaneously in a single organism. That whole-animal context captures biological complexity that cell-based assays simply can't replicate, making zebrafish an incredibly powerful bridge between in vitro testing and mammalian models,” shares Dr. Lisa Truong, Deputy Director of Sinnhuber Aquatic Research Laboratory (SARL) and Associate Professor, Department of Environmental and Molecular Toxicology, Oregon State University.
SARL at Oregon State University is the world’s largest specific pathogen-free zebrafish aquatic toxicology facility. Including a 17,000 square foot self-contained fish hatchery for rearing fish, it has a low maintenance system housing over 40,000 fish, including more than 20 transgenic zebrafish lines.
“SARL's mission is to advance chemical safety science through high-throughput biological testing, including high-throughput zebrafish screening,” says Dr. Truong. “Our goal is to generate the kind of robust, reproducible toxicological data that industry and regulatory agencies like the EPA and FDA can rely on to make evidence-based decisions about chemical risk. We have screened approximately 6,000 chemicals to date.”
Dr. Truong’s interest in toxicology and her research focus grew out of curiosity; she is driven to find out why some chemicals cause adverse effects while others have no impact on living systems. “What gets me up in the morning is wanting to understand how the intrinsic properties of a chemical drive toxicity and then using that knowledge to inform chemical safety decisions and protect human health,” says Dr. Truong.

Dr. Robyn Tanguay, Director of the Sinnhuber Aquatic Research Laboratory (SARL) at Oregon State University, pioneered the use of zebrafish for toxicity testing at the facility. This work led to a partnership with Hudson Lab Automation to develop a scalable zebrafish toxicology research platform.
Challenges in chemical safety science
One of the core challenges faced by chemical safety scientists is that existing safety information lives across disconnected platforms. This means labs spend time, money and resources trying to resolve issues when someone else, whether in the same institution or across the world, may have already addressed the problem. There are additional issues with reproducibility as labs often work in silos and methods are allowed to drift.
“As a lab funded primarily by tax dollars, we have a real obligation to ensure the data we generate is robust, rigorous, and reproducible,” explains Dr. Truong. “We work hard to minimize variability study to study, but zebrafish are sensitive; even subtle differences in how an experiment is handled can shift developmental timing and change your answers. In toxicology especially, where our findings can directly influence regulatory decisions and public health, reproducibility is an obligation.”
The power of toxicology automation
The real scientific opportunity in toxicology research lies in uncovering structure-activity relationships and understanding how the structural properties of a molecule can predict its biological behaviour. However, as Dr. Truong and her team started to scale up their research to accommodate this, their labour costs increased and capacity became an issue.
“So much of what we were doing was scheduled and routine. One of the most honest realizations we had was that our limiting factor wasn't scientific — it was finding enough undergraduate workers willing to come in to perform the same repetitive tasks day after day,” states Dr. Truong. “When your ability to advance science is constrained by scheduling and human fatigue, you know it's time to rethink the model.”
Being able to scale with automation frees up scientists’ time for them to focus on higher-value work, while ensuring that the routine, precision-critical steps are handled consistently and reliably every single time. “Automation has given us the consistency we needed,” continues Dr. Truong. “When your science depends on experiments being executed at the right time, and the same way every time, that opportunity practically screams for automation.”
Collaborating to accelerate toxicology
Dr. Truong’s advice for those considering automation is to ensure that there is a step-by-step understanding of the team’s processes, including where the repetitive tasks are, where human error can creep in, and what levels of consistency are expected.
“Automation will pay for itself many times over in consistency, scalability, and the ability to focus your team's talent on the science rather than the logistics, but it’s important to remember that automation is only as good as the clarity you bring to it,” Dr. Truong explains. “You also need to go in with realistic expectations, as implementation takes time and reproducible biological workflows will need refinement.”
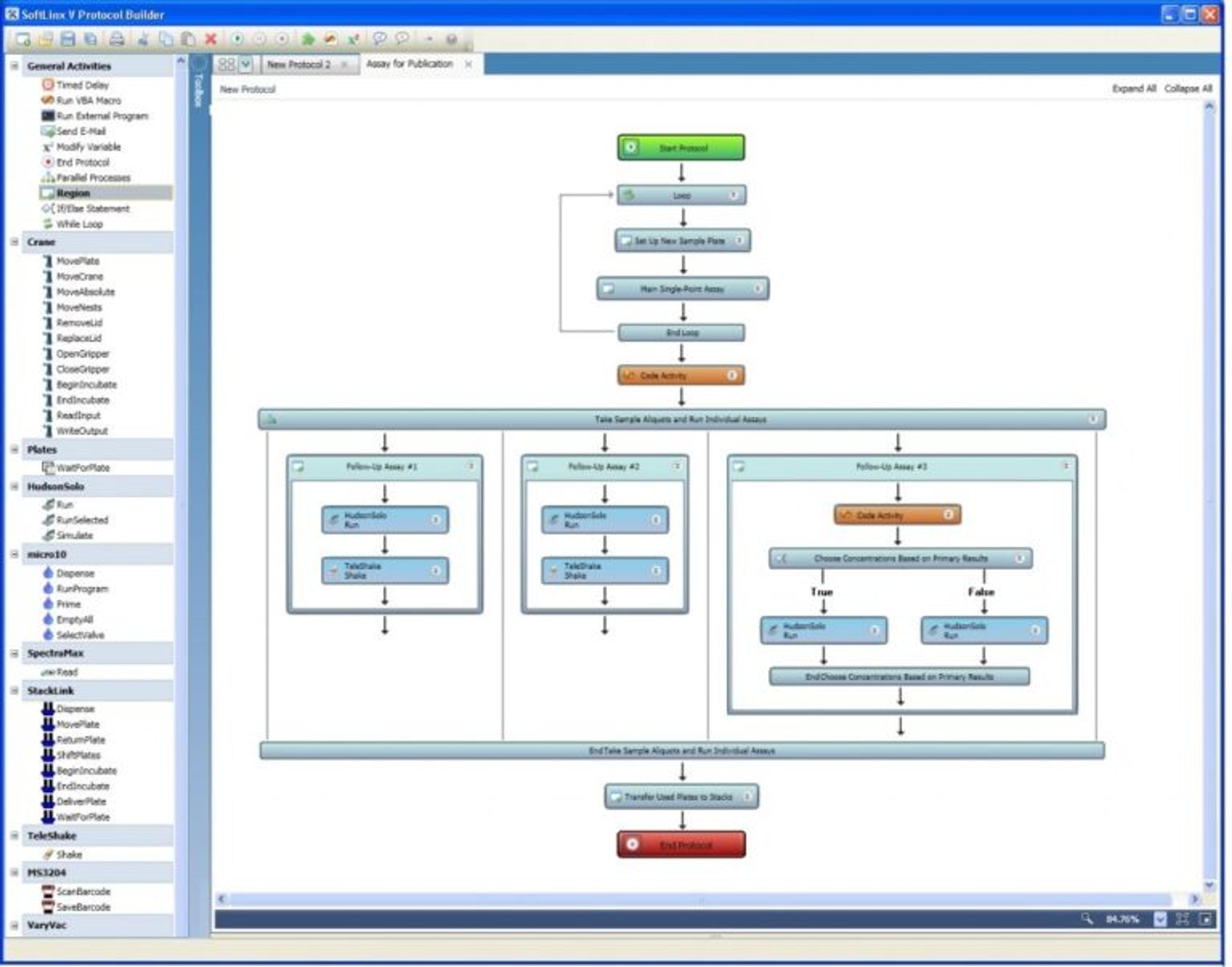
Dr. Truong and her team operate a highly automated workflow orchestrated through a single, unified platform that connects and coordinates eight instruments end to end. The system combines Hudson Lab Automation’s SciClops™ robotic arm for plate handling with Ramona Optics’ MCAM™ AI-powered, multi-camera imaging for high‑throughput zebrafish analysis, all synchronized through Hudson’s SoftLinx™ lab automation software. Acting as the central command layer, SoftLinx manages system-wide timing, sequencing, and device handshakes, enabling disparate technologies to function as one cohesive workflow rather than a collection of standalone instruments. Hudson Lab Automation’s collaborative integration approach ensured seamless interoperability at every step, demonstrating the value of true platform-level orchestration.

PlateCrane™ SciClops™ Robotic Arm is Hudson Lab Automation’s latest robotic arm with extended reach for accessing tough to reach instruments
“What Hudson’s technologies have enabled us to do is incorporate complex, multi-day assays with best-in-class instrumentation and associated software from different third parties into a coordinated, scheduled workflow managed on a single screen,” says Dr. Truong. “We are currently working with Hudson to bring in an additional third-party instrument that will not only expand our automated toxicity screening capabilities, but also increase our precision and accuracy. Working with a partner that is invested in solving scientific problems alongside you is what has helped us to advance the platform rather than being limited by it.”
The future of data-driven science at SARL
The foundation that Dr. Truong and her team have built at SARL, with the automated platform supporting scaling up and reproducible workflows, has put them in a position to do something really meaningful with the data.
“We are integrating AI into our screening platform to give us the ability to start finding patterns that would be impossible to detect manually as we test more chemicals and expand into broader chemical space. We will be able to accelerate our understanding of structure-activity relationships and why certain chemical structures drive toxicity while others don't,” she explains. “This gives us the opportunity to demonstrate, at scale, that the zebrafish is a powerful and practical tool for chemical safety evaluation, and that automation is what makes that feasible. Every chemical we screen, every relationship we uncover, brings us closer to being able to predict toxicity rather than just measure it. That's the future I'm working toward — a world where we have enough data, the right tools, and the computational power to proactively protect human health rather than react to it.”