Calla Lily Clinical Care doses first patients in clinical trial for intravaginal drug delivery platform
FREEDOM study evaluates progesterone delivery using Callavid platform for threatened miscarriage
11 May 2026Industry news
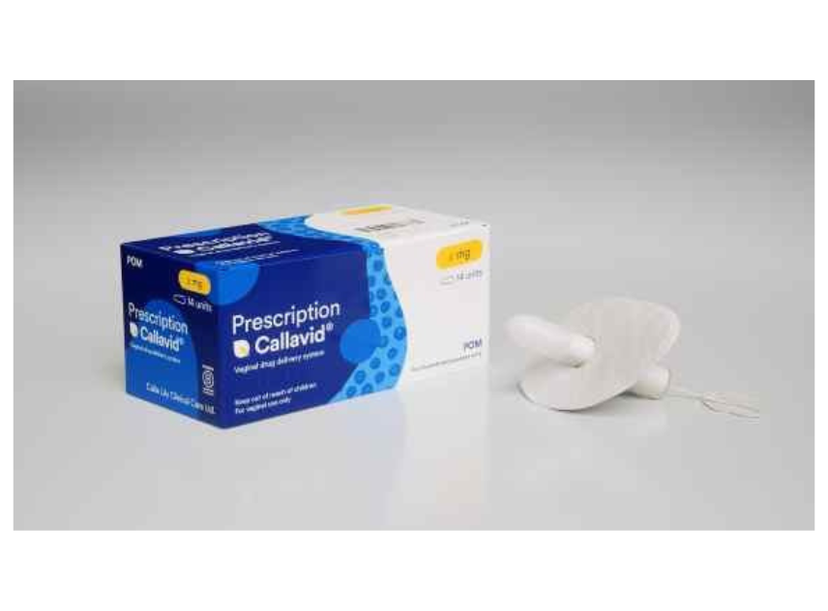
Calla Lily Clinical Care’s Callavid platform for intravaginal drug delivery
Calla Lily Clinical Care, a UK-based women’s health medical technology company, has dosed the first patients in the NIHR-funded FREEDOM clinical trial at University Hospitals Coventry and Warwickshire (UHCW) NHS Trust. The study is evaluating 400 mg progesterone Callavid®, an intravaginal drug-device combination platform designed to improve progesterone delivery for women diagnosed with luteal phase insufficiency and those experiencing threatened miscarriage, with the aim of supporting early pregnancy and reducing the risk of infertility and recurrent miscarriage.
Background on miscarriage and progesterone treatment
The Government’s Renewed Women’s Health Strategy for England cites estimates ranging from 120,000 to 250,000 miscarriages per year in the UK1. The National Institute for Health and Care Excellence (NICE) recommends administering 400 mg micronised progesterone twice daily for women who have had a previous miscarriage and experience bleeding in early pregnancy, a condition known clinically as threatened miscarriage.
Luteal phase insufficiency, where progesterone levels may be too low to sustain early pregnancy, is associated with infertility and recurrent miscarriage. Optimising progesterone delivery is therefore a key focus in reproductive medicine and early pregnancy care.
Callavid intravaginal drug-device combination platform
Callavid is positioned to become the world’s first intravaginal drug-device combination product to support treatment of threatened miscarriage and to provide luteal phase support as part of Assisted Reproductive Technologies, including in vitro fertilization (IVF).
Current progesterone delivery methods typically rely on self-administered pessaries. These can be prone to leakage, uncertain placement and movement during use, which may reduce the efficiency and consistency of drug absorption and potentially compromise delivery of the intended dose. Patients are often advised to lie horizontally for extended periods after each administration.
Callavid’s patented, leak-free, tampon-like design is intended to address these challenges in vaginal therapeutic administration. The platform aims to enable cleaner, more comfortable delivery of medicines and hormones, supporting more reliable dosing and improving the overall treatment experience.
FREEDOM clinical trial design and objectives
The FiRst in human safEty and Ease of use assessment of 400mg progesterone CallaviD in wOMen with luteal phase insufficiency (FREEDOM) study is led by Clinical Chief Investigator Professor Siobhan Quenby MBE, a world-leading authority on miscarriage and preterm birth and an Honorary Consultant at UHCW NHS Trust.
The trial is funded by the National Institute for Health and Care Research (NIHR) and is being run collaboratively with the Trial Management Unit at UHCW. FREEDOM is designed to evaluate the safety, user acceptability and progesterone absorption profile of 400 mg progesterone Callavid in women with luteal phase insufficiency.
By assessing these parameters, the study aims to generate evidence of improved usability in self-administration and to demonstrate the capabilities of the easy-to-use Callavid vaginal delivery platform in maximising adherence and supporting patient wellbeing.
Professor Quenby, said, “Through my clinical practice, I see the difficulties patients face with existing vaginal progesterone products at an already very stressful time. Callavid offers a promising new solution to ensure delivery of the correct progesterone dosage and give women greater confidence in their treatment. There is genuine excitement among both clinicians and patients at the prospect of Callavid progressing into clinical trials.”
References
1. Department of Health and Social Care. Renewed Women’s Health Strategy for England. 2024. Available at: The Renewed Women’s Health Strategy for England
Want the latest science news straight to your inbox? Become a SelectScience member for free today>>
Frequently asked questions
How does Calla Lily Clinical Care’s Callavid intravaginal platform aim to improve progesterone delivery for women with luteal phase insufficiency and threatened miscarriage?
Callavid®, developed by Calla Lily Clinical Care in London, UK, is an intravaginal drug-device combination platform designed to improve progesterone delivery for women diagnosed with luteal phase insufficiency and those experiencing threatened miscarriage. The 400 mg progesterone Callavid product uses a patented, leak-free, tampon-like design intended to overcome limitations of current self-administered vaginal pessaries, such as leakage, uncertain placement and movement during use.
By providing cleaner, more comfortable administration and supporting more reliable dosing and absorption, Callavid aims to better sustain early pregnancy, reduce the risk of infertility and recurrent miscarriage, and enhance the overall treatment experience in reproductive medicine and early pregnancy care.
What is the FREEDOM clinical trial evaluating about the 400 mg progesterone Callavid platform at University Hospitals Coventry and Warwickshire NHS Trust?
The NIHR-funded FREEDOM clinical trial (FiRst in human safEty and Ease of use assessment of 400mg progesterone CallaviD in wOMen with luteal phase insufficiency) at University Hospitals Coventry and Warwickshire (UHCW) NHS Trust is assessing 400 mg progesterone Callavid in women with luteal phase insufficiency. Led by Clinical Chief Investigator Professor Siobhan Quenby MBE, the study is designed to evaluate the safety, user acceptability and progesterone absorption profile of the Callavid intravaginal platform.
By examining these parameters, the trial aims to generate evidence of improved usability in self-administration, demonstrate the platform’s ability to maximise adherence, and support patient wellbeing, positioning Callavid as a differentiated intravaginal delivery modality for women’s health.
Why is optimizing intravaginal progesterone delivery important in the context of miscarriage, IVF and the UK Women’s Health Strategy?
Optimizing intravaginal progesterone delivery is important because luteal phase insufficiency, where progesterone levels may be too low to sustain early pregnancy, is associated with infertility and recurrent miscarriage. In the UK, the Government’s Renewed Women’s Health Strategy for England cites estimates of 120,000 to 250,000 miscarriages per year, and NICE recommends 400 mg micronised progesterone twice daily for women with a previous miscarriage who experience bleeding in early pregnancy (threatened miscarriage).
Current vaginal progesterone treatments, typically pessaries, can be affected by leakage and uncertain placement, potentially compromising dose delivery. Callavid’s tampon-like, leak-free design is intended to provide more consistent progesterone absorption and improved patient experience, supporting early pregnancy, threatened miscarriage treatment and luteal phase support in Assisted Reproductive Technologies such as in vitro fertilisation (IVF), while also offering pharmaceutical companies a differentiated intravaginal delivery modality that could extend the lifecycle of existing drugs.
